93. Construction
Chapter Editors: Knut Ringen, Jane L. Seegal and James L. Weeks
Table of Contents
Tables and Figures
Health, Prevention and Management
Health and Safety Hazards in the Construction Industry
James L. Weeks
Health Risks of Underground Construction Work
Bohuslav Málek
Preventive Health Services in Construction
Pekka Roto
Health and Safety Regulations: The Netherlands Experience
Leen Akkers
Organizational Factors Affecting Health and Safety
Doug J. McVittie
Integrating Prevention and Quality Management
Rudolf Scholbeck
Major Sectors and Their Hazards
Major Sectors
Jeffrey Hinksman
Types of Projects and their Associated Hazards
Jeffrey Hinksman
Trenching
Jack L. Mickle
Tools, Equipment and Materials
Tools
Scott P. Schneider
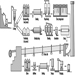
Equipment, Machinery and Material
Hans Göran Linder
Cranes
Francis Hardy
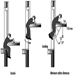
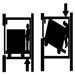
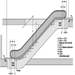
Elevators, Escalators and Hoists
J. Staal and John Quackenbush
Cement and Concrete
L. Prodan and G. Bachofen
Case Studies: Prevention of Occupational Dermatosis among Workers Exposed to Cement Dust
Pekka Roto
Asphalt
John Finklea
Gravel
James L. Weeks
Tables
Click a link below to view the table in the article context.
- Selected construction occupations
- Primary hazards encountered in skilled construction trades
- Occupations excessing standardized mortality & incidence rates
- Value of construction projects in Canada, 1993
- Contractors on industrial/commercial/institutional projects
- Clearance for normal voltage near high-voltage power lines
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Children categories
Asphalt
Asphalts can generally be defined as complex mixtures of chemical compounds of high molecular weight, predominantly asphaltenes, cyclic hydrocarbons (aromatic or naphthenic) and a lesser quantity of saturated components of low chemical reactivity. The chemical composition of asphalts depends both on the original crude oil and on the process used during refining. Asphalts are predominantly derived from crude oils, especially heavier residue crude oil. Asphalt also occurs as a natural deposit, where it is usually the residue resulting from the evaporation and oxidation of liquid petroleum. Such deposits have been found in California, China, the Russian Federation, Switzerland, Trinidad and Tobago and Venezuela. Asphalts are non-volatile at ambient temperatures and soften gradually when heated. Asphalt should not be confused with tar, which is physically and chemically dissimilar.
A wide variety of applications include paving streets, highways and airfields; making roofing, waterproofing and insulating materials; lining irrigation canals and reservoirs; and the facing of dams and levees. Asphalt is also a valuable ingredient of some paints and varnishes. It is estimated that the current annual world production of asphalts is over 60 million tonnes, with more than 80% being used in need construction and maintenance and more than 15% used in roofing materials.
Asphalt mixes for road construction are produced by first heating and drying mixtures of graded crushed stone (such as granite or limestone), sand and filler and then mixing with penetration bitumen, referred to in the US as straight-run asphalt. This is a hot process. The asphalt is also heated using propane flames during application to a road bed.
Exposures and Hazards
Exposures to particulate polynuclear aromatic hydrocarbons (PAHs) in asphalt fumes have been measured in a variety of settings. Most of the PAHs found was composed of napthalene derivatives, not the four- to six-ring compounds which are more likely to pose a significant carcinogenic risk. In refinery asphalt processing units, respirable PAH levels range from non-detectable to 40 mg/m3. During drum-filling operations, 4 hour breathing zone samples ranged from 1.0 mg/m3upwind to 5.3 mg/m3 downwind. At asphalt mixing plants, exposures to benzene-soluble organic compounds ranged from 0.2 to 5.4 mg/m3. During paving operations, exposures to respirable PAH ranged from less than 0.1 mg/m3 to 2.7 mg/m3. Potentially noteworthy worker exposures may also occur during the manufacture and application of asphalt roofing materials. Little information is available regarding exposures to asphalt fumes in other industrial situations and during the application or use of asphalt products.
