Platcow, Philip A.
Surface Treatment of Metals
Adapted from the 3rd edition, Encyclopaedia of Occupational Health and Safety.
There is a wide variety of techniques for finishing the surfaces of metal products so that they resist corrosion, fit better and look better (see table 1). Some products are treated by a sequence of several of these techniques. This article will briefly describe some of those most commonly used.
Table 1. Summary of the hazards associated with the different metal treatment methods
|
Metal treatment method |
Hazards |
Precautions |
|
Electrolytic polishing |
Burns and irritation from caustic and corrosive chemicals |
Use appropriate personal protective equipment. Install effective exhaust ventilation. |
|
Electroplating |
Exposure to potentially cancer causing chromium and nickel; exposure to cyanides; burns and irritation from caustic and corrosive chemicals; electric shock; the process can be wet, causing slip and fall hazards; potential explosive dust generation; ergonomic hazards |
Use appropriate personal protective equipment. Install effective exhaust ventilation, often slotted, push-pull system. Clean up spills immediately. Install non-skid flooring. Use effective design of work procedures and stations to avoid ergonomic stress. |
|
Enamels and glazing |
Physical hazards from grinders, conveyers, mills; burn hazard from high temperature liquids and equipment; exposure to dusts that may cause lung disease |
Install proper machine guards, including interlocks. Use appropriate personal protective equipment. Install effective exhaust ventilation to avoid dust exposure. HEPA-filtered equipment may be necessary. |
|
Etching |
Exposure to hydrofluoric acid; burns and irritation from caustic and corrosive chemicals; burn hazard from high temperature liquids and equipment |
Implement a programme to avoid exposure to hydrofluoric acid. Use appropriate personal protective equipment. Install effective exhaust ventilation. |
|
Galvanizing |
Burn hazard from high temperature liquids, metals, and equipment; burns and irritation from caustic and corrosive chemicals; metal fume fever; potential lead exposure |
Use appropriate personal protective equipment. Install effective exhaust ventilation. Implement a lead exposure reduction/monitoring programme. |
|
Heat treatment |
Burn hazard from high temperature liquids, metals and equipment; burns and irritation from caustic and corrosive chemicals; possible explosive atmospheres of hydrogen; potential exposure to carbon monoxide; potential exposure to cyanides; fire hazard from oil quenching |
Use appropriate personal protective equipment. Install effective exhaust ventilation. Display signs warning of high temperature equipment and surfaces. Install systems to monitor the concentration of carbon monoxide. Install adequate fire-suppression systems. |
|
Metallizing |
Burn hazard from high temperature metals and equipment; possible explosive atmospheres of dust, acetylene; zinc metal fume fever |
Install adequate fire suppression systems. Properly separate chemicals and gases. Use appropriate personal protective equipment. Install effective exhaust ventilation. |
|
Phosphating |
Burns and irritation from caustic and corrosive chemicals |
Use appropriate personal protective equipment. Install effective exhaust ventilation. |
|
Plastics coating |
Exposure to chemical sensitizers |
Seek alternatives to sensitizers. Use appropriate personal protective equipment. Install effective exhaust ventilation. |
|
Priming |
Exposure to various solvents which are potentially toxic and flammable, exposure to chemical sensitizers, exposure to potentially carcinogenic chromium |
Seek alternatives to sensitizers. Use appropriate personal protective equipment. Install effective exhaust ventilation. Properly separate chemicals/gases. |
Before any of these techniques can be applied, the products must be thoroughly cleaned. A number of methods of cleaning are used, individually or in sequence. They include mechanical grinding, brushing and polishing (which produce metallic or oxidic dust—aluminium dust may be explosive), vapour degreasing, washing with organic grease solvents, “pickling” in concentrated acid or alkaline solutions and electrolytic degreasing. The last involves immersion in baths containing cyanide and concentrated alkali in which electrolytically formed hydrogen or oxygen remove the grease, resulting in “blank” metal surfaces that are free from oxides and grease. The cleaning is followed by adequate rinsing and drying of the product.
Proper design of the equipment and effective LEV will reduce some of the risk. Workers exposed to the hazard of splashes must be provided with protective goggles or eye shields and protective gloves, aprons and clothing. Showers and eyewash fountains should be nearby and in good working order, and splashes and spills should be washed away promptly. With electrolytic equipment, the gloves and shoes must be non-conducting, and other standard electrical precautions, such as the installation of ground fault circuit interrupters and lockout/tagout procedures should be followed.
Treatment Processes
Electrolytic polishing
Electrolytic polishing is used to produce a surface of improved appearance and reflectivity, to remove excess metal to accurately fit the required dimensions and to prepare the surface for inspection for imperfections. The process involves preferential anodic dissolution of high spots on the surface after vapour degreasing and hot alkaline cleaning. Acids are frequently used as the electrolyte solutions; accordingly, adequate rinsing is required afterwards.
Electroplating
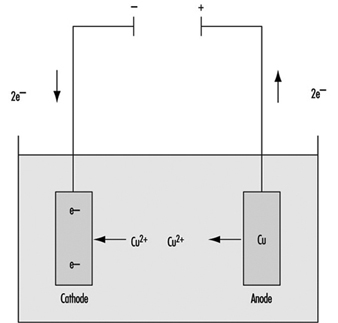
Electroplating is a chemical or electrochemical process for applying a metallic layer to the product—for example, nickel to protect against corrosion, hard chromium to improve the surface properties or silver and gold to beautify it. Occasionally, non-metallic materials are used. The product, wired as the cathode, and an anode of the metal to be deposited are immersed in an electrolyte solution (which can be acidic, alkaline or alkaline with cyanide salts and complexes) and connected externally to a source of direct current. The positively charged cations of the metallic anode migrate to the cathode, where they are reduced to the metal and deposited as a thin layer (see figure 1). The process is continued until the new coating reaches the desired thickness, and the product is then washed, dried and polished.
Figure 1. Electroplating: Schematic representation
Anode: Cu → Cu+2 + 2e- ; Cathode: Cu+2 + 2e- → Cu
In electroforming, a process closely related to electroplating, objects moulded of, for example, plaster or plastic are made conductive by the application of graphite and then are connected as the cathode so that the metal is deposited on them.
In anodization, a process that has become increasingly important in recent years, products of aluminium (titanium and other metals are also used) are connected as the anode and immersed in dilute sulphuric acid. However, instead of the formation of positive aluminium ions and migrating for deposition on the cathode, they are oxidized by the oxygen atoms arising at the anode and become bound to it as an oxide layer. This oxide layer is partially dissolved by the sulphuric acid solution, making the surface layer porous. Subsequently, coloured or light-sensitive materials can be deposited in these pores, as in the fabrication of nameplates, for example.
Enamels and glazes
Vitreous enamel or porcelain enamel is used to give a high heat-, stain- and corrosion-resistant covering to metals, usually iron or steel, in a wide range of fabricated products including bath tubs, gas and electric cookers, kitchen ware, storage tanks and containers, and electrical equipment. In addition, enamels are used in the decoration of ceramics, glass, jewellery and decorative ornaments. The specialized use of enamel powders in the production of such ornamental ware as Cloisonné and Limoges has been known for centuries. Glazes are applied to pottery ware of all kinds.
