48. Radiation: Ionizing
Chapter Editor: Robert N. Cherry, Jr.
Table of Contents
Introduction
Robert N. Cherry, Jr.
Radiation Biology and Biological Effects
Arthur C. Upton
Sources of Ionizing Radiation
Robert N. Cherry, Jr.
Workplace Design for Radiation Safety
Gordon M. Lodde
Radiation Safety
Robert N. Cherry, Jr.
Planning for and Management of Radiation Accidents
Sydney W. Porter, Jr.
Introduction
Ionizing radiation is everywhere. It arrives from outer space as cosmic rays. It is in the air as emissions from radioactive radon and its progeny. Naturally occurring radioactive isotopes enter and remain in all living things. It is inescapable. Indeed, all species on this planet evolved in the presence of ionizing radiation. While humans exposed to small doses of radiation may not immediately show any apparent biological effects, there is no doubt that ionizing radiation, when given in sufficient amounts, can cause harm. These effects are well known both in kind and in degree.
While ionizing radiation can cause harm, it also has many beneficial uses. Radioactive uranium generates electricity in nuclear power plants in many countries. In medicine, x rays produce radiographs for diagnosis of internal injuries and diseases. Nuclear medicine physicians use radioactive material as tracers to form detailed images of internal structures and to study metabolism. Therapeutic radiopharmaceuticals are available to treat disorders such as hyperthyroidism and cancer. Radiotherapy physicians use gamma rays, pion beams, electron beams, neutrons and other types of radiation to treat cancer. Engineers use radioactive material in oil well logging operations and in soil moisture density gauges. Industrial radiographers use x rays in quality control to look at internal structures of manufactured devices. Exit signs in buildings and aircraft contain radioactive tritium to make them glow in the dark in the event of a power failure. Many smoke detectors in homes and commercial buildings contain radioactive americium.
These many uses of ionizing radiation and radioactive materials enhance the quality of life and help society in many ways. The benefits of each use must always be compared with the risks. The risks may be to workers directly involved in applying the radiation or radioactive material, to the public, to future generations and to the environment or to any combination of these. Beyond political and economic considerations, benefits must always outweigh risks when ionizing radiation is involved.
Ionizing Radiation
Ionizing radiation consists of particles, including photons, which cause the separation of electrons from atoms and molecules. However, some types of radiation of relatively low energy, such as ultraviolet light, can also cause ionization under certain circumstances. To distinguish these types of radiation from radiation that always causes ionization, an arbitrary lower energy limit for ionizing radiation usually is set around 10 kiloelectron volts (keV).
Directly ionizing radiation consists of charged particles. Such particles include energetic electrons (sometimes called negatrons), positrons, protons, alpha particles, charged mesons, muons and heavy ions (ionized atoms). This type of ionizing radiation interacts with matter primarily through the Coulomb force, repelling or attracting electrons from atoms and molecules by virtue of their charges.
Indirectly ionizing radiation consists of uncharged particles. The most common kinds of indirectly ionizing radiation are photons above 10 keV (x rays and gamma rays) and all neutrons.
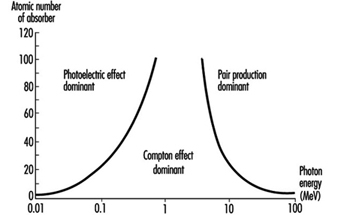
X-ray and gamma-ray photons interact with matter and cause ionization in at least three different ways:
- Lower-energy photons interact mostly via the photoelectric effect, in which the photon gives all of its energy to an electron, which then leaves the atom or molecule. The photon disappears.
- Intermediate-energy photons mostly interact through the Compton effect, in which the photon and an electron essentially collide as particles. The photon continues in a new direction with reduced energy while the released electron goes off with the remainder of the incoming energy (less the electron’s binding energy to the atom or molecule).
- Pair production is possible only for photons with energy in excess of 1.02 MeV. (However, near 1.02 MeV, the Compton effect still dominates. Pair production dominates at higher energies.) The photon disappears and an electron-positron pair appears in its place (this occurs only in the vicinity of a nucleus because of conservation of momentum and energy considerations). The total kinetic energy of the electron-positron pair is equal to the energy of the photon less the sum of the rest-mass energies of the electron and positron (1.02 MeV). These energetic electrons and positrons then proceed as directly ionizing radiation. As it loses kinetic energy, a positron will eventually encounter an electron, and the particles will annihilate each other. Two (usually) 0.511 MeV photons are then emitted from the annihilation site at 180 degrees from each other.
a given photon any of these can occur, except that pair production is possible only for photons with energy greater than 1.022 MeV. The energy of the photon and the material with which it interacts determine which interaction is the most likely to occur.
Figure 1 shows the regions in which each type of photon interaction dominates as a function of photon energy and atomic number of absorber.
Figure 1. Relative importance of the three principal interactions of photons in matter
The most common neutron interactions with matter are inelastic collisions, neutron capture (or activation) and fission. All of these are interactions with nuclei. A nucleus colliding inelastically with a neutron is left at a higher energy level. It can release this energy in the form of a gamma ray or by emitting a beta particle, or both. In neutron capture, an affected nucleus may absorb the neutron and eject energy as gamma or x rays or beta particles, or both. The secondary particles then cause ionization as discussed above. In fission, a heavy nucleus absorbs the neutron and splits into two lighter nuclei that are almost always radioactive.
Quantities, Units and Related Definitions
The International Commission on Radiation Units and Measurements (ICRU) develops internationally accepted formal definitions of quantities and units of radiation and radioactivity. The International Commission on Radiological Protection (ICRP) also sets standards for definition and use of various quantities and units used in radiation safety. A description of some quantities, units and definitions commonly used in radiation safety follows.
Absorbed dose. This is the fundamental dosimetric quantity for ionizing radiation. Basically, it is the energy ionizing radiation imparts to matter per unit mass. Formally,
![]()
where D is the absorbed dose, de is the mean energy imparted to matter of mass dm. Absorbed dose has units of joules per kilogram (J kg–1). The special name for the unit of absorbed dose is the gray (Gy).
Activity. This quantity represents the number of nuclear transformations from a given nuclear energy state per unit time. Formally,
![]()
where A is the activity, dN is the expectation value of the number of spontaneous nuclear transitions from the given energy state in the time interval dt. It is related to the number of radioactive nuclei N by:
![]()
where l is the decay constant. Activity has units of inverse seconds (s–1). The special name for the unit of activity is the becquerel (Bq).
Decay constant (l). This quantity represents the probability per unit time that a nuclear transformation will occur for a given radionuclide. The decay constant has units of inverse seconds (s–1). It is related to the half-life t½ of a radionuclide by:
![]()
The decay constant l is related to the mean lifetime, t, of a radionuclide by:
![]()
The time dependence of activity A(t) and of the number of radioactive nuclei N(t) can be expressed by ![]() and
and ![]() respectively.
respectively.
Deterministic biological effect. This is a biological effect caused by ionizing radiation and whose probability of occurrence is zero at small absorbed doses but will increase steeply to unity (100%) above some level of absorbed dose (the threshold). Cataract induction is an example of a stochastic biological effect.
Effective dose. The effective dose E is the sum of the weighted equivalent doses in all the tissues and organs of the body. It is a radiation safety quantity, so its use is not appropriate for large absorbed doses delivered in a relatively short period of time. It is given by:
![]()
where w T is the tissue weighting factor and HT is the equivalent dose for tissue T. Effective dose has units of J kg–1. The special name for the unit of effective dose is the sievert (Sv).
Equivalent dose. The equivalent dose HT is the absorbed dose averaged over a tissue or organ (rather than at a point) and weighted for the radiation quality that is of interest. It is a radiation safety quantity, so its use is not appropriate for large absorbed doses delivered in a relatively short period of time. The equivalent dose is given by:
![]()
where DT,R is the absorbed dose averaged over the tissue or organ T due to radiation R and w R
is the radiation weighting factor. Equivalent dose has units of J kg–1. The special name for the unit of equivalent dose is the sievert (Sv).
Half-life. This quantity is the amount of time required for the activity of a radionuclide sample to reduce by a factor of one-half. Equivalently, it is the amount of time required for a given number of nuclei in a given radioactive state to reduce by a factor of one-half. It has fundamental units of seconds (s), but is also commonly expressed in hours, days and years. For a given radionuclide, half-life t½ is related to the decay constant l by:
![]()
Linear energy transfer. This quantity is the energy a charged particle imparts to matter per unit length as it traverses the matter. Formally,
![]()
where L is the linear energy transfer (also called linear collision stopping power) and de is the mean energy lost by the particle in traversing a distance dl. Linear energy transfer (LET) has units of J m–1.