Handling of hot asphalt can cause severe burns because it is sticky and is not readily removed from the skin. The principal concern from the industrial toxicological aspect is irritation of the skin and eyes by fumes of hot asphalt. These fumes may cause dermatitis and acne-like lesions as well as mild keratoses on prolonged and repeated exposure. The greenish-yellow fumes given off by boiling asphalt can also cause photosensitization and melanosis.
Although all asphaltic materials will combust if heated sufficiently, asphalt cements and oxidized asphalts will not normally burn unless their temperature is raised about 260°C. The flammability of the liquid asphalts is influenced by the volatility and amount of petroleum solvent added to the base material. Thus, the rapid-curing liquid asphalts present the greatest fire hazard, which becomes progressively lower with the medium- and slow-curing types.
Because of its insolubility in aqueous media and the high molecular weight of its components, asphalt has a low order of toxicity.
The effects on the tracheobronchial tree and lungs of mice inhaling an aerosol of petroleum asphalt and another group inhaling smoke from heated petroleum asphalt included congestion, acute bronchitis, pneumonitis, bronchial dilation, some peribronchiolar round cell infiltration, abscess formation, loss of cilia, epithelial atrophy and necrosis. The pathological changes were patchy, and in some animals were relatively refractory to treatment. It was concluded that these changes were a non-specific reaction to breathing air polluted with aromatic hydrocarbons, and that their extent was dose dependent. Guinea pigs and rats inhaling fumes from heated asphalt showed effects such as chronic fibrosing pneumonitis with peribronchial adenomatosis, and the rats developed squamous cell metaplasia, but none of the animals had malignant lesions.
Steam-refined petroleum asphalts were tested by application to the skin of mice. Skin tumours were produced by undiluted asphalts, dilutions in benzene and a fraction of steam-refined asphalt. When air-refined (oxidized) asphalts were applied to the skin of mice, no tumour was found with undiluted material, but, in one experiment, an air-refined asphalt in solvent (toluene) produced topical skin tumours. Two cracking-residue asphalts produced skin tumours when applied to the skin of mice. A pooled mixture of steam- and air-blown petroleum asphalts in benzene produced tumours at the site of application on the skin of mice. One sample of heated, air-refined asphalt injected subcutaneously into mice produced a few sarcomas at the injection sites. A pooled mixture of steam- and air-blown petroleum asphalts produced sarcomas at the site of subcutaneous injection in mice. Steam-distilled asphalts injected intramuscularly produced local sarcomas in one experiment in rats. Both an extract of road-surfacing asphalt and its emissions were mutagenic to Salmonella typhimurium.
Evidence for carcinogenicity to humans is not conclusive. A cohort of roofers exposed to both asphalts and coal tar pitches showed an excess risk for respiratory cancer. Likewise, two Danish studies of asphalt workers found an excess risk for lung cancer, but some of these workers may also have been exposed to coal tar, and they were more likely to be smokers than the comparison group. Among Minnesota (but not California) highway workers, increases were noted for leukaemia and urological cancers. Even though the epidemiological data to date are inadequate to demonstrate with a reasonable degree of scientific certainty that asphalt presents a cancer risk to humans, general agreement exists, on the basis of experimental studies, that asphalt may pose such a risk.
Safety and Health Measures
Since heated asphalt will cause severe skin burns, those working with it should wear loose clothing in good condition, with the neck closed and the sleeves rolled down. Hand and arm protection should be worn. Safety shoes should be about 15 cm high and laced so that no openings are left through which hot asphalt may reach the skin. Face and eye protection is also recommended when heated asphalt is handled. Changing rooms and proper washing and bathing facilities are desirable. At crushing plants where dust is produced and at boiling pans from which fumes escape, adequate exhaust ventilation should be provided.
Asphalt kettles should be set securely and be levelled to preclude the possibility of their tipping. Workers should stand upwind of a kettle. The temperature of heated asphalt should be checked frequently in order to prevent overheating and possible ignition. If the flash point is approached, the fire under a kettle must be put out at once and no open flame or other source of ignition should be permitted nearby. Where asphalt is being heated, fire-extinguishing equipment should be within easy reach. For asphalt fires, dry chemical or carbon dioxide types of extinguishers are considered most appropriate. The asphalt spreader and the driver of an asphalt paving machine should be offered half-face respirators with organic vapour cartridges. In addition, to prevent the inadvertent swallowing of toxic materials, workers should not eat, drink or smoke near a kettle.