The materials used in the manufacture of vitreous enamels and glazes include:
- refractories, such as quartz, feldspar and clay
- fluxes, such as borax (sodium borate decahydrate), soda ash (anhydrous sodium carbonate), sodium nitrate, fluorspar, cryolite, barium carbonate, magnesium carbonate, lead monoxide, lead tetroxide and zinc oxide
- colours, such as oxides of antimony, cadmium, cobalt, iron, nickel, manganese, selenium, vanadium, uranium and titanium
- opacifiers, such as oxides of antimony, titanium, tin and zirconium, and sodium antimoninate
- electrolytes, such as borax, soda ash, magnesium carbonate and sulphate, sodium nitrite and sodium aluminate
- flocculating agents, such as clay, gums, ammonium alginate, bentonite and colloidal silica.
The first step in all types of vitreous enamelling or glazing is the making of the frit, the enamel powder. This involves preparation of the raw materials, smelting and frit handing.
After careful cleaning of the metal products (e.g., shot blasting, pickling, degreasing), the enamel may be applied by a number of procedures:
- In the wet process, the object is dipped into the aqueous enamel slip, withdrawn and allowed to drain or, in “slushing”, the enamel slip is thicker and must be shaken from the object.
- In the dry process, the ground-coated object is heated to the enamelling temperature and then dry enamel powder is dusted through sieves onto it. The enamel sinters into place and, when the object is returned to the furnace, it melts down to a smooth surface.
- Spray application is being used increasingly, usually in a mechanized operation. It requires a cabinet under exhaust ventilation.
- Decorative enamels are usually applied by hand, using brushes or similar tools.
- Glazes for porcelain and pottery articles are usually applied by dipping or spraying. Although some dipping operations are being mechanized, pieces are usually dipped by hand in the domestic porcelain industry. The object is held in the hand, dipped into a large tub of glaze, the glaze is removed by a flick of the wrist and the object is placed in a dryer. An enclosed hood or cabinet with efficient exhaust ventilation should be provided when the glaze is sprayed.
The prepared objects are then “fired” in a furnace or kiln, which usually is gas fuelled.
Etching
Chemical etching produces a satin or matte finish. Most frequently, it is used as a pre-treatment prior to anodizing, lacquering, conversion coating, buffing or chemical brightening. It is most frequently applied to aluminium and stainless steel, but is also used for many other metals.
Aluminium is usually etched in alkaline solutions containing various mixtures of sodium hydroxide, potassium hydroxide, trisodium phosphate and sodium carbonate, together with other ingredients to prevent sludge formation. One of the most common processes uses sodium hydroxide at a concentration of 10 to 40 g/l maintained at a temperature of 50 to 85°C with an immersion time as long as 10 minutes.
The alkaline etching is usually preceded and followed by treatment in various mixtures of hydrochloric, hydrofluoric, nitric, phosphoric, chromic or sulphuric acid. A typical acid treatment involves immersions of 15 to 60 seconds in a mixture of 3 parts by volume of nitric acid and 1 part by volume of hydrofluoric acid that is maintained at a temperature of 20°C.
Galvanizing
Galvanizing applies a zinc coating to a variety of steel products to protect against corrosion. The product must be clean and oxide-free for the coating to adhere properly. This usually involves a number of cleaning, rinsing, drying or annealing processes before the product enters the galvanizing bath. In “hot dip” galvanizing, the product is passed through a bath of molten zinc; “cold” galvanizing is essentially electroplating, as described above.
Manufactured products are usually galvanized in a batch process, while the continuous strip method is used for steel strip, sheet or wire. Flux may be employed to maintain satisfactory cleaning of both the product and the zinc bath and to facilitate drying. A prefluxing step may be followed by an ammonium chloride flux cover on the surface of the zinc bath, or the latter may be used alone. In galvanizing pipe, the pipe is immersed in a hot solution of zinc ammonium chloride after cleaning and before the pipe enters the molten zinc bath. The fluxes decompose to form irritating hydrogen chloride and ammonia gas, requiring LEV.
The various types of continuous hot-dip galvanizing differ essentially in how the product is cleaned and whether the cleaning is done on-line:
- cleaning by flame oxidation of the surface oils with subsequent reduction in the furnace and annealing done in-line
- electrolytic cleaning done prior to in-line annealing
- cleaning by acid pickling and alkali cleaning, using a flux prior to the preheat furnace and annealing in a furnace before galvanizing
- cleaning by acid pickling and alkali cleaning, eliminating the flux and preheating in a reducing gas (e.g., hydrogen) prior to galvanizing.
The continuous galvanizing line for light-gauge strip steel omits pickling and the use of flux; it uses alkaline cleaning and maintains the clean surface of the strip by heating it in a chamber or furnace with a reducing atmosphere of hydrogen until it passes below the surface of the molten zinc bath.
Continuous galvanizing of wire requires annealing steps, usually with a molten lead pan in front of the cleaning and galvanizing tanks; air or water cooling; pickling in hot, dilute hydrochloric acid; rinsing; application of a flux; drying; and then galvanizing in the molten zinc bath.
A dross, an alloy of iron and zinc, settles to the bottom of the molten zinc bath and must be removed periodically. Various types of materials are floated on the surface of the zinc bath to prevent oxidation of the molten zinc. Frequent skimming is needed at the points of entry and exit of the wire or strip being galvanized.
Heat treatment
Heat treatment, the heating and cooling of a metal which remains in the solid state, is usually an integral part of the processing of metal products. It almost always involves a change in the crystalline structure of the metal which results in a modification of its properties (e.g., annealing to make the metal more malleable, heating and slow cooling to reduce hardness, heating and quenching to increase hardness, low-temperature heating to minimize internal stresses).
Annealing
Annealing is a “softening” heat treatment widely used to allow further cold working of the metal, improve machinability, stress-relieve the product before it is used and so on. It involves heating the metal to a specific temperature, holding it at that temperature for a specific length of time and allowing it to cool at a particular rate. A number of annealing techniques are used:
- Blue annealing, in which a layer of blue oxide is produced on the surface of iron-based alloys
- Bright annealing, which is carried out in a controlled atmosphere to minimize surface oxidation
- Close annealing or box annealing, a method in which both ferrous and non-ferrous metals are heated in a sealed metal container with or without a packing material and then slowly cooled
- Full annealing, usually carried out in a protective atmosphere, aimed at obtaining the maximum softness economically feasible
- Malleablizing, a special kind of anneal given to iron castings to make them malleable by transforming the combined carbon in the iron to fine carbon (i.e., graphite)
- Partial annealing, a low-temperature process to remove internal stresses induced in the metal by cold working
- Sub-critical or spheroidizing annealing, which produces improved machinability by allowing the iron carbide in the crystalline structure to acquire a spheroid shape.
Age-hardening
Age-hardening is a heat treatment often used on aluminium-copper alloys in which the natural hardening that takes place in the alloy is accelerated by heating to about 180°C for about 1 hour.
Homogenizing
Homogenizing, usually applied to ingots or powdered metal compacts, is designed to remove or greatly reduce segregation. It is achieved by heating to a temperature about 20°C below the metal’s melting point for about 2 hours or more and then quenching.
Normalizing
A process similar to full annealing, ensures the uniformity of the mechanical properties to be obtained and also produces greater toughness and resistance to mechanical loading.
Patenting
Patenting is a special type of annealing process that is usually applied to materials of small cross-section which are intended to be drawn (e.g., 0.6% carbon steel wire). The metal is heated in an ordinary furnace to above the transformation range and then passes from the furnace directly into, for example, a lead bath held at a temperature of about 170°C.