Mean lifetime. This quantity is the average time a nuclear state will survive before it undergoes a transformation to a lower energy state by emitting ionizing radiation. It has fundamental units of seconds (s), but may also be expressed in hours, days or years. It is related to the decay constant by:
![]()
where t is the mean lifetime and l is the decay constant for a given nuclide in a given energy state.
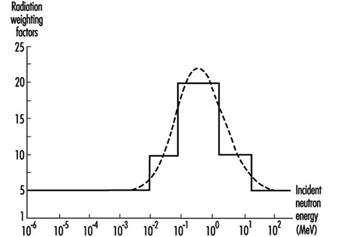
Radiation weighting factor. This is a number w R that, for a given type and energy of radiation R, is representative of values of the relative biological effectiveness of that radiation in inducing stochastic effects at low doses. The values of w R are related to linear energy transfer (LET) and are given in table 1. Figure 2 (overleaf) shows the relationship between w R and LET for neutrons.
Table 1. Radiation weighting factors wR
|
Type and energy range |
wR 1 |
|
Photons, all energies |
1 |
|
Electrons and muons, all energies2 |
1 |
|
Neutrons, energy 10 keV |
5 |
|
10 keV to 100 keV |
10 |
|
>100 keV to 2 MeV |
20 |
|
>2 MeV to 20 MeV |
10 |
|
>20 MeV |
5 |
|
Protons, other than recoil protons, energy >2 MeV |
5 |
|
Alpha particles, fission fragments, heavy nuclei |
20 |
1 All values relate to the radiation incident on the body or, for internal sources, emitted from the source.
2 Excluding Auger electrons emitted from nuclei bound to DNA.
Relative biological effectiveness (RBE). The RBE of one type of radiation compared with another is the inverse ratio of the absorbed doses producing the same degree of a defined biological end point.
Figure 2. Radiation weighting factors for neutrons (the smooth curve is to be treated as an approximation)
Stochastic biological effect. This is a biological effect caused by ionizing radiation whose probability of occurrence increases with increasing absorbed dose, probably with no threshold, but whose severity is independent of absorbed dose. Cancer is an example of a stochastic biological effect.
Tissue weighting factor w T. This represents the contribution of tissue or organ T to the total detriment due to all of the stochastic effects resulting from uniform irradiation of the whole body. It is used because the probability of stochastic effects due to an equivalent dose depends on the tissue or organ irradiated. A uniform equivalent dose over the whole body should give an effective dose numerically equal to the sum of effective doses for all tissues and organs of the body. Therefore, the sum of all tissue weighting factors is normalized to unity. Table 2 gives values for tissue weighting factors.
Table 2. Tissue weighting factors wT
|
Tissue or organ |
wT 1 |
|
Gonads |
0.20 |
|
Bone marrow (red) |
0.12 |
|
Colon |
0.12 |
|
Lung |
0.12 |
|
Stomach |
0.12 |
|
Bladder |
0.05 |
|
Breast |
0.05 |
|
Liver |
0.05 |
|
Oesophagus |
0.05 |
|
Thyroid |
0.05 |
|
Skin |
0.01 |
|
Bone surface |
0.01 |
|
Remainder |
0.052, 3 |
1 The values have been developed from a reference population of equal numbers of both sexes and a wide range of ages. In the definition of effective dose they apply to workers, to the whole population, and to either sex.
2 For purposes of calculation, the remainder is composed of the following additional tissues and organs: adrenals, brain, upper large intestine, small intestine, kidneys, muscle, pancreas, spleen, thymus and uterus. The list includes organs that are likely to be selectively irradiated. Some organs in the list are known to be susceptible to cancer induction.
3 In those exceptional cases in which a single one of the remainder tissues or organs receives an equivalent dose in excess of the highest dose in any of the twelve organs for which a weighting factor is specified, a weighting factor of 0.025 should be applied to that tissue or organ and a weighting factor of 0.025 to the average dose in the rest of the remainder as defined above.
Radiation Biology and Biological Effects
After its discovery by Roentgen in 1895, the x ray was introduced so rapidly into the diagnosis and treatment of disease that injuries from excessive radiation exposure began to be encountered almost immediately in pioneer radiation workers, who had yet to become aware of the dangers (Brown 1933). The first such injuries were predominantly skin reactions on the hands of those working with the early radiation equipment, but within a decade many other types of injury also had been reported, including the first cancers attributed to radiation (Stone 1959).
Throughout the century since these early findings, study of the biological effects of ionizing radiation has received continuing impetus from the growing uses of radiation in medicine, science and industry, as well as from the peaceful and military applications of atomic energy. As a result, the biological effects of radiation have been investigated more thoroughly than those of virtually any other environmental agent. The evolving knowledge of radiation effects has been influential in shaping measures for the protection of human health against many other environmental hazards as well as radiation.
Nature and Mechanisms of the Biological Effects of Radiation
Energy deposition. In contrast to other forms of radiation, ionizing radiation is capable of depositing enough localized energy to dislodge electrons from the atoms with which it interacts. Thus, as radiation collides randomly with atoms and molecules in passing through living cells, it gives rise to ions and free radicals which break chemical bonds and cause other molecular changes that injure the affected cells. The spatial distribution of the ionizing events depends on the radiation weighting factor, w R of the radiation (see table 1 and figure 1).
Table 1. Radiation weighting factors wR
|
Type and energy range |
wR 1 |
|
Photons, all energies |
1 |
|
Electrons and muons, all energies2 |
1 |
|
Neutrons, energy <10 keV |
5 |
|
10 keV to 100 keV |
10 |
|
>100 keV to 2 MeV |
20 |
|
>2 MeV to 20 MeV |
10 |
|
>20 MeV |
5 |
|
Protons, other than recoil protons, energy >2 MeV |
5 |
|
Alpha particles, fission fragments, heavy nuclei |
20 |
1 All values relate to the radiation incident on the body or, for internal sources, emitted from the source.
2 Excluding Auger electrons emitted from nuclei bound to DNA.
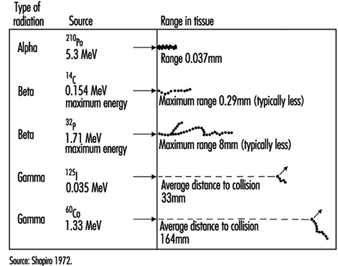
Figure 1. Differences among various types of ionizing radiation in penetrating power in tissue
Effects on DNA. Any molecule in the cell may be altered by radiation, but DNA is the most critical biological target because of the limited redundancy of the genetic information it contains. An absorbed dose of radiation large enough to kill the average dividing cell—2 gray (Gy)—suffices to cause hundreds of lesions in its DNA molecules (Ward 1988). Most such lesions are reparable, but those produced by a densely ionizing radiation (for example, a proton or an alpha particle) are generally less reparable than those produced by a sparsely ionizing radiation (for example, an x ray or a gamma ray) (Goodhead 1988). Densely ionizing (high LET) radiations, therefore, typically have a higher relative biological effectiveness (RBE) than sparsely ionizing (low LET) radiations for most forms of injury (ICRP 1991).
Effects on genes. Damage to DNA that remains unrepaired or is misrepaired may be expressed in the form of mutations, the frequency of which appears to increase as a linear, non-threshold function of the dose, approximately 10–5 to 10–6 per locus per Gy (NAS 1990). The fact that the mutation rate appears to be proportional to the dose is interpreted to signify that traversal of the DNA by a single ionizing particle may, in principle, suffice to cause a mutation (NAS 1990). In Chernobyl accident victims, the dose-response relationship for glycophorin mutations in bone marrow cells closely resembles that observed in atomic bomb survivors (Jensen, Langlois and Bigbee 1995).
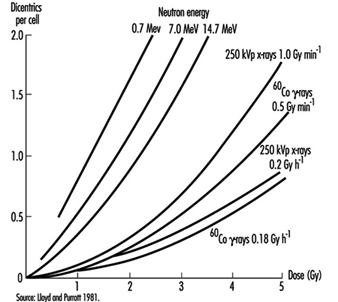
Effects on chromosomes. Radiation damage to the genetic apparatus may also cause changes in chromosome number and structure, the frequency of which has been observed to increase with the dose in radiation workers, atomic bomb survivors, and others exposed to ionizing radiation. The dose-response relationship for chromosome aberrations in human blood lymphocytes (figure 2) has been characterized well enough so that the frequency of aberrations in such cells can serve as a useful biological dosimeter (IAEA 1986).
Figure 2. Frequency of dicentric chromosome aberrations in human lymphocytes in relation to dose, dose rate, and quality of irradiation in vitro
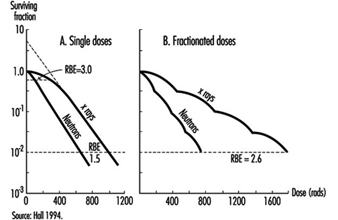
Effects on cell survival. Among the earliest reactions to irradiation is the inhibition of cell division, which appears promptly after exposure, varying both in degree and duration with the dose (figure 3). Although the inhibition of mitosis is characteristically transitory, radiation damage to genes and chromosomes may be lethal to dividing cells, which are highly radiosensitive as a class (ICRP 1984). Measured in terms of proliferative capacity, the survival of dividing cells tends to decrease exponentially with increasing dose, 1 to 2 Gy generally sufficing to reduce the surviving population by about 50% (figure 4).