If molten asphalt strikes the exposed skin, it should be cooled immediately by quenching with cold water or by some other method recommended by medical advisers. An extensive burn should be covered with a sterile dressing and the patient should be taken to a hospital; minor burns should be seen by a physician. Solvents should not be used to remove asphalt from burned flesh. No attempt should be made to remove particles of asphalt from the eyes; instead the victim should be taken to a physician at once.
Classes of bitumens / asphalts
Class 1: Penetration bitumens are classified by their penetration value. They are usually produced from the residue from atmospheric distillation of petroleum crude oil by applying further distillation under vacuum, partial oxidation (air rectification), solvent precipitation or a combination of these processes. In Australia and the United States, bitumens that are approximately equivalent to those described here are called asphalt cements or viscosity-graded asphalts, and are specified on the basis of viscosity measurements at 60°C.
Class 2: Oxidized bitumens are classified by their softening points and penetration values. They are produced by passing air through hot, soft bitumen under controlled temperature conditions. This process alters the characteristics of the bitumen to give reduced temperature susceptibility and greater resistance to different types of imposed stress. In the United States, bitumens produced using air blowing are known as air-blown asphalts or roofing asphalts and are similar to oxidized bitumens.
Class 3: Cutback bitumens are produced by mixing penetration bitumens or oxidized bitumens with suitable volatile diluents from petroleum crudes such as white spirit, kerosene or gas oil, to reduce their viscosity and render them more fluid for ease of handling. When the diluent evaporates, the initial properties of bitumen are recovered. In the United States, cutback bitumens are sometimes referred to as road oils.
Class 4: Hard bitumens are normally classified by their softening point. They are manufactured similarly to penetration bitumens, but have lower penetration values and higher softening points (i.e., they are more brittle).
Class 5: Bitumen emulsions are fine dispersions of droplets of bitumen (from classes 1, 3 or 6) in water. They are manufactured using high-speed shearing devices, such as colloid mills. The bitumen content can range from 30 to 70% by weight. They can be anionic, cationic or non-ionic. In the United States, they are referred to as emulsified asphalts.
Class 6: Blended or fluxed bitumens may be produced by blending bitumens (primarily penetration bitumens) with solvent extracts (aromatic by-products from the refining of base oils), thermally cracked residues or certain heavy petroleum distillates with final boiling points above 350°C.
Class 7: Modified bitumens contain appreciable quantities (typically 3 to 15% by weight) of special addidtives, such as polymers, elastomers, sulphur and other products used to modify their properties; they are used for specialized applications.
Class 8: Thermal bitumens were produced by extended distillation, at high temperature, of a petroleum residue. Currently, they are not manufactured in Europe or in the United States.
Source: IARC1985
Gravel
Gravel is a loose conglomerate of stones that have been mined from a surface deposit, dredged from a river bottom or obtained from a quarry and crushed into desired sizes. Gravel has a variety of uses, including: for rail beds; in roadways, walkways and roofs; as filler in concrete (often for foundations); in landscaping and gardening; and as a filter medium.
The principal safety and health hazards to those who work with gravel are airborne silica dust, musculoskeletal problems and noise. Free crystalline silicon dioxide occurs naturally in many rocks that are used to make gravel. The silica content of bulk species of stone varies and is not a reliable indicator of the percentage of airborne silica dust in a dust sample. Granite contains about 30% silica by weight. Limestone and marble have less free silica.
Silica can become airborne during quarrying, sawing, crushing, sizing and, to a lesser extent, spreading of gravel. Generation of airborne silica can usually be prevented with water sprays and jets, and sometimes with local exhaust ventilation (LEV). In addition to construction workers, workers exposed to silica dust from gravel include quarry workers, railroad workers and landscape workers. Silicosis is more common among quarry or stone-crushing workers than among construction workers who work with gravel as a finished product. An elevated risk of mortality from pneumoconiosis and other non-malignant respiratory disease has been observed in one cohort of workers in the crushed-stone industry in the United States.