Quench-hardening and tempering
An increase in hardness can be produced in an iron-based alloy by heating to above the transformation range and rapidly cooling to room temperature by quenching in oil, water or air. The article is often too highly stressed to be put into service and, in order to increase its toughness, it is tempered by reheating to a temperature below the transformation range and allowing it to cool at the desired rate.
Martempering and austempering are similar processes except that the article is quenched, for example, in a salt or lead bath held at a temperature of 400°C.
Surface- and case-hardening
This is another heat-treatment process applied most frequently to iron-based alloys, which allows the surface of the object to remain hard while its core remains relatively ductile. It has a number of variations:
- Flame hardening involves hardening the surfaces of the object (e.g., gear teeth, bearings, slideways) by heating with a high-temperature gas torch and then quenching in oil, water or another suitable medium.
- Electrical induction hardening is similar to flame hardening except that the heating is produced by eddy currents induced in the surface layers.
- Carburizing increases the carbon content of the surface of an iron-based alloy by heating the object in a solid, liquid or gaseous carbonaceous medium (e.g., solid charcoal and barium carbonate, liquid sodium cyanide and sodium carbonate, gaseous carbon monoxide, methane and so on) at a temperature of about 900°C.
- Nitriding increases the nitrogen content of the surface of a special low-alloy cast iron or steel object by heating it in a nitrogenous medium, usually ammonia gas, at about 500 to 600°C.
- Cyaniding is a method of case-hardening in which the surface of a low-carbon steel object is enriched in both carbon and nitrogen simultaneously. It usually involves heating the object for 1 hour in a bath of molten 30% sodium cyanide at 870°C, and then quenching in oil or water.
- Carbo-nitriding is a gaseous process for the simultaneous absorption of carbon and nitrogen into the surface layer of steel by heating it to 800 to 875°C in an atmosphere of a carburizing gas (see above) and a nitriding gas (e.g., 2 to 5% anhydrous ammonia).
Metallizing
Metallizing, or metal spraying, is a technique for applying a protective metallic coating to a mechanically roughened surface by spraying it with molten droplets of metal. It is also used to build up worn or corroded surfaces and for salvaging badly-machined component parts. The process is widely known as Schooping, after the Dr. Schoop who invented it.
It uses the Schooping gun, a hand-held, pistol-shaped spray gun through which the metal in wire form is fed into a fuel gas/oxygen blowpipe flame which melts it and, using compressed air, sprays it onto the object. The heat source is a mixture of oxygen and either acetylene, propane or compressed natural gas. The coiled wire is usually straightened before being fed into the gun. Any metal that can be made into a wire may be used; the gun can also accept the metal in powder form.
Vacuum metallizing is a process in which the object is placed in a vacuum jar into which the coating metal is sprayed.
Phosphating
Phosphating is used mainly on mild and galvanized steel and aluminium to augment the adhesion and corrosion resistance of paint, wax and oil finishes. It is also used to form a layer which acts as a parting film in the deep drawing of sheet metal and improves its wear resistance. It essentially consists of allowing the metal surface to react with a solution of one or more phosphates of iron, zinc, manganese, sodium or ammonium. Sodium and ammonium phosphate solutions are used for combined cleaning and phosphating. The need to phosphate multi-metal objects and the desire to increase line speeds in automated operations have led to reducing reaction times by the addition of accelerators such as fluorides, chlorates, molybdates and nickel compounds to the phosphating solutions.To reduce crystal size and, consequently, increase the flexibility of zinc phosphate coatings, crystal refining agents such as tertiary zinc phosphate or titanium phosphate are added to the pre-treatment rinse.
The phosphating sequence typically includes the following steps:
- hot caustic cleaning
- brushing and rinsing
- further hot caustic cleaning
- conditioning water rinse
- spraying or dipping in hot solutions of acid phosphates
- cold water rinse
- warm chromic acid rinse
- another cold water rinse
- drying.
Priming
Organic paint primers are applied to metal surfaces to promote the adhesion of subsequently applied paints and to retard corrosion at the paint-metal interface. The primers usually contain resins, pigments and solvents and may be applied to the prepared metal surfaces by brush, spray, immersion, roller coating or electrophoresis.
The solvents may be any combination of aliphatic and aromatic hydrocarbons, ketones, esters, alcohols and ethers. The most commonly used resins are polyvinyl butynol, phenolic resins, drying oil alkyds, epoxidized oils, epoxyesters, ethyl silicates and chlorinated rubbers. In complex primers, cross-linking agents such as tetraethylene pentamine, pentaethylene hexamine, isocyanates and urea formaldehyde are used. Inorganic pigments used in primer formulations include lead, barium, chromium, zinc and calcium compounds.
Plastic coating
Plastic coatings are applied to metals in liquid form, as powders which are subsequently cured or sintered by heating, or in the form of fabricated sheets which are laminated to the metal surface with an adhesive. The most commonly used plastics include polyethylene, polyamides (nylons) and PVC. The latter may include plasticizers based on monomeric and polymeric esters and stabilizers such as lead carbonate, fatty acid salts of barium and cadmium, dibutyltin dilaurate, alkyltin mercaptides and zinc phosphate. Although generally of low toxicity and non-irritating, some of the plasticizers are skin sensitizers.
Hazards and Their Prevention
As might be deduced from the complexity of the processes outlined above, there is a large variety of safety and health hazards associated with the surface treatment of metals. Many are regularly encountered in manufacturing operations; others are presented by the uniqueness of the techniques and materials employed. Some are potentially life threatening. By and large, however, they can be prevented or controlled.
Workplace design
The workplace should be designed to allow the delivery of raw materials and supplies and the removal of the finished products without interfering with the ongoing processing. Since many of the chemicals are flammable or prone to react when mixed, proper separation in storage and in transit is essential. Many of the metal finishing operations involve liquids, and when leaks, spills or splashes of acids or alkalis occur they must be washed away promptly. Accordingly, adequately drained, slip-resistant floors must be provided. Housekeeping must be diligent to keep the work areas and other spaces clean and free from accumulations of materials. Systems for disposal of solid and liquid wastes and effluents from furnaces and exhaust ventilation must be designed with environmental concerns in mind.
Work stations and work assignments should use ergonomic principles to minimize strains, sprains, excessive fatigue and RSIs. Machine guards must have automatic lockout so the machine is de-energized if the guard is removed. Splash guards are essential. Because of the danger of splashes of hot acid and alkali solutions, eyewash fountains and whole-body showers must be installed within easy reach. Signs should be posted to warn other production and maintenance personnel of such dangers as chemical baths and hot surfaces.
Chemical assessment
All chemicals should be evaluated for potential toxicity and physical hazards, and less hazardous materials should be substituted where possible. However, since the less toxic material may be more flammable, the hazard of fire and explosion must also be considered. In addition, the chemical compatibility of materials must be considered. For example, mixing of nitrate and cyanide salts by accident could cause an explosion due to the strong oxidizing properties of nitrates.
Ventilation
Most of the metal coating processes require LEV that is strategically placed to draw the vapours or other contaminants away from the worker. Some systems push fresh air across the tank to “push” airborne contaminants to the exhaust side of the system. Fresh air intakes must be located away from exhaust vents so that potentially toxic gases are not recirculated.