Figure 3. Mitotic inhibition induced by x rays in rat corneal epithelial cells
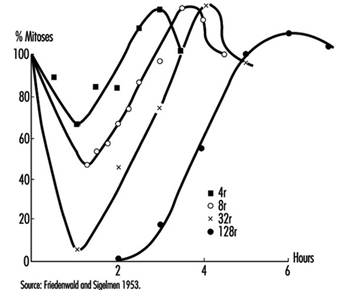
Figure 4. Typical dose-survival curves for mammalian cells exposed to x rays and fast neutrons
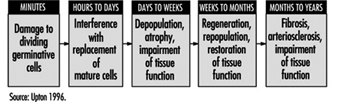
Effects on tissues. Mature, non-dividing cells are relatively radioresistant, but the dividing cells in a tissue are radiosensitive and may be killed in sufficient numbers by intensive irradiation to cause the tissue to become atrophic (figure 5). The rapidity of such atrophy depends on cell population dynamics within the affected tissue; that is, in organs characterized by slow cell turnover, such as the liver and vascular endothelium, the process is typically much slower than in organs characterized by rapid cell turnover, such as the bone marrow, epidermis and intestinal mucosa (ICRP 1984). It is noteworthy, moreover, that if the volume of tissue irradiated is sufficiently small, or if the dose is accumulated gradually enough, the severity of injury may be greatly reduced by the compensatory proliferation of surviving cells.
Figure 5. Characteristic sequence of events in the pathogenesis of nonstochastic effects of ionizing radiation
Clinical Manifestations of Injury
Types of effects. Radiation effects encompass a wide variety of reactions, varying markedly in their dose-response relationships, clinical manifestations, timing and prognosis (Mettler and Upton 1995). The effects are often subdivided, for convenience, into two broad categories: (1) heritable effects, which are expressed in the descendants of exposed individuals, and (2) somatic effects, which are expressed in exposed individuals themselves. The latter include acute effects, which occur relatively soon after irradiation, as well as late (or chronic) effects, such as cancer, which may not appear until months, years or decades later.
Acute effects. The acute effects of radiation result predominantly from the depletion of progenitor cells in affected tissues (figure 5) and can be elicited only by doses that are large enough to kill many such cells (for example, table 2). For this reason, such effects are viewed as nonstochastic, or deterministic, in nature (ICRP 1984 and 1991), in contradistinction to the mutagenic and carcinogenic effects of radiation, which are viewed as stochastic phenomena resulting from random molecular alterations in individual cells that increase as linear-nonthreshold functions of the dose (NAS 1990; ICRP 1991).
Table 2. Approximate threshold doses of conventionally fractionated therapeutic x-radiation for clinically detrimental nonstochastic effects in various tissues
|
Organ |
Injury at 5 years |
Threshold |
Irradiation |
|
Skin |
Ulcer, severe fibrosis |
55 |
100 cm2 |
|
Oral mucosa |
Ulcer, severe fibrosis |
60 |
50 cm2 |
|
Oesophagus |
Ulcer, stricture |
60 |
75 cm2 |
|
Stomach |
Ulcer, perforation |
45 |
100 cm2 |
|
Small intestine |
Ulcer, stricture |
45 |
100 cm2 |
|
Colon |
Ulcer, stricture |
45 |
100 cm2 |
|
Rectum |
Ulcer, stricture |
55 |
100 cm2 |
|
Salivary glands |
Xerostomia |
50 |
50 cm2 |
|
Liver |
Liver failure, ascites |
35 |
whole |
|
Kidney |
Nephrosclerosis |
23 |
whole |
|
Urinary bladder |
Ulcer, contracture |
60 |
whole |
|
Testes |
Permanent sterility |
5-15 |
whole |
|
Ovary |
Permanent sterility |
2-3 |
whole |
|
Uterus |
Necrosis, perforation |
>100 |
whole |
|
Vagina |
Ulcer, fistula |
90 |
5 cm2 |
|
Breast, child |
Hypoplasia |
10 |
5 cm2 |
|
Breast, adult |
Atrophy, necrosis |
>50 |
whole |
|
Lung |
Pneumonitis, fibrosis |
40 |
lobe |
|
Capillaries |
Telangiectasis, fibrosis |
50-60 |
s |
|
Heart |
Pericarditis, pancarditis |
40 |
whole |
|
Bone, child |
Arrested growth |
20 |
10 cm2 |
|
Bone, adult |
Necrosis, fracture |
60 |
10 cm2 |
|
Cartilage, child |
Arrested growth |
10 |
whole |
|
Cartilage, adult |
Necrosis |
60 |
whole |
|
Central nervous system (brain) |
Necrosis |
50 |
whole |
|
Spinal cord |
Necrosis, transection |
50 |
5 cm2 |
|
Eye |
Panophthalmitis, haemorrhage |
55 |
whole |
|
Cornea |
Keratitis |
50 |
whole |
|
Lens |
Cataract |
5 |
whole |
|
Ear (inner) |
Deafness |
>60 |
whole |
|
Thyroid |
Hypothyroidism |
45 |
whole |
|
Adrenal |
Hypoadrenalism |
>60 |
whole |
|
Pituitary |
Hypopituitarism |
45 |
whole |
|
Muscle, child |
Hypoplasia |
20-30 |
whole |
|
Muscle, adult |
Atrophy |
>100 |
whole |
|
Bone marrow |
Hypoplasia |
2 |
whole |
|
Bone marrow |
Hypoplasia, fibrosis |
20 |
localized |
|
Lymph nodes |
Atrophy |
33-45 |
s |
|
Lymphatics |
Sclerosis |
50 |
s |
|
Foetus |
Death |
2 |
whole |
* Dose causing effect in 1-5 per cent of exposed persons.
Source: Rubin and Casarett 1972.
Acute injuries of the types that were prevalent in pioneer radiation workers and early radiotherapy patients have been largely eliminated by improvements in safety precautions and treatment methods. Nevertheless, most patients treated with radiation today still experience some injury of the normal tissue that is irradiated. In addition, serious radiation accidents continue to occur. For example, some 285 nuclear reactor accidents (excluding the Chernobyl accident) were reported in various countries between 1945 and 1987, irradiating more than 1,350 persons, 33 of them fatally (Lushbaugh, Fry and Ricks 1987). The Chernobyl accident alone released enough radioactive material to require the evacuation of tens of thousands of people and farm animals from the surrounding area, and it caused radiation sickness and burns in more than 200 emergency personnel and fire-fighters, injuring 31 fatally (UNSCEAR 1988). The long-term health effects of the radioactive material released cannot be predicted with certainty, but estimates of the resulting risks of carcinogenic effects, based on nonthreshold dose-incidence models (discussed below), imply that up to 30,000 additional cancer deaths may occur in the population of the northern hemisphere during the next 70 years as a result of the accident, although the additional cancers in any given country are likely to be too few to be detectable epidemiologically (USDOE 1987).
Less catastrophic, but far more numerous, than reactor accidents have been accidents involving medical and industrial gamma ray sources, which also have caused injuries and loss of life. For example, the improper disposal of a caesium-137 radiotherapy source in Goiânia, Brazil, in 1987, resulted in the irradiation of dozens of unsuspecting victims, four of them fatally (UNSCEAR 1993).
A comprehensive discussion of radiation injuries is beyond the scope of this review, but acute reactions of the more radiosensitive tissues are of widespread interest and are, therefore, described briefly in the following sections.
Skin. Cells in the germinal layer of the epidermis are highly radiosensitive. As a result, rapid exposure of the skin to a dose of 6 Sv or more causes erythema (reddening) in the exposed area, which appears within a day or so, typically lasts a few hours, and is followed two to four weeks later by one or more waves of deeper and more prolonged erythema, as well as by epilation (hair loss). If the dose exceeds 10 to 20 Sv, blistering, necrosis and ulceration may ensue within two to four weeks, followed by fibrosis of the underlying dermis and vasculature, which may lead to atrophy and a second wave of ulceration months or years later (ICRP 1984).
Bone marrow and lymphoid tissue. Lymphocytes also are highly radiosensitive; a dose of 2 to 3 Sv delivered rapidly to the whole body can kill enough of them to depress the peripheral lymphocyte count and impair the immune response within hours (UNSCEAR 1988). Haemopoietic cells in the bone marrow are similarly radiosensitive and are depleted sufficiently by a comparable dose to cause granulocytopenia and thrombocytopenia to ensue within three to five weeks. Such reductions in granulocyte and platelet counts may be severe enough after a larger dose to result in haemorrhage or fatal infection (table 3).