Musculoskeletal problems can occur as a result of manual loading or unloading of gravel or during manual spreading. The larger the individual pieces of stone and the larger the shovel or other tool used, the more difficult it is to manage the material with hand tools. The risk of sprains and strains can be reduced if two or more workers work together on strenuous tasks, and more so if draught animals or powered machines are used. Smaller shovels or rakes carry or push less weight than larger ones and can reduce the risk of musculoskeletal problems.
Noise accompanies mechanical processing or handling of stone or gravel. Stone crushing using a ball mill generates considerable low-frequency noise and vibration. Transporting gravel through metal chutes and mixing it in drums are both noisy processes. Noise can be controlled by using sound-absorbing or -reflecting materials around the ball mill, by using chutes lined with wood or other sound-absorbing (and durable) material or by using noise-insulated mixing drums.
Case Studies: Prevention of Occupational Dermatosis Among Workers Exposed to Cement Dust
The most common form of occupational dermatosis to be found among construction workers is caused by exposure to cement. Depending on the country, 5 to 15% of construction workers—most of them masons—acquire dermatosis during their work lives. Two types of dermatosis are caused by exposure to cement: (1) toxic contact dermatitis, which is local irritation of skin exposed to wet cement and is caused mainly by the alkalinity of the cement; and (2) allergic contact dermatitis, which is a generalized allergic skin reaction to exposure to the water-soluble chromium compound found in most cement. One kilogramme of normal cement dust contains 5 to 10 mg of water-soluble chromium. The chromium originates both in the raw material and the production process (mainly from steel structures used in production).
Allergic contact dermatitis is chronic and debilitating. If not properly treated, it can lead to decreased worker productivity and, in some cases, early retirement. In the 1960s and 1970s, cement dermatitis was the most common reported cause of early retirement among construction workers in Scandinavia. Therefore, technical and hygienic procedures were undertaken to prevent cement dermatitis. In 1979, Danish scientists suggested that reducing hexavalent water-soluble chromium to trivalent insoluble chromium by adding ferrous sulphate during production would prevent chromium-induced dermatitis (Fregert, Gruvberger and Sandahl 1979).
Denmark passed legislation requiring the use of cement with lower levels of hexavalent chromium in 1983. Finland followed with a legislative decision at the beginning of 1987, and Sweden and Germany adopted administrative decisions in 1989 and 1993, respectively. For the four countries, the accepted level of water-soluble chromium in cement was determined to be less than 2 mg/kg.
Before Finland’s action in 1987, the Board of Labour Protection wanted to evaluate the occurrence of chromium dermatitis in Finland. The Board asked the Finnish Institute of Occupational Health to monitor the incidence of occupational dermatosis among construction workers to assess the effectiveness of adding ferrous sulphate to cement in order to prevent chromium-induced dermatitis. The Institute monitored the incidence of occupational dermatitis through the Finnish Register of Occupational Diseases from 1978 through 1992. The results indicated that chromium-induced hand dermatitis practically disappeared among construction workers, whereas the incidence of toxic contact dermatitis remained unchanged during the study period (Roto et al. 1996).
In Denmark, chromate sensitization from cement was detected in only one case among 4,511 patch tests conducted between 1989 and 1994 among patients of a large dermatological clinic, 34 of whom were construction workers. The expected number of chromate-positive construction workers was 10 of 34 subjects (Zachariae, Agner and Menn J1996).
There seems to be increasing evidence that the addition of ferrous sulphate to cement prevents chromate sensitization among construction workers. In addition, there has been no indication that, when added to cement, ferrous sulphate has negative effects on the health of exposed workers. The process is economically feasible, and the properties of the cement do not change. It has been calculated that adding ferrous sulphate to cement increases the production costs by US$1.00 per tonne. The reductive effect of ferrous sulphate lasts 6 months; the product must be kept dry before mixing because humidity neutralizes the effect of the ferrous sulphate.
The addition of ferrous sulphate to cement does not change its alkalinity. Therefore workers should use proper skin protection. In all circumstances, construction workers should avoid touching wet cement with unprotected skin. This precaution is especially important in initial cement production, where minor adjustments to moulded elements are made manually.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."