Personal protective equipment
Processes should be engineered to prevent potentially toxic exposures, but since they cannot always be totally avoided, employees will have to be provided with appropriate PPE (e.g., goggles with or without face shields as appropriate, gloves, aprons or coveralls and shoes). Because many of the exposures involve hot corrosive or caustic solutions, the protective items should be insulated and chemical-resistant. If there is possible exposure to electricity, PPE should be non-conductive. PPE must be available in adequate quantity to allow contaminated, wet items to be cleaned and dried before re-using them. Insulated gloves and other protective clothing should be available where there is the risk of thermal burns from hot metal, furnaces and so on.
An important adjunct is the availability of wash-up facilities and clean lockers and dressing rooms, so that workers’ clothing remains uncontaminated and workers do not carry toxic materials back into their homes.
Employee training and supervision
Employee education and training are essential both when new to the job or when there have been changes in the equipment or the process. MSDSs must be provided for each of the chemical products which explain the chemical and physical hazards, in languages and at educational levels that ensure they will be understood by the workers. Competence testing and periodic retraining will assure that workers have retained the needed information. Close supervision is advisable to make sure that the proper procedures are being followed.
Selected hazards
Certain hazards are unique to the metal coating industry and deserve special consideration.
Alkaline and acid solutions
The heated alkaline and acid solutions used in cleaning and treatment of metals are particularly corrosive and caustic. They are irritating to the skin and mucous membranes and are especially dangerous when splashed into the eye. Eyewash fountains and emergency showers are essential. Proper protective clothing and goggles will guard against the inevitable splashes; when a splash reaches the skin, the area should be immediately and copiously rinsed with cool, clean water for at least 15 minutes; medical attention may be necessary, particularly when the eye is involved.
Care should be exercised when utilizing chlorinated hydrocarbons as phosgene may result from a reaction of the chlorinated hydrocarbon, acids and metals. Nitric and hydrofluoric acid are particularly dangerous when their gases are inhaled, because it may take 4 hours or more before the effects on the lungs become apparent. Bronchitis, pneumonitis and even potentially fatal pulmonary oedema may appear belatedly in a worker who apparently had no initial effect from the exposure. Prompt prophylactic medical treatment and, often, hospitalization are advisable for workers who have been exposed. Skin contact with hydrofluoric acid can cause severe burns without pain for several hours. Prompt medical attention is essential.
Dust
Metallic and oxidic dusts are a particular problem in grinding and polishing operations, and are most effectively removed by LEV as they are created. Ductwork should be designed to be smooth and air velocity should be sufficient to keep the particulates from settling out of the air stream. Aluminium and magnesium dust may be explosive and should be collected in a wet trap. Lead has become less of a problem with the decline of its use in ceramics and porcelain glazes, but it remains the ubiquitous occupational hazard and must always be guarded against. Beryllium and its compounds have received interest recently due to the possibility of carcinogenicity and chronic beryllium disease.
Certain operations present a risk of silicosis and pneumoconiosis: the calcining, crushing and drying of flint, quartz or stone; the sieving, mixing and weighing out of these substances in the dry state; and the charging of furnaces with such materials. They also represent a danger when they are used in a wet process and are splashed about the workplace and on workers’ clothing, to become dusts again when they dry out. LEV and rigorous cleanliness and personal hygiene are important preventive measures.
Organic solvents
Solvents and other organic chemicals used in degreasing and in certain processes are dangerous when inhaled. In the acute phase, their narcotic effects may lead to respiratory paralysis and death. In chronic exposure, toxicity of the central nervous system and liver and kidney damage are most frequent. Protection is provided by LEV with a safety zone of at least 80 to 100 cm between the source and the breathing area of the worker. Bench ventilation must also be installed to remove residual vapours from the finished workpieces. Defatting of the skin by organic solvents may be a precursor of dermatitis. Many solvents are also flammable.
Cyanide
Baths containing cyanides are frequently used in electrolytic degreasing, electroplating and cyaniding. Reaction with acid will form the volatile, potentially lethal hydrogen cyanide (prussic acid). The lethal concentration in air is 300 to 500 ppm. Fatal exposures may also result from skin absorption or ingestion of cyanides. Optimum cleanliness is essential for workers using cyanide. Food should not be eaten before washing, and should never be in the work area. Hands and clothing must be carefully cleaned following a potential cyanide exposure.
First aid measures for cyanide poisoning include transport into the open air, removal of contaminated clothing, copious washing of the exposed areas with water, oxygen therapy and inhalation of amyl nitrite. LEV and skin protection are essential.
Chromium and nickel
Chromic and nickel compounds used in galvanic baths in electroplating may be hazardous. Chromium compounds can cause burns, ulceration and eczema of the skin and mucosa and a characteristic perforation of the nasal septum. Bronchial asthma may occur. Nickel salts can cause obstinate allergic or toxic-irritative skin injury. There is evidence that both chromium and nickel compounds may be carcinogenic. LEV and skin protection are essential.
Furnaces and ovens
Special precautions are needed when working with the furnaces employed, for example, in the heat treatment of metals where components are handled at high temperatures and the materials used in the process may either be toxic or explosive or both. The gaseous media (atmospheres) in the furnace may react with the metal charge (oxidizing or reducing atmospheres) or they may be neutral and protective. Most of the latter contain up to 50% hydrogen and 20% carbon monoxide, which, in addition to being combustible, form highly explosive mixtures with air at elevated temperatures. The ignition temperature varies from 450 to 750 °C, but a local spark may cause ignition even at lower temperatures. The danger of explosion is greater when the furnace is being started up or shut down. Since a cooling furnace tends to suck in air (a particular danger when the fuel or power supply is interrupted), a supply of inert gas (e.g., nitrogen or carbon dioxide) should be available for purging when the furnace is shut down as well as when a protective atmosphere is introduced into a hot furnace.
Carbon monoxide is perhaps the greatest hazard from furnaces and ovens. Since it is colourless and odourless, it frequently reaches toxic levels before the worker becomes aware of it. Headache is one of the earliest symptoms of toxicity, and, therefore, a worker developing a headache on the job should immediately be removed into fresh air. Danger zones include recessed pockets in which the carbon monoxide may collect; it should be remembered that brickwork is porous and may retain the gas during normal purging and emit it when the purging is completed.
Lead furnaces may be dangerous since lead tends to vaporize quite rapidly at temperatures above 870°C. Accordingly, an effective fume extraction system is required. A pot breakage or failure may also be hazardous; a sufficiently large well or pit should be provided to capture the molten metal if this occurs.
Fire and explosion
Many of the compounds used in metal coating are flammable and, under certain circumstances, explosive. For the most part, the furnaces and drying ovens are gas fired, and special precautions such as flame-failure devices at burners, low-pressure cut-off valves in the supply lines and explosion relief panels in the structure of the stoves should be installed. In electrolytic operations, hydrogen formed in the process may collect at the surface of the bath and, if not exhausted, may reach explosive concentrations. Furnaces should be properly ventilated and burners protected from being clogged by dripping material.
Oil quenching is also a fire hazard, especially if the metal charge is not completely immersed. Quenching oils should have a high flashpoint, and their temperature should not exceed 27°C.
Compressed oxygen and fuel gas cylinders used in metallizing are fire and explosion hazards if not stored and operated properly. See the article “Welding and thermal cutting” in this chapter for detailed precautions.
As required by local ordinances, firefighting equipment, including alarms, should be provided and maintained in working order, and the workers drilled in using it properly.