Table 3. Major forms and features of the acute radiation syndrome
|
Time after |
Cerebral form |
Gastro- |
Hemopoietic form |
Pulmonary form |
|
First day |
nausea |
nausea |
nausea |
nausea |
|
Second week |
nausea |
|||
|
Third to sixth |
weakness |
|||
|
Second to eighth |
cough |
Source: UNSCEAR 1988.
Intestine. Stem cells in the epithelium lining the small bowel also are extremely radiosensitive, acute exposure to 10 Sv depleting their numbers sufficiently to cause the overlying intestinal villi to become denuded within days (ICRP 1984; UNSCEAR 1988). Denudation of a large area of the mucosa can result in a fulminating, rapidly fatal dysentery-like syndrome (table 3).
Gonads. Mature spermatozoa can survive large doses (100 Sv), but spermatogonia are so radiosensitive that as little as 0.15 Sv delivered rapidly to both testes suffices to cause oligospermia, and a dose of 2 to 4 Sv can cause permanent sterility. Oocytes, likewise, are radiosensitive, a dose of 1.5 to 2.0 Sv delivered rapidly to both ovaries causing temporary sterility, and a larger dose, permanent sterility, depending on the age of the woman at the time of exposure (ICRP 1984).
Respiratory tract. The lung is not highly radiosensitive, but rapid exposure to a dose of 6 to 10 Sv can cause acute pneumonitis to develop in the exposed area within one to three months. If a large volume of lung tissue is affected, the process may result in respiratory failure within weeks, or may lead to pulmonary fibrosis and cor pulmonale months or years later (ICRP 1984; UNSCEAR 1988).
Lens of the eye. Cells of the anterior epithelium of the lens, which continue to divide throughout life, are relatively radiosensitive. As a result, rapid exposure of the lens to a dose exceeding 1 Sv may lead within months to the formation of a microscopic posterior polar opacity; and 2 to 3 Sv received in a single brief exposure—or 5.5 to 14 Sv accumulated over a period of months—may produce a vision-impairing cataract (ICRP 1984).
Other tissues. In comparison with the tissues mentioned above, other tissues of the body are generally appreciably less radiosensitive (for example, table 2); however, the embryo constitutes a notable exception, as discussed below. Noteworthy also is the fact that the radiosensitivity of every tissue is increased when it is in a rapidly growing state (ICRP 1984).
Whole-body radiation injury. Rapid exposure of a major part of the body to a dose in excess of 1 Gy can cause the acute radiation syndrome. This syndrome includes: (1) an initial prodromal stage, characterized by malaise, anorexia, nausea and vomiting, (2) an ensuing latent period, (3) a second (main) phase of illness and (4) ultimately, either recovery or death (table 3). The main phase of the illness typically takes one of the following forms, depending on the predominant locus of radiation injury: (1) haematological, (2) gastro-intestinal, (3) cerebral or (4) pulmonary (table 3).
Localized radiation injury. Unlike the clinical manifestations of acute whole-body radiation injury, which typically are dramatic and prompt, the reaction to sharply localized irradiation, whether from an external radiation source or from an internally deposited radionuclide, tends to evolve slowly and to produce few symptoms or signs unless the volume of tissue irradiated and/or the dose are relatively large (for example, table 3).
Effects of radionuclides. Some radionuclides - for example, tritium (3H), carbon-14 (14C) and cesium-137 (137Cs) - tend to be distributed systemically and to irradiate the body as a whole, whereas other radionuclides are characteristically taken up and concentrated in specific organs, producing injuries that are correspondingly localized. Radium (Ra) and strontium-90
(90Sr), for example, are deposited predominantly in bone and thus injure skeletal tissues primarily, whereas radioactive iodine concentrates in the thyroid gland, the primary site of any resulting injury (Stannard 1988; Mettler and Upton 1995).
Carcinogenic Effects
General features. The carcinogenicity of ionizing radiation, first manifested early in this century by the occurrence of skin cancers and leukaemias in pioneer radiation workers (Upton 1986), has since been documented extensively by dose-dependent excesses of many types of neoplasms in radium-dial painters, underground hardrock miners, atomic bomb survivors, radiotherapy patients and experimentally irradiated laboratory animals (Upton 1986; NAS 1990).
The benign and malignant growths induced by irradiation characteristically take years or decades to appear and exhibit no known features by which they can be distinguished from those produced by other causes. With few exceptions, moreover, their induction has been detectable only after relatively large dose equivalents (0.5 Sv), and it has varied with the type of neoplasm as well as the age and sex of those exposed (NAS 1990).
Mechanisms. The molecular mechanisms of radiation carcinogenesis remain to be elucidated in detail, but in laboratory animals and cultured cells the carcinogenic effects of radiation have been observed to include initiating effects, promoting effects, and effects on the progression of neoplasia, depending on the experimental conditions in question (NAS 1990). The effects also appear to involve the activation of oncogenes and/or the inactivation or loss of tumor-suppressor genes in many, if not all, instances. In addition, the carcinogenic effects of radiation resemble those of chemical carcinogens in being similarly modifiable by hormones, nutritional variables and other modifying factors (NAS 1990). It is noteworthy, moreover, that the effects of radiation may be additive, synergistic or mutually antagonistic with those of chemical carcinogens, depending on the specific chemicals and exposure conditions in question (UNSCEAR 1982 and 1986).
Dose-effect relationship. Existing data do not suffice to describe the dose-incidence relationship unambiguously for any type of neoplasm or to define how long after irradiation the risk of the growth may remain elevated in an exposed population. Any risks attributable to low-level irradiation can, therefore, be estimated only by extrapolation, based on models incorporating assumptions about such parameters (NAS 1990). Of various dose-effect models that have been used to estimate the risks of low-level irradiation, the one that has been judged to provide the best fit to the available data is of the form:
![]()
where R0 denotes the age-specific background risk of death from a specific type of cancer, D the radiation dose, f(D) a function of dose that is linear-quadratic for leukaemia and linear for some other types of cancer, and g(b) is a risk function dependent on other parameters, such as sex, age at exposure and time after exposure (NAS 1990).
Non-threshold models of this type have been applied to epidemiological data from the Japanese atomic-bomb survivors and other irradiated populations to derive estimates of the lifetime risks of different forms of radiation-induced cancer (for example, table 4). Such estimates must be interpreted with caution, however, in attempting to predict the risks of cancer attributable to small doses or doses that are accumulated over weeks, months or years, since experiments with laboratory animals have shown the carcinogenic potency of x rays and gamma rays to be reduced by as much as an order of magnitude when the exposure is greatly prolonged. In fact, as has been emphasized elsewhere (NAS 1990), the available data do not exclude the possibility that there may be a threshold in the millisievert (mSv) dose equivalent range, below which radiation may lack carcinogenicity.
Table 4. Estimated lifetime risks of cancer attributable to 0.1 Sv rapid irradiation
|
Type or site of cancer |
Excess cancer deaths per 100,000 |
|
|
(No.) |
(%)* |
|
|
Stomach |
110 |
18 |
|
Lung |
85 |
3 |
|
Colon |
85 |
5 |
|
Leukaemia (excluding CLL) |
50 |
10 |
|
Urinary bladder |
30 |
5 |
|
Oesophagus |
30 |
10 |
|
Breast |
20 |
1 |
|
Liver |
15 |
8 |
|
Gonads |
10 |
2 |
|
Thyroid |
8 |
8 |
|
Osteosarcoma |
5 |
5 |
|
Skin |
2 |
2 |
|
Remainder |
50 |
1 |
|
Total |
500 |
2 |
* Percentage increase in “background” expectation for a non-irradiated population.
Source: ICRP 1991.
It is also noteworthy that the estimates tabulated are based on population averages and are not necessarily applicable to any given individual; that is, susceptibility to certain types of cancer (for example, cancers of the thyroid and breast) is substantially higher in children than in adults, and susceptibility to certain cancers is also increased in association with some hereditary disorders, such as retinoblastoma and the nevoid basal cell carcinoma syndrome (UNSCEAR 1988, 1994; NAS 1990). Such differences in susceptibility notwithstanding, population-based estimates have been proposed for use in compensation cases as a basis for gauging the probability that a cancer arising in a previously irradiated person may have been caused by the exposure in question (NIH 1985).