Heat
The use of furnaces, open flames, ovens, heated solutions and molten metals inevitably presents the risk of excessive heat exposure, which is compounded in hot, humid climates and, particularly, by occlusive protective garments and gear. Complete air conditioning of a plant may not be economically feasible, but supplying cooled air in local ventilation systems is helpful. Rest breaks in cool surroundings and adequate fluid intake (fluids taken at the work station should be free of toxic contaminants) will help to avert heat toxicity. Workers and supervisors should be trained in the recognition of heat stress symptoms.
Conclusion
Surface treatment of metals involves a multiplicity of processes entailing a broad range of potentially toxic exposures, most of which can be prevented or controlled by the diligent application of well-recognized preventive measures.
Welding and Thermal Cutting
This article is a revision of the 3rd edition of the Encyclopaedia of Occupational Health and Safety article “Welding and thermal cutting” by G.S. Lyndon.
Process Overview
Welding is a generic term referring to the union of pieces of metal at joint faces rendered plastic or liquid by heat or pressure, or both. The three common direct sources of heat are:
- flame produced by the combustion of fuel gas with air or oxygen
- electrical arc, struck between an electrode and a workpiece or between two electrodes
- electrical resistance offered to passage of current between two or more workpieces.
Other sources of heat for welding are discussed below (see table 1).
Table 1. Process materials inputs and pollution outputs for lead smelting and refining
|
Process |
Material input |
Air emissions |
Process wastes |
Other wastes |
|
Lead sintering |
Lead ore, iron, silica, limestone flux, coke, soda, ash, pyrite, zinc, caustic, baghouse dust |
Sulphur dioxide, particulate matter contain-ing cadmium and lead |
||
|
Lead smelting |
Lead sinter, coke |
Sulphur dioxide, particulate matter contain-ing cadmium and lead |
Plant washdown wastewater, slag granulation water |
Slag containing impurities such as zinc, iron, silica and lime, surface impoundment solids |
|
Lead drossing |
Lead bullion, soda ash, sulphur, baghouse dust, coke |
Slag containing such impurities as copper, surface impoundment solids |
||
|
Lead refining |
Lead drossing bullion |
In gas welding and cutting, oxygen or air and a fuel gas are fed to a blowpipe (torch) in which they are mixed prior to combustion at the nozzle. The blowpipe is usually hand held (see figure 1). The heat melts the metal faces of the parts to be joined, causing them to flow together. A filler metal or alloy is frequently added. The alloy often has a lower melting point than the parts to be joined. In this case, the two pieces are generally not brought to fusion temperature (brazing, soldering). Chemical fluxes may be used to prevent oxidation and facilitate the joining.
Figure 1. Gas welding with a torch & rod of filter metal. The welder is protected by a leather apron, gauntlets and goggles
In arc welding, the arc is struck between an electrode and the workpieces. The electrode can be connected to either an alternating current (AC) or direct current (DC) electric supply. The temperature of this operation is about 4,000°C when the workpieces fuse together. Usually it is necessary to add molten metal to the joint either by melting the electrode itself (consumable electrode processes) or by melting a separate filler rod which is not carrying current (non-consumable electrode processes).
Most conventional arc welding is done manually by means of a covered (coated) consumable electrode in a hand-held electrode holder. Welding is also accomplished by many semi or fully automatic electric welding processes such as resistance welding or continuous electrode feed.
During the welding process, the welding area must be shielded from the atmosphere in order to prevent oxidation and contamination. There are two types of protection: flux coatings and inert gas shielding. In flux-shielded arc welding, the consumable electrode consists of a metal core surrounded by a flux coating material, which is usually a complex mixture of mineral and other components. The flux melts as welding progresses, covering the molten metal with slag and enveloping the welding area with a protective atmosphere of gases (e.g., carbon dioxide) generated by the heated flux. After welding, the slag must be removed, often by chipping.
In gas-shielded arc welding, a blanket of inert gas seals off the atmosphere and prevents oxidation and contamination during the welding process. Argon, helium, nitrogen or carbon dioxide are commonly used as the inert gases. The gas selected depends upon the nature of the materials to be welded. The two most popular types of gas-shielded arc welding are metal- and tungsten inert gas (MIG and TIG).
Resistance welding involves using the electrical resistance to the passage of a high current at low voltage through components to be welded to generate heat for melting the metal. The heat generated at the interface between the components brings them to welding temperatures.
Hazards and Their Prevention
All welding involves hazards of fire, burns, radiant heat (infrared radiation) and inhalation of metal fumes and other contaminants. Other hazards associated with specific welding processes include electrical hazards, noise, ultraviolet radiation, ozone, nitrogen dioxide, carbon monoxide, fluorides, compressed gas cylinders and explosions. See table 2 for additional detail.
Table 2. Description and hazards of welding processes
|
Welding Process |
Description |
Hazards |
|
Gas welding and cutting |
||
|
Welding |
The torch melts the metal surface and filler rod, causing a joint to be formed. |
Metal fumes, nitrogen dioxide, carbon monoxide, noise, burns, infrared radiation, fire, explosions |
|
Brazing |
The two metal surfaces are bonded without melting the metal. The melting temperature of the filler metal is above 450 °C. Heating is done by flame heating, resistance heating and induction heating. |
Metal fumes (especially cadmium), fluorides, fire, explosion, burns |
|
Soldering |
Similar to brazing, except the melting temperature of the filler metal is below 450 °C. Heating is also done using a soldering iron. |
Fluxes, lead fumes, burns |
|
Metal cutting and flame gouging |
In one variation, the metal is heated by a flame, and a jet of pure oxygen is directed onto the point of cutting and moved along the line to be cut. In flame gouging, a strip of surface metal is removed but the metal is not cut through. |
Metal fumes, nitrogen dioxide, carbon monoxide, noise, burns, infrared radiation, fire, explosions |
|
Gas pressure welding |
The parts are heated by gas jets while under pressure, and become forged together. |
Metal fumes, nitrogen dioxide, carbon monoxide, noise, burns, infrared radiation, fire, explosions |
|
Flux-shielded arc welding |
||
|
Shielded metal arc welding (SMAC); “stick” arc welding; manual metal arc welding (MMA); open arc welding |
Uses a consumable electrode consisting of a metal core surrounded by a flux coating |
Metal fumes, fluorides (especially with low-hydrogen electrodes), infrared and ultraviolet radiation, burns, electrical, fire; also noise, ozone, nitrogen dioxide |
|
Submerged arc welding (SAW) |
A blanket of granulated flux is deposited on the workpiece, followed by a consumable bare metal wire electrode. The arc melts the flux to produce a protective molten shield in the welding zone. |
Fluorides, fire, burns, infrared radiation, electrical; also metal fumes, noise, ultraviolet radiation, ozone, and nitrogen dioxide |
|
Gas-shielded arc welding |
||
|
Metal inert gas (MIG); gas metal arc welding (GMAC) |
The electrode is normally a bare consumable wire of similar composition to the weld metal and is fed continuously to the arc. |
Ultraviolet radiation, metal fumes, ozone, carbon monoxide (with CO2 gas), nitrogen dioxide, fire, burns, infrared radiation, electrical, fluorides, noise |
|
Tungsten inert gas (TIG); gas tungsten arc welding (GTAW); heliarc |
The tungsten electrode is non-consumable, and filler metal is introduced as a consumable into the arc manually. |
Ultraviolet radiation, metal fumes, ozone, nitrogen dioxide, fire, burns, infrared radiation, electrical, noise, fluorides, carbon monoxide |
Plasma arc welding (PAW) and plasma arc spraying; tungsten arc cutting |
Similar to TIG welding, except that the arc and stream of inert gases pass through a small orifice before reaching the workpiece, creating a “plasma” of highly ionized gas which can achieve temperatures of over 33,400°C.This is also used for metallizing. |
Metal fumes, ozone, nitrogen dioxide, ultraviolet and infrared radiation, noise; fire, burns, electrical, fluorides, carbon monoxide, possible x rays |
|
Flux core arc welding (FCAW); metal active gas welding (MAG) |
Uses a flux-cored consumable electrode; may have carbon dioxide shield (MAG) |
Ultraviolet radiation, metal fumes, ozone, carbon monoxide (with CO2 gas), nitrogen dioxide, fire, burns, infrared radiation, electrical, fluorides, noise |
|
Electric resistance welding |
||
|
Resistance welding (spot, seam, projection or butt welding) |
A high current at low voltage flows through the two components from electrodes. The heat generated at the interface between the components brings them to welding temperatures. During the passage of the current, pressure by the electrodes produces a forge weld. No flux or filler metal is used. |
Ozone, noise (sometimes), machinery hazards, fire, burns, electrical, metal fumes |
|
Electro-slag welding |
Used for vertical butt welding. The workpieces are set vertically, with a gap between them, and copper plates or shoes are placed on one or both sides of the joint to form a bath. An arc is established under a flux layer between one or more continuously fed electrode wires and a metal plate. A pool of molten metal is formed, protected by molten flux or slag, which is kept molten by resistance to the current passing between the electrode and the workpieces. This resistance-generated heat melts the sides of the joint and the electrode wire, filling the joint and making a weld. As welding progresses, the molten metal and slag are retained in position by shifting the copper plates. |
Burns, fire, infrared radiation, electrical, metal fumes |
|
Flash welding |
The two metal parts to be welded are connected to a low-voltage, high-current source. When the ends of the components are brought into contact, a large current flows, causing “flashing” to occur and bringing the ends of the components to welding temperatures. A forge weld is obtained by pressure. |
Electrical, burns, fire, metal fumes |
Other welding processes |
||
|
Electron beam welding |
A workpiece in an vacuum chamber is bombarded by a beam of electrons from an electron gun at high voltages. The energy of the electrons is transformed into heat upon striking the workpiece, thus melting the metal and fusing the workpiece. |
X rays at high voltages, electrical, burns, metal dusts, confined spaces |
|
Arcair cutting |
An arc is struck between the end of a carbon electrode (in a manual electrode holder with its own supply of compressed air) and the workpiece. The molten metal produced is blown away by jets of compressed air. |
Metal fumes, carbon monoxide, nitrogen dioxide, ozone, fire, burns, infrared radiation, electrical |
|
Friction welding |
A purely mechanical welding technique in which one component remains stationary while the other is rotated against it under pressure. Heat is generated by friction, and at forging temperature the rotation ceases. A forging pressure then effects the weld. |
Heat, burns, machinery hazards |
|
Laser welding and drilling |
Laser beams can be used in industrial applications requiring exceptionally high precision, such as miniature assemblies and micro techniques in the electronics industry or spinnerets for the artificial fibre industry. The laser beam melts and joins the workpieces. |
Electrical, laser radiation, ultraviolet radiation, fire, burns, metal fumes, decomposition products of workpiece coatings |
|
Stud welding |
An arc is struck between a metal stud (acting as the electrode) held in a stud welding gun and the metal plate to be joined, and raises the temperature of the ends of the components to melting point. The gun forces the stud against the plate and welds it. Shielding is provided by a ceramic ferrule surrounding the stud. |
Metal fumes, infrared and ultraviolet radiation, burns, electrical, fire, noise, ozone, nitrogen dioxide |
|
Thermite welding |
A mixture of aluminium powder and a metal oxide powder (iron, copper, etc.) is ignited in a crucible, producing molten metal with the evolution of intense heat. The crucible is tapped and the molten metal flows into the cavity to be welded (which is surrounded by a sand mould). This is often used to repair castings or forgings. |
Fire, explosion, infrared radiation, burns |
Much welding is not done in shops where conditions can generally be controlled, but in the field in the construction or repair of large structures and machinery (e.g., frameworks of buildings, bridges and towers, ships, railroad engines and cars, heavy equipment and so on). The welder may have to carry all his or her equipment to the site, set it up and work in confined spaces or on scaffolds. Physical strain, inordinate fatigue and musculoskeletal injuries may follow being required to reach, kneel or work in other uncomfortable and awkward positions. Heat stress may result from working in warm weather and the occlusive effects of the personal protective equipment, even without the heat generated by the welding process.
Compressed gas cylinders
In high-pressure gas welding installations, oxygen and the fuel gas (acetylene, hydrogen, town gas, propane) are supplied to the torch from cylinders. The gases are stored in these cylinders at high pressure. The special fire and explosion hazards and precautions for the safe use and storage of the fuel gases are also discussed elsewhere in this Encyclopaedia. The following precautions should be observed:
- Only pressure regulators designed for the gas in use should be fitted to cylinders. For example, an acetylene regulator should not be used with coal gas or hydrogen (although it may be used with propane).
- Blowpipes must be kept in good order and cleaned at regular intervals. A hardwood stick or soft brass wire should be used for cleaning the tips. They should be connected to regulators with special canvas-reinforced hoses placed in such a way that they are unlikely to be damaged.
- Oxygen and acetylene cylinders must be stored separately and only on fire-resistant premises devoid of flammable material and must be so located that they may be readily removed in case of fire. Local building and fire protection codes must be consulted.
- The colour coding in force or recommended for identification of cylinders and accessories should be scrupulously observed. In many countries, the internationally accepted colour codes used for the transport of dangerous materials are applied in this field. The case for enforcement of uniform international standards in this respect is strengthened by safety considerations bound up with the increasing international migration of industrial workers.
Acetylene generators
In the low-pressure gas welding process, acetylene is generally produced in generators by reaction of calcium carbide and water. The gas is then piped to the welding or cutting torch into which oxygen is fed.
Stationary generating plants should be installed either in the open air or in a well-ventilated building away from the main workshops. The ventilation of the generator house should be such as to prevent the formation of an explosive or toxic atmosphere. Adequate lighting should be provided; switches, other electrical gear and electrical lamps should either be located outside the building or be explosion-proof. Smoking, flames, torches, welding plant or flammable materials must be excluded from the house or from the vicinity of an open-air generator. Many of these precautions also apply to portable generators. Portable generators should be used, cleaned and recharged only in the open air or in a well-ventilated shop, away from any flammable material.
Calcium carbide is supplied in sealed drums. The material should be stored and kept dry, on a platform raised above the floor level. Stores must be situated under cover, and if they adjoin another building the party wall must be fireproof. The storeroom should be suitably ventilated through the roof. Drums should be opened only immediately before the generator is charged. A special opener should be provided and used; a hammer and chisel should never be used to open drums. It is dangerous to leave calcium carbide drums exposed to any source of water.
Before a generator is dismantled, all calcium carbide must be removed and the plant filled with water. The water should remain in the plant for at least half an hour to ensure that every part is free from gas. The dismantling and servicing should be carried out only by the manufacturer of the equipment or by a specialist. When a generator is being recharged or cleaned, none of the old charge must be used again.
Pieces of calcium carbide wedged in the feed mechanism or adhering to parts of the plant should be carefully removed, using non-sparking tools made of bronze or another suitable non-ferrous alloy.