Low-dose risk assessment. Epidemiological studies to ascertain whether the risks of cancer from low-level exposure to radiation actually vary with dose in the manner predicted by the above estimates have been inconclusive thus far. Populations residing in areas of elevated natural background radiation levels manifest no definitely attributable increases in cancer rates (NAS 1990; UNSCEAR 1994); conversely, a few studies have even suggested an inverse relationship between background radiation levels and cancer rates, which has been interpreted by some observers as evidence for the existence of beneficial (or hormetic) effects of low-level irradiation, in keeping with the adaptive responses of certain cellular systems (UNSCEAR 1994). The inverse relationship is of questionable significance, however, since it has not persisted after controlling for the effects of confounding variables (NAS 1990). Likewise in today’s radiation workers—except for certain cohorts of underground hardrock miners (NAS 1994; Lubin, Boice and Edling 1994)—the rates of cancers other than leukaemia are no longer detectably increased (UNSCEAR 1994), thanks to advances in radiation protection; furthermore, the rates of leukaemia in such workers are consistent with the estimates tabulated above (IARC 1994). In summary, therefore, the data available at present are consistent with the estimates tabulated above (table 4), which imply that less than 3% of cancers in the general population are attributable to natural background radiation (NAS 1990; IARC 1994), although up to 10% of lung cancers may be attributable to indoor radon (NAS 1990; Lubin, Boice and Edling 1994).
High levels of radioactive fallout from a thermonuclear weapons test at Bikini in 1954 have been observed to cause a dose-dependent increase in the frequency of thyroid cancer in Marshall Islanders who received large doses to the thyroid gland in childhood (Robbins and Adams 1989). Similarly, children living in areas of Belarus and the Ukraine contaminated by radionuclides released from the Chernobyl accident have been reported to show an increased incidence of thyroid cancer (Prisyazhuik, Pjatak and Buzanov 1991; Kasakov, Demidchik and Astakhova 1992), but the findings are at variance with those of the International Chernobyl Project, which found no excess of benign or malignant thyroid nodules in children living in the more heavily contaminated areas around Chernobyl (Mettler, Williamson and Royal 1992). The basis for the discrepancy, and whether the reported excesses may have resulted from heightened surveillance alone, remain to be determined. In this connection, it is noteworthy that children of south-western Utah and Nevada who were exposed to fallout from nuclear weapons tests in Nevada during the 1950s have shown increase in the frequency of any type of thyroid cancer (Kerber et al. 1993), and the prevalence of acute leukaemia appears to have been elevated in such children dying between 1952 and 1957, the period of greatest exposure to fallout (Stevens et al. 1990).
The possibility that excesses of leukaemia among children residing in the vicinity of nuclear plants in the United Kingdom may have been caused by radioactivity released from the plants has also been suggested. The releases, however, are estimated to have increased the total radiation dose to such children by less than 2%, from which it is inferred that other explanations are more likely (Doll, Evans and Darby 1994). An ineffective aetiology for the observed clusters of leukaemia is implied by the existence of comparable excesses of childhood leukaemia at sites in the UK that lack nuclear facilities but otherwise resemble nuclear sites in having similarly experienced large influxes of population in recent times (Kinlen 1988; Doll, Evans and Darby 1994). Another hypothesis—namely, that the leukaemias in question may have been caused by occupational irradiation of the fathers of the affected children—also has been suggested by the results of a case-control study (Gardner et al. 1990), but this hypothesis is generally discounted for reasons that are discussed in the section to follow.
Heritable Effects
Heritable effects of irradiation, although well documented in other organisms, have yet to be observed in humans. For example, intensive study of more than 76,000 children of the Japanese atomic-bomb survivors, carried out over four decades, has failed to disclose any heritable effects of radiation in this population, as measured by untoward pregnancy outcomes, neonatal deaths, malignancies, balanced chromosomal rearrangements, sex-chromosome aneuploidy, alterations of serum or erythrocyte protein phenotypes, changes in sex ratio or disturbances in growth and development (Neel, Schull and Awa 1990). Consequently, estimates of the risks of heritable effects of radiation must rely heavily on extrapolation from findings in the laboratory mouse and other experimental animals (NAS 1990; UNSCEAR 1993).
From the available experimental and epidemiological data, it is inferred that the dose required to double the rate of heritable mutations in human germ cells must be at least 1.0 Sv (NAS 1990; UNSCEAR 1993). On this basis, it is estimated that less than 1% of all genetically determined diseases in the human population can be attributed to natural background irradiation (table 5).
Table 5. Estimated frequencies of heritable disorders attributable to natural background ionizing irradiation
|
Type of disorder |
Natural prevalence |
Contribution from natural background |
|
|
First generation |
Equilibrium |
||
|
Autosomal |
180,000 |
20-100 |
300 |
|
X-linked |
400 |
<1 |
<15 |
|
Recessive |
2,500 |
<1 |
very slow increase |
|
Chromosomal |
4,400 |
<20 |
very slow increase |
|
Congenital |
20,000-30,000 |
30 |
30-300 |
|
Other disorders of complex aetiology: |
|||
|
Heart disease |
600,000 |
not estimated4 |
not estimated4 |
|
Cancer |
300,000 |
not estimated4 |
not estimated4 |
|
Selected others |
300,000 |
not estimated4 |
not estimated4 |
1 Equivalent to » 1 mSv per year, or » 30 mSv per generation (30 years).
2 Values rounded.
3 After hundreds of generations, the addition of unfavorable radiation-induced mutations eventually becomes balanced by their loss from the population, resulting in a genetic "equilibrium".
4 Quantitative risk estimates are lacking because of uncertainty about the mutational component of the disease(s) indicated.
Source: National Research Council 1990.
The hypothesis that the excess of leukaemia and non-Hodgkin’s lymphoma in young people residing in the village of Seascale resulted from heritable oncogenic effects caused by the occupational irradiation of the children’s fathers at the Sellafield nuclear installation has been suggested by the results of a case-control study (Gardner et al. 1990), as noted above. Arguments against this hypothesis, however, are:
- the lack of any comparable excess in larger numbers of children born outside Seascale to fathers who had received similar, or even larger, occupational doses at the same nuclear plant (Wakeford et al. 1994a)
- the lack of similar excesses in French (Hill and LaPlanche 1990), Canadian (McLaughlin et al. 1993) or Scottish (Kinlen, Clarke and Balkwill 1993) children born to fathers with comparable occupational exposures
- the lack of excesses in the children of atomic-bomb survivors (Yoshimoto et al. 1990)
- the lack of excesses in US counties containing nuclear plants (Jablon, Hrubec and Boice 1991)
- the fact that the frequency of radiation-induced mutations implied by the interpretation is far higher than established rates (Wakeford et al. 1994b).
On balance, therefore, the available data fail to support the paternal gonadal irradiation hypothesis (Doll, Evans and Darby 1994; Little, Charles and Wakeford 1995).
Effects of Prenatal Irradiation
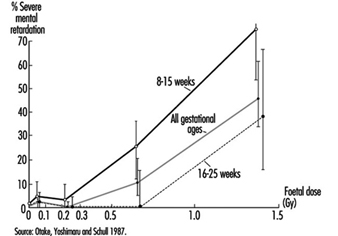
Radiosensitivity is relatively high throughout prenatal life, but the effects of a given dose vary markedly, depending on the developmental stage of the embryo or foetus at the time of exposure (UNSCEAR 1986). During the pre-implantation period, the embryo is most susceptible to killing by irradiation, while during critical stages in organogenesis it is susceptible to the induction of malformations and other disturbances of development (table 6). The latter effects are dramatically exemplified by the dose-dependent increase in the frequency of severe mental retardation (figure 6) and the dose-dependent decrease in IQ test scores in atomic-bomb survivors who were exposed between the eighth and fifteenth weeks (and, to a lesser extent, between the sixteenth and twenty-fifth weeks) (UNSCEAR 1986 and 1993).
Table 6. Major developmental abnormalities produced by prenatal irradiation
|
Brain |
||
|
Anencephaly |
Porencephaly |
Microcephaly* |
|
Encephalocoele |
Mongolism* |
Reduced medulla |
|
Cerebral atrophy |
Mental retardation* |
Neuroblastoma |
|
Narrow aqueduct |
Hydrocephalus* |
Dilatation of ventricles* |
|
Spinal cord anomalies* |
Cranial nerve anomalies |
|
|
Eyes |
||
|
Anophthalmia |
Microphthalmia* |
Microcornia* |
|
Coloboma* |
Deformed iris |
Absence of lens |
|
Absence of retina |
Open eyelids |
Strabismus* |
|
Nystagmus* |
Retinoblastoma |
Hypermetropia |
|
Glaucoma |
Cataract* |
Blindness |
|
Chorioretinitis* |
Partial albinism |
Ankyloblepharon |
|
Skeleton |
||
|
General stunting |
Reduced size of skull |
Skull deformities* |
|
Head ossification defects* |
Vaulted cranium |
Narrow head |
|
Cranial blisters |
Cleft palate* |
Funnel chest |
|
Dislocation of hip |
Spina bifida |
Deformed tail |
|
Deformed feet |
Club foot* |
Digital anomalies* |
|
Calcaneo valgus |
Odontogenesis imperfecta* |
Tibial exostosis |
|
Amelanogenesis* |
Scleratomal necrosis |
|
|
Miscellaneous |
||
|
Situs inversus |
Hydronephrosis |
Hydroureter |
|
Hydrocoele |
Absence of kidney |
Gonadal anomalies* |
|
Congenital heart disease |
Facial deformities |
Pituitary disturbances |
|
Deformities of ears |
Motor disturbances |
Dermatomal necrosis |
|
Myotomal necrosis |
Abnormalities in skinpigmentation |
|
* These abnormalities have been observed in humans exposed prenatally to large doses of radiation and have, therefore, been tentatively attributed to irradiation.
Source: Brill and Forgotson 1964.
Susceptibility to the carcinogenic effects of radiation also appears to be relatively high throughout the prenatal period, judging from the association between childhood cancer (including leukaemia) and prenatal exposure to diagnostic x rays reported in case-control studies (NAS 1990). The results of such studies imply that prenatal irradiation may cause a 4,000% per Sv increase in the risk of leukaemia and other childhood cancers (UNSCEAR 1986; NAS 1990), which is a far larger increase than is attributable to postnatal irradiation (UNSCEAR 1988; NAS 1990). Although, paradoxically, no excess of childhood cancer was recorded in A-bomb survivors irradiated prenatally (Yoshimoto et al. 1990), as noted above, there were too few such survivors to exclude an excess of the magnitude in question.
Figure 6. The frequency of severe mental retardation in relation to radiation dose in prenatally irradiated atomic bomb survivors
Summary and Conclusions
The adverse effects of ionizing radiation on human health are widely diverse, ranging from rapidly fatal injuries to cancers, birth defects, and hereditary disorders that appear months, years or decades later. The nature, frequency and severity of effects depend on the quality of the radiation in question as well as on the dose and conditions of exposure. Most such effects require relatively high levels of exposure and are, therefore, encountered only in accident victims, radiotherapy patients, or other heavily irradiated persons. The genotoxic and carcinogenic effects of ionizing radiation, by contrast, are presumed to increase in frequency as linear non-threshold functions of the dose; hence, although the existence of thresholds for these effects cannot be excluded, their frequency is assumed to increase with any level of exposure. For most effects of radiation, the sensitivity of exposed cells varies with their rate of proliferation and inversely with their degree of differentiation, the embryo and growing child being especially vulnerable to injury.
Sources of Ionizing Radiation
Ionizing Radiation Types
Alpha particles
An alpha particle is a tightly bound collection of two protons and two neutrons. It is identical to a helium-4 (4He) nucleus. Indeed, its ultimate fate after it loses most of its kinetic energy is to capture two electrons and become a helium atom.
Alpha-emitting radionuclides are generally relatively massive nuclei. Almost all alpha emitters have atomic numbers greater than or equal to that of lead (82Pb). When a nucleus decays by emitting an alpha particle, both its atomic number (number of protons) and its number of neutrons are reduced by two and its atomic mass number is reduced by four. For example, the alpha decay of uranium-238 (238U) to thorium-234 (234Th) is represented by:
![]()
![]()
The left superscript is the atomic mass number (number of protons plus neutrons), the left subscript is the atomic number (number of protons), and the right subscript is the number of neutrons.
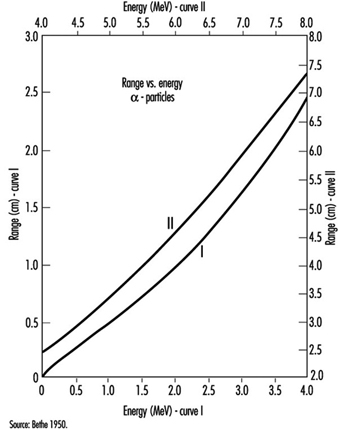
Common alpha emitters emit alpha particles with kinetic energies between about 4 and 5.5 MeV. Such alpha particles have a range in air of no more than about 5 cm (see figure 1). Alpha particles with an energy of at least 7.5 MeV are required to penetrate the epidermis (the protective layer of skin, 0.07 mm thick). Alpha emitters generally do not pose an external radiation hazard. They are hazardous only if taken within the body. Because they deposit their energy in a short distance, alpha particles are high linear energy transfer (LET) radiation and have a large radiation weighting factor; typically, w R=20.
Figure 1. Range-energy radiation of slow alpha particles in air at 15 and 760 m
Beta particles
A beta particle is a highly energetic electron or positron. (A positron is the anti-particle of the electron. It has the same mass and most other properties of an electron except for its charge, which is exactly the same magnitude as that of an electron but is positive.) Beta-emitting radionuclides can be of high or low atomic weight.
Radionuclides that have an excess of protons in comparison with stable nuclides of about the same atomic mass number can decay when a proton in the nucleus converts to a neutron. When this occurs, the nucleus emits a positron and an extremely light, very non-interacting particle called a neutrino. (The neutrino and its anti-particle are of no interest in radiation protection.) When it has given up most of its kinetic energy, the positron ultimately collides with an electron and both are annihilated. The annihilation radiation produced is almost always two 0.511 keV (kiloelectron volt) photons travelling in directions 180 degrees apart. A typical positron decay is represented by:
![]()
where the positron is represented by β+ and the neutrino by n. Note that the resulting nuclide has the same atomic mass number as the parent nuclide and an atomic (proton) number larger by one and a neutron number lesser by one than those of the original nuclide.
Electron capture competes with positron decay. In electron capture decay, the nucleus absorbs an orbital electron and emits a neutrino. A typical electron capture decay is given by:
![]()
Electron capture is always possible when the resulting nucleus has a lower total energy than the initial nucleus. However, positron decay requires that the total energy of the initial atom is greater than that of the resulting atom by more than 1.02 MeV (twice the rest mass energy of the positron).
Similar to positron and electron capture decay, negatron (β–) decay occurs for nuclei that have an excess of neutrons compared to stable nuclei of about the same atomic mass number. In this case, the nucleus emits a negatron (energetic electron) and an anti-neutrino. A typical negatron decay is represented by:
![]()
where the negatron is represented by β– and the anti-neutrino by`n Here the resulting nucleus gains one neutron at the expense of one proton but again does not change its atomic mass number.
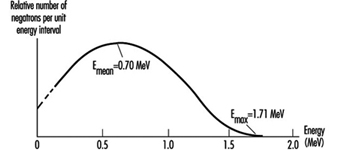
Alpha decay is a two-body reaction, so alpha particles are emitted with discrete kinetic energies. However, beta decay is a three-body reaction, so beta particles are emitted over a spectrum of energies. The maximum energy in the spectrum depends on the decaying radionuclide. The average beta energy in the spectrum is approximately one-third of the maximum energy (see figure 2).
Figure 2. Energy spectrum of negatrons emitted from 32P
Typical maximum beta energies range from 18.6 keV for tritium (3H) to 1.71 MeV for phosphorus-32 (32P).
The range of beta particles in air is approximately 3.65 m per MeV of kinetic energy. Beta particles of at least 70 keV energy are required to penetrate the epidermis. Beta particles are low-LET radiation.
Gamma radiation
Gamma radiation is electromagnetic radiation emitted by a nucleus when it undergoes a transition from a higher to a lower energy state. The number of protons and neutrons in the nucleus does not change in such a transition. The nucleus may have been left in the higher energy state following an earlier alpha or beta decay. That is, gamma rays are often emitted immediately following alpha or beta decays. Gamma rays can also result from neutron capture and inelastic scattering of subatomic particles by nuclei. The most energetic gamma rays have been observed in cosmic rays.
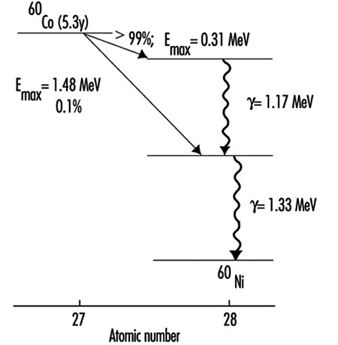
Figure 3 is a picture of the decay scheme for cobalt-60 (60Co). It shows a cascade of two gamma rays emitted in nickel-60 (60Ni) with energies of 1.17 MeV and 1.33 MeV following the beta decay of 60Co.
Figure 3. Radioactive decay scheme for 60Co
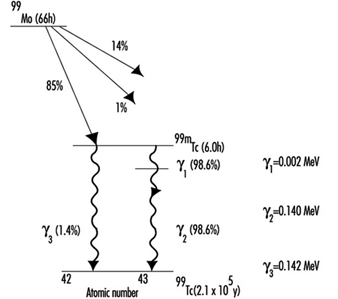
Figure 4 is a picture of the decay scheme for molybdenum-99 (99Mo). Note that the resulting technetium-99 (99Tc) nucleus has an excited state that lasts for an exceptionally long time (t½ = 6 h). Such an excited nucleus is called an isomer. Most excited nuclear states have half-lives between a few picoseconds (ps) and 1 microsecond (μs).
Figure 4. Radioactive decay scheme for 99Mo
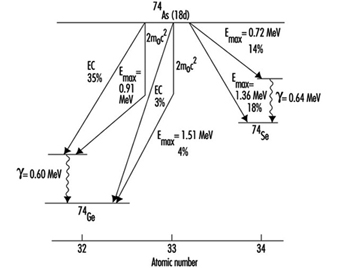
Figure 5 is a picture of the decay scheme for arsenic-74 (74As). It illustrates that some radionuclides decay in more than one way.
Figure 5. Radioactive decay scheme for 74As, illustrating competing processes of negatron emission, positron emission and electron capture (m0 is the rest mass of the electron)
While alpha and beta particles have definite ranges in matter, gamma rays are attenuated exponentially (ignoring build-up that results from scattering within a material) as they pass through matter. When build-up can be ignored the attenuation of gamma rays is given by:
![]()
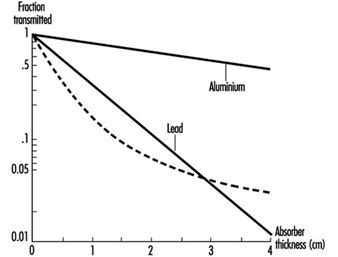
where I(x) is the gamma ray intensity as a function of distance x into the material and μ is the mass attenuation coefficient. The mass attenuation coefficient depends on gamma-ray energy and on the material with which the gamma rays are interacting. Mass attenuation coefficient values are tabulated in many references. Figure 6 shows the absorption of gamma rays in matter in conditions of good geometry (build-up can be ignored).
Figure 6. Atenuation of 667 keV gamma rays in Al and Pb under conditions of good geometry (dashed line represents attenuation of a poly-energetic photon beam)
Build-up occurs when a broad gamma-ray beam interacts with matter. The measured intensity at points within the material is increased relative to the expected “good geometry” (narrow beam) value due to gamma rays scattered from the sides of the direct beam into the measuring device. The degree of build-up depends on the geometry of the beam, on the material and on the energy of the gamma rays.
Internal conversion competes with gamma emission when a nucleus transforms from a higher energy state to a lower one. In internal conversion, an inner orbital electron is ejected from the atom instead of the nucleus emitting a gamma ray. The ejected electron is directly ionizing. As outer orbital electrons drop to lower electronic energy levels to fill the vacancy left by the ejected electron, the atom emits x rays. Internal conversion probability relative to gamma emission probability increases with increasing atomic number.
X rays
X rays are electromagnetic radiation and, as such, are identical to gamma rays. The distinction between x rays and gamma rays is their origin. Whereas gamma rays originate in the atomic nucleus, x rays result from electron interactions. Although x rays often have lower energies than gamma rays, this is not a criterion for differentiating them. It is possible to produce x rays with energies much higher than gamma rays resulting from radioactive decay.
Internal conversion, discussed above, is one method of x ray production. In this case, the resulting x rays have discrete energies equal to the difference in the energy levels between which the orbital electrons transit.
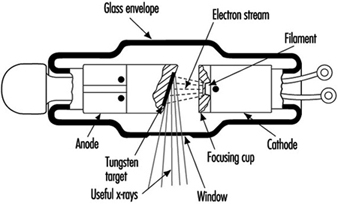
Charged particles emit electromagnetic radiation whenever they are accelerated or decelerated. The amount of radiation emitted is inversely proportional to the fourth power of the particle’s mass. As a result, electrons emit much more x radiation than heavier particles such as protons, all other conditions being equal. X-ray systems produce x rays by accelerating electrons across a large electric potential difference of many kV or MV. The electrons are then quickly decelerated in a dense, heat-resistant material, such as tungsten (W).
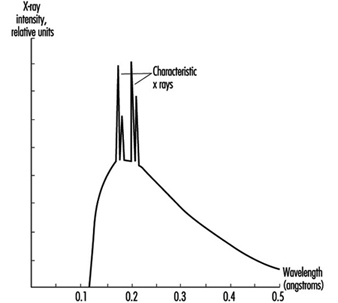
The x rays emitted from such systems have energies spread over a spectrum ranging from about zero up to the maximum kinetic energy possessed by the electrons before deceleration. Often superimposed on this continuous spectrum are x rays of discrete energy. They are produced when the decelerating electrons ionize the target material. As other orbital electrons move to fill vacancies left after ionization, they emit x rays of discrete energies similar to the way x rays are emitted following internal conversion. They are called characteristic x rays because they are characteristic of the target (anode) material. See figure 7 for a typical x ray spectrum. Figure 8 depicts a typical x ray tube.
Figure 7. X-ray spectrum illustrating the contribution of characteristic x rays produced as electrons fill holes in the K shell of W (the wavelength of x rays is inversely proportional to their energy)
X rays interact with matter the same way gamma rays do, but a simple exponential attenuation equation does not adequately describe the attenuation of x rays with a continuous range of energies (see figure 6). However, as lower energy x rays are removed more rapidly from the beam than higher energy x rays as they pass through material, the description of attenuation approaches an exponential function.
Figure 8. A simplified x-ray tube with a stationary anode and a heated filament
Neutrons
Generally, neutrons are not emitted as a direct result of natural radioactive decay. They are produced during nuclear reactions. Nuclear reactors produce neutrons in the greatest abundance but particle accelerators and special neutron sources, called (α, n) sources, also can yield neutrons.
Nuclear reactors produce neutrons when uranium (U) nuclei in nuclear fuel split, or fission. Indeed, the production of neutrons is essential in maintaining nuclear fission in a reactor.
Particle accelerators produce neutrons by accelerating charged particles, such as protons or electrons, to high energies to bombard stable nuclei in a target. Neutrons are only one of the particles that can result from such nuclear reactions. For example, the following reaction produces neutrons in a cyclotron that is accelerating deuterium ions to bombard a beryllium target:
![]()
Alpha emitters mixed with beryllium are portable sources of neutrons. These (α, n) sources produce neutrons via the reaction:
![]()
The source of the alpha particles can be such isotopes as polonium-210 (210Po),
plutonium-239 (239Pu) and americium-241 (241Am).
Neutrons are generally classified according to their energy as illustrated in table 1. This classification is somewhat arbitrary and may vary in different contexts.
Table 1. Classification of neutrons according to kinetic energy
|
Type |
Energy range |
|
Slow or thermal |
0-0.1 keV |
|
Intermediate |
0.1-20 keV |
|
Fast |
20 keV-10 MeV |
|
High-energy |
>10 MeV |
A number of possible modes of neutron interaction with matter exist, but the two main modes for the purposes of radiation safety are elastic scattering and neutron capture.
Elastic scattering is the means by which higher-energy neutrons are reduced to thermal energies. Higher-energy neutrons interact primarily by elastic scattering and generally do not cause fission or produce radioactive material by neutron capture. It is thermal neutrons that are primarily responsible for the latter types of interaction.
Elastic scattering occurs when a neutron interacts with a nucleus and bounces off with reduced energy. The interacting nucleus takes up the kinetic energy the neutron loses. After being excited in this manner, the nucleus soon gives up this energy as gamma radiation.
When the neutron eventually reaches thermal energies (so-called because the neutron is in thermal equilibrium with its environment), it is easily captured by most nuclei. Neutrons, having no charge, are not repelled by the positively charged nucleus as are protons. When a thermal neutron approaches a nucleus and comes within the range of the strong nuclear force, on the order of a few fm (fm = 10–15 metres), the nucleus captures the neutron. The result can then be a radioactive nucleus that emits a photon or other particle or, in the case of fissionable nuclei such as 235U and 239Pu, the capturing nucleus can fission into two smaller nuclei and more neutrons.
The laws of kinematics indicate that neutrons will reach thermal energies more rapidly if the elastic scattering medium includes a large number of light nuclei. A neutron rebounding off a light nucleus loses a much larger percentage of its kinetic energy than when it bounces off of a heavy nucleus. For this reason, water and hydrogenous materials are the best shielding material to slow down neutrons.
A monoenergetic beam of neutrons will attenuate exponentially in material, obeying an equation similar to that given above for photons. The probability of a neutron interacting with a given nucleus is described in terms of the quantity cross section. Cross section has units of area. The special unit for cross section is the barn (b), defined by:
![]()
It is extremely difficult to produce neutrons without accompanying gamma and x rays. It may be generally assumed that if neutrons are present, so are high energy photons.
Ionizing Radiation Sources
Primordial radionuclides
Primordial radionuclides occur in nature because their half-lives are comparable with the age of the earth. Table 2 lists the most important primordial radionuclides.
Table 2. Primordial radionuclides
|
Radioisotope |
Half-life (109 Y) |
Abundance (%) |
|
238U |
4.47 |
99.3 |
|
232Th |
14.0 |
100 |
|
235U |
0.704 |
0.720 |
|
40K |
1.25 |
0.0117 |
|
87Rb |
48.9 |
27.9 |
Uranium and thorium isotopes head a long chain of progeny radioisotopes that, as a result, are also naturally occurring. Figure 9, A-C, illustrates the decay chains for 232Th, 238U and 235U, respectively. Because alpha decay is common above atomic mass number 205 and an alpha particle’s atomic mass number is 4, there are four distinct decay chains for heavy nuclei. One of these chains (see figure 9, D), that for 237Np, does not occur in nature. This is because it does not contain a primordial radionuclide (that is, no radionuclide in this chain has a half-life comparable with the age of the earth).
Figure 9. Decay series (Z = atomic number; N = atomic mass number)
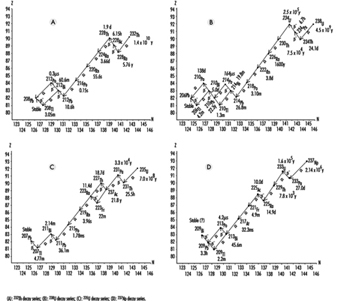 Note that radon (Rn) isotopes occur in each chain (219Rn, 220Rn and 222Rn). Since Rn is a gas, once Rn is produced, it has a chance of escape to the atmosphere from the matrix in which it was formed. However, the half-life of 219Rn is much too short to allow significant amounts of it to reach a breathing zone. The relatively short half-life of 220Rn usually makes it a lesser health hazard concern than 222Rn.
Note that radon (Rn) isotopes occur in each chain (219Rn, 220Rn and 222Rn). Since Rn is a gas, once Rn is produced, it has a chance of escape to the atmosphere from the matrix in which it was formed. However, the half-life of 219Rn is much too short to allow significant amounts of it to reach a breathing zone. The relatively short half-life of 220Rn usually makes it a lesser health hazard concern than 222Rn.
Not including Rn, primordial radionuclides external to the body deliver on the average about 0.3 mSv annual effective dose to the human population. The actual annual effective dose varies widely and is determined primarily by the concentration of uranium and thorium in the local soil. In some parts of the world where monazite sands are common, the annual effective dose to a member of the population is as high as about 20 mSv. In other places such as on coral atolls and near seashores, the value may be as low as 0.03 mSv (see figure 9).
Radon is usually considered separately from other naturally occurring terrestrial radionuclides. It seeps into the air from the soil. Once in the air, Rn further decays to radioactive isotopes of Po, bismuth (Bi) and Pb. These progeny radionuclides attach themselves to dust particles that may be breathed in and trapped in the lungs. Being alpha emitters, they deliver almost all of their radiation energy to the lungs. It is estimated that the average annual lung equivalent dose from such exposure is about 20 mSv. This lung equivalent dose is comparable to a whole body effective dose of about 2 mSv. Clearly, Rn and its progeny radionuclides are the most significant contributors to background radiation effective dose (see figure 9).
Cosmic rays
Cosmic radiation includes energetic particles of extraterrestrial origin that strike the atmosphere of the earth (primarily particles and mostly protons). It also includes secondary particles; mostly photons, neutrons and muons, generated by interactions of primary particles with gases in the atmosphere.
By virtue of these interactions, the atmosphere serves as a shield against cosmic radiation, and the thinner this shield, the greater the effective dose rate. Thus, the cosmic-ray effective dose rate increases with altitude. For example, the dose rate at an altitude of 1,800 metres is about double that at sea level.
Because primary cosmic radiation consists mostly of charged particles, it is influenced by the earth’s magnetic field. People living in higher latitudes receive greater effective doses of cosmic radiation than those closer to the earth’s equator. Variation due to this effect is of the order
of 10%.
Finally, the cosmic-ray effective dose rate varies according to modulation of the sun’s cosmic-ray output. On the average, cosmic rays contribute about 0.3 mSv to background radiation whole-body effective dose.
Cosmogenic radionuclides
Cosmic rays produce cosmogenic radionuclides in the atmosphere. The most prominent of these are tritium (3H), beryllium-7 (7Be), carbon-14 (14C) and sodium-22 (22Na). They are produced by cosmic rays interacting with atmospheric gases. Cosmogenic radionuclides deliver about 0.01 mSv annual effective dose. Most of this comes from 14C.
Nuclear fallout
From the 1940s through the 1960s, extensive testing of nuclear weapons above ground occurred. This testing produced large quantities of radioactive materials and distributed them to the environment throughout the world as fallout. Although much of this debris has since decayed to stable isotopes, small amounts that remain will be a source of exposure for many years to come. In addition, nations that continue to occasionally test nuclear weapons in the atmosphere add to the worldwide inventory.
The primary fallout contributors to effective dose currently are strontium-90 (90Sr) and caesium-137 (137Cs), both of which have half-lives around 30 years. The average annual effective dose from fallout is about 0.05 mSv.
Radioactive material in the body
The deposition of naturally occurring radionuclides in the human body results primarily from the inhalation and ingestion of these materials in air, food and water. Such nuclides include radioisotopes of Pb, Po, Bi, Ra, K (potassium), C, H, U and Th. Of these, 40K is the largest contributor. Naturally occurring radionuclides deposited in the body contribute about 0.3 mSv to the annual effective dose.
Machine-produced radiation
The use of x rays in the healing arts is the largest source of exposure to machine-produced radiation. Millions of medical x ray systems are in use around the world. The average exposure to these medical x ray systems is greatly dependent on a population’s access to care. In developed countries, the average annual effective dose from medically prescribed radiation from x rays and radioactive material for diagnosis and therapy is on the order of 1 mSv.
X rays are a by-product of most high-energy physics particle accelerators, especially those that accelerate electrons and positrons. However, appropriate shielding and safety precautions plus the limited population at risk make this source of radiation exposure less significant than the above sources.
Machine-produced radionuclides
Particle accelerators can produce a large variety of radionuclides in varying quantities by way of nuclear reactions. Accelerated particles include protons, deuterons (2H nuclei), alpha particles, charged mesons, heavy ions and so on. Target materials can be made of almost any isotope.
Particle accelerators are virtually the only source for positron-emitting radioisotopes. (Nuclear reactors tend to produce neutron-rich radioisotopes that decay by negatron emission.) They are also being increasingly used to produce short-lived isotopes for medical use, especially for positron-emission tomography (PET).
Technologically enhanced material and consumer products
X rays and radioactive materials appear, wanted and unwanted, in a great number of modern-day operations. Table 3 lists these radiation sources.
Table 3. Sources and estimates of associated population effective doses from technologically enhanced material and consumer products
|
Group I - Involves large numbers of people and the individual effective dose is very |
|
|
Tobacco products |
Combustible fuels |
|
Domestic water supplies |
Glass and ceramics |
|
Building materials |
Ophthalmic glass |
|
Mining and agricultural products |
|
|
Group II - Involves many people but the effective dose is relatively small or is limited |
|
|
Television receivers |
Highway and road construction materials |
|
Radioluminous products |
Aircraft transport of radioactive materials |
|
Airport inspection systems |
Spark gap irradiators and electron tubes |
|
Gas and aerosol (smoke) detectors |
Thorium products - fluorescent lamp starters |
|
Group III - Involves relatively few people and the collective effective dose is small |
|
|
Thorium products - tungsten welding rods |
|
Source: NCRP 1987.
Workplace Design for Radiation Safety
Basic Design Features of Radiation Facilities
Hazards associated with the handling and use of radiation sources necessitate special features of design and construction that are not required for conventional laboratories or working areas. These special design features are incorporated so that the facility worker is not unduly hampered while ensuring that he or she is not exposed to undue external or internal radiation hazards.
Access to all areas where exposure to radiation sources or radioactive materials could occur must be controlled not only with respect to the facility workers who may be permitted to enter such work areas, but also with respect to the type of clothing or protective equipment that they should wear and the precautions that they should take in controlled areas. In the administration of such control measures, it helps to classify radiation work areas based on the presence of ionizing radiation, on the presence of radioactive contamination or both. The introduction of such work area classification concepts in early planning stages will result in the facility having all the features necessary to make operations with radiation sources less hazardous.
Classification of working areas and laboratory types
The basis for the classification of the work area is the grouping of radionuclides according to their relative radiotoxicities per unit activity. Group I should be classified as very high toxicity radionuclides, Group II as moderate-to-high toxicity radionuclides, Group III as moderate toxicity radionuclides, and Group IV as low toxicity radionuclides. Table 1 shows the toxicity group classification of many radionuclides.
Table 1. Radionuclides classified according to relative radiotoxicity per unit activity
|
Group I: Very high toxicity |
|||||||||
|
210Pb |
210Po |
223Ra |
226Ra |
228Ra |