All concerned should be fully conversant with the manufacturer’s instructions, which should be conspicuously displayed. The following precautions should also be observed:
- A properly designed back-pressure valve must be fitted between the generator and each blowpipe to prevent backfire or reverse flow of gas. The valve should be regularly inspected after backfire, and the water level checked daily.
- Only blowpipes of the injector type designed for low-pressure operation should be used. For heating and cutting, town gas or hydrogen at low pressure are sometimes employed. In these cases, a non-return valve should be placed between each blowpipe and the supply main or pipeline.
- An explosion may be caused by “flash-back”, which results from dipping the nozzle-tip into the molten metal pool, mud or paint, or from any other stoppage. Particles of slag or metal that become attached to the tip should be removed. The tip should also be cooled frequently.
- Local building and fire codes should be consulted.
Fire and explosion prevention
In locating welding operations, consideration should be given to surrounding walls, floors, nearby objects and waste material. The following procedures should be followed:
- All combustible material must be removed or adequately protected by sheet metal or other suitable materials; tarpaulins should never be used.
- Wood structures should be discouraged or similarly protected. Wood floors should be avoided.
- Precautionary measures should be taken in the case of openings or cracks in walls and floors; flammable material in adjoining rooms or on the floor below should be removed to a safe position. Local building and fire codes should be consulted.
- Suitable fire-extinguishing apparatus should always be at hand. In the case of low-pressure plant using an acetylene generator, buckets of dry sand should also be kept available; fire extinguishers of dry powder or carbon dioxide types are satisfactory. Water must never be used.
- Fire brigades may be necessary. A responsible person should be assigned to keep the site under observation for at least half an hour after completion of the work, in order to deal with any outbreak of fire.
- Since explosions can occur when acetylene gas is present in air in any proportion between 2 and 80%, adequate ventilation and monitoring are required to ensure freedom from gas leaks. Only soapy water should be used to search for gas leaks.
- Oxygen must be carefully controlled. For example, it should never be released into the air in a confined space; many metals, clothing and other materials become actively combustible in the presence of oxygen. In gas cutting, any oxygen which may not be consumed will be released into the atmosphere; gas cutting should never be undertaken in a confined space without proper ventilation arrangements.
- Alloys rich in magnesium or other combustible metals should be kept away from welding flames or arcs.
- Welding of containers can be extremely hazardous. If the previous contents are unknown, a vessel should always be treated as if it had contained a flammable substance. Explosions may be prevented either by removing any flammable material or by making it non-explosive and non-flammable.
- The mixture of aluminium and iron oxide used in thermite welding is stable under normal conditions. However, in view of the ease with which aluminium powder will ignite, and the quasi-explosive nature of the reaction, appropriate precautions should be taken in handling and storage (avoidance of exposure to high heat and possible ignition sources).
- A written hot-work permit programme is required for welding in some jurisdictions. This programme outlines the precautions and procedures to be followed during welding, cutting, burning and so on. This programme should include the specific operations conducted along with the safety precautions to be implemented. It must be plant specific and may include an internal permit system that must be completed with each individual operation.
Protection from heat and burn hazards
Burns of the eyes and exposed parts of the body may occur due to contact with hot metal and spattering of incandescent metal particles or molten metal. In arc welding, a high-frequency spark used to initiate the arc can cause small, deep burns if concentrated at a point on the skin. Intense infrared and visible radiation from a gas welding or cutting flame and incandescent metal in the weld pool can cause discomfort to the operator and persons in the vicinity of the operation. Each operation should be considered in advance, and necessary precautions designed and implemented. Goggles made specifically for gas welding and cutting should be worn to protect the eyes from heat and light radiated from the work. Protective covers over filter glass should be cleaned as required and replaced when scratched or damaged. Where molten metal or hot particles are emitted, the protective clothing being worn should deflect spatter. The type and thickness of fire-resistant clothing worn should be chosen according to the degree of hazard. In cutting and arc welding operations, leather shoe coverings or other suitable spats should be worn to prevent hot particles from falling into boots or shoes. For protecting the hands and forearms against heat, spatter, slag and so on, the leather gauntlet type of glove with canvas or leather cuffs is sufficient. Other types of protective clothing include leather aprons, jackets, sleeves, leggings and head covering. In overhead welding, a protective cape and cap are necessary. All protective clothing should be free from oil or grease, and seams should be inside, so as not to trap globules of molten metal. Clothing should not have pockets or cuffs that could trap sparks, and it should be worn so sleeves overlap gloves, leggings overlap shoes and so on. Protective clothing should be inspected for burst seams or holes through which molten metal or slag may enter. Heavy articles left hot on completion of welding should always be marked “hot” as a warning to other workers. With resistance welding, the heat produced may not be visible, and burns can result from handling of hot assemblies. Particles of hot or molten metal should not fly out of spot, seam or projection welds if conditions are correct, but non-flammable screens should be used and precautions taken. Screens also protect passers-by from eye burns. Loose parts should not be left in the throat of the machine because they are liable to be projected with some velocity.
Electrical safety
Although no-load voltages in manual arc welding are relatively low (about 80 V or less), welding currents are high, and transformer primary circuits present the usual hazards of equipment operated at power supply line voltage. The risk of electric shock should therefore not be ignored, especially in cramped spaces or in insecure positions.
Before welding commences, the grounding installation on arc welding equipment should always be checked. Cables and connections should be sound and of adequate capacity. A proper grounding clamp or bolted terminal should always be used. Where two or more welding machines are grounded to the same structure, or where other portable electric tools are also in use, grounding should be supervised by a competent person. The working position should be dry, secure and free from dangerous obstructions. A well-arranged, well-lighted, properly ventilated and tidy workplace is important. For work in confined spaces or dangerous positions, additional electrical protection (no-load, low-voltage devices) can be installed in the welding circuit, ensuring that only extremely low-voltage current is available at the electrode holder when welding is not taking place. (See discussion of confined spaces below.) Electrode holders in which the electrodes are held by a spring grip or screw thread are recommended. Discomfort due to heating can be reduced by effective heat insulation on that part of the electrode holder which is held in the hand. Jaws and connections of electrode holders should be cleaned and tightened periodically to prevent overheating. Provision should be made to accommodate the electrode holder safely when not in use by means of an insulated hook or a fully insulated holder. The cable connection should be designed so that continued flexing of the cable will not cause wear and failure of the insulation. Dragging of cables and plastic gas supply tubes (gas-shielded processes) across hot plates or welds must be avoided. The electrode lead should not come in contact with the job or any other earthed object (ground). Rubber tubes and rubber-covered cables must not be used anywhere near the high-frequency discharge, because the ozone produced will rot the rubber. Plastic tubes and polyvinyl chloride (PVC) covered cables should be used for all supplies from the transformer to the electrode holder. Vulcanized or tough rubber-sheathed cables are satisfactory on the primary side. Dirt and metallic or other conducting dust can cause a breakdown in the high-frequency discharge unit. To avoid this condition, the unit should be cleaned regularly by blowing-out with compressed air. Hearing protection should be worn when using compressed air for more than a few seconds. For electron-beam welding, the safety of the equipment used must be checked prior to each operation. To protect against electric shock, a system of interlocks must be fitted to the various cabinets. A reliable system of grounding of all units and control cabinets is necessary. For plasma welding equipment used for cutting heavy thicknesses, the voltages may be as high as 400 V and danger should be anticipated. The technique of firing the arc by a high-frequency pulse exposes the operator to the dangers of an unpleasant shock and a painful, penetrating high-frequency burn.
Ultraviolet radiation
The brilliant light emitted by an electric arc contains a high proportion of ultraviolet radiation. Even momentary exposure to bursts of arc flash, including stray flashes from other workers’ arcs, may produce a painful conjunctivitis (photo-ophthalmia) known as “arc eye” or “eye flash”. If any person is exposed to arc flash, immediate medical attention must be sought. Excessive exposure to ultraviolet radiation may also cause overheating and burning of the skin (sunburn effect). Precautions include:
- A shield or helmet fitted with correct grade of filter should be used (see the article “Eye and face protection” elsewhere in this Encyclopaedia). For the gas-shielded arc welding processes and carbon-arc cutting, flat handshields provide insufficient protection from reflected radiation; helmets should be used. Filtered goggles or eyeglasses with sideshields should be worn under the helmet to avoid exposure when the helmet is lifted up for inspection of the work. Helmets will also provide protection from spatter and hot slag. Helmets and handshields are provided with a filter glass and a protective cover glass on the outside. This should be regularly inspected, cleaned and replaced when scratched or damaged.
- The face, nape of the neck and other exposed parts of the body should be properly protected, especially when working close to other welders.
- Assistants should wear suitable goggles at a minimum and other PPE as the risk requires.
- All arc welding operations should be screened to protect other persons working nearby. Where the work is carried out at fixed benches or in welding shops, permanent screens should be erected where possible; otherwise, temporary screens should be used. All screens should be opaque, of sturdy construction and of a flame-resistant material.
- The use of black paints for the inside of welding booths has become an accepted practice, but the paint should produce a matte finish. Adequate ambient lighting should be provided to prevent eye strain leading to headaches and accidents.
- Welding booths and portable screens should be checked regularly to ensure that there is no damage which might result in the arc affecting persons working nearby.
Chemical hazards
Airborne contaminants from welding and flame cutting, including fumes and gases, arise from a variety of sources:
- the metal being welded, the metal in the filler rod or constituents of various types of steel such as nickel or chromium)
- any metallic coating on the article being welded or on the filler rod (e.g., zinc and cadmium from plating, zinc from galvanizing and copper as a thin coating on continuous mild steel filler rods)
- any paint, grease, debris and the like on the article being welded (e.g., carbon monoxide, carbon dioxide, smoke and other irritant breakdown products)
- flux coating on the filler rod (e.g., inorganic fluoride)
- the action of heat or ultraviolet light on the surrounding air (e.g., nitrogen dioxide, ozone) or on chlorinated hydrocarbons (e.g., phosgene)
- inert gas used as a shield (e.g., carbon dioxide, helium, argon).
Fumes and gases should be removed at the source by LEV. This can be provided by partial enclosure of the process or by the installation of hoods which supply sufficiently high air velocity across the weld position so as to ensure capture of the fumes.
Special attention should be paid to ventilation in the welding of non-ferrous metals and certain alloy steels, as well as to protection from the hazard of ozone, carbon monoxide and nitrogen dioxide which may be formed. Portable as well as fixed ventilation systems are readily available. In general, the exhausted air should not be recirculated. It should be recirculated only if there are not hazardous levels of ozone or other toxic gases and the exhaust air is filtered through a high-efficiency filter.
With electron-beam welding and if materials being welded are of a toxic nature (e.g., beryllium, plutonium and so on), care must be taken to protect the operator from any dust cloud when opening the chamber.
When there is a risk to health from toxic fumes (e.g., lead) and LEV is not practicable—for example, when lead-painted structures are being demolished by flame cutting—the use of respiratory protective equipment is necessary. In such circumstances, an approved, high-efficiency full-facepiece respirator or ahigh-efficiency positive pressure powered air-purified respirator (PAPR) should be worn. A high standard of maintenance of the motor and the battery is necessary, especially with the original high-efficiency positive pressure power respirator. The use of positive pressure compressed air line respirators should be encouraged where a suitable supply of breathing-quality compressed air is available. Whenever respiratory protective equipment is to be worn, the safety of the workplace should be reviewed to determine whether extra precautions are necessary, bearing in mind the restricted vision, entanglement possibilities and so on of persons wearing respiratory protective equipment.
Metal fume fever
Metal fume fever is commonly seen in workers exposed to the fumes of zinc in the galvanizing or tinning process, in brass founding, in the welding of galvanized metal and in metallizing or metal spraying, as well as from exposure to other metals such as copper, manganese and iron. It occurs in new workers and those returning to work after a weekend or holiday hiatus. It is an acute condition that occurs several hours after the initial inhalation of particles of a metal or its oxides. It starts with a bad taste in the mouth followed by dryness and irritation of the respiratory mucosa resulting in cough and occasionally dyspnoea and “tightness” of the chest. These may be accompanied by nausea and headache and, some 10 to 12 hours after the exposure, chills and fever which may be quite severe. These last several hours and are followed by sweating, sleep and often by polyuria and diarrhoea. There is no particular treatment, and recovery is usually complete in about 24 hours with no residua. It can be prevented by keeping exposure to the offending metallic fumes well within the recommended levels through the use of efficient LEV.
Confined spaces
For entry into confined spaces, there may be a risk of the atmosphere being explosive, toxic, oxygen deficient or combinations of the above. Any such confined space must be certified by a responsible person as safe for entry and for work with an arc or flame. A confined-space entry programme, including an entry permit system, may be required and is highly recommended for work that must be carried out in spaces that are typically not constructed for continuous occupancy. Examples include, but are not limited to, manholes, vaults, ship holds and the like. Ventilation of confined spaces is crucial, since gas welding not only produces airborne contaminants but also uses up oxygen. Gas-shielded arc welding processes can decrease the oxygen content of the air. (See figure 2.)
Figure 2. Welding in an enclosed space
S. F. Gilman
Noise
Noise is a hazard in several welding processes, including plasma welding, some types of resistance welding machines and gas welding. In plasma welding, the plasma jet is ejected at very high speeds, producing intense noise (up to 90 dBA), particularly in the higher frequency bands. The use of compressed air to blow off dust also creates high noise levels. To prevent hearing damage, ear plugs or muffs must be worn and a hearing conservation programme should be instituted, including audiometric (hearing capacity) examinations and employee training.
Ionizing radiation
In welding shops where welds are inspected radiographically with x-ray or gamma-ray equipment, the customary warning notices and instructions must be strictly observed. Workers must be kept at a safe distance from such equipment. Radioactive sources must be handled only with the required special tools and subject to special precautions.
Local and governmental regulations must be followed. See the chapter Radiation, ionizing elsewhere in this Encyclopaedia.
Sufficient shielding must be provided with electron-beam welding to prevent x rays from penetrating the walls and windows of the chamber. Any parts of the machine providing shields against x-ray radiation should be interlocked so that the machine cannot be energized unless they are in position. Machines should be checked at the time of installation for leaks of x-ray radiation, and regularly thereafter.
Other hazards
Resistance welding machines have at least one electrode, which moves with considerable force. If a machine is operated while a finger or hand is lying between the electrodes, severe crushing will result. Where possible, a suitable means of guarding must be devised to safeguard the operator. Cuts and lacerations can be minimized by first deburring components and by wearing protective gloves or gauntlets.
Lockout/tagout procedures should be used when machinery with electrical, mechanical or other energy sources is being maintained or repaired.
When slag is being removed from welds by chipping and so on, the eyes should be protected by goggles or other means.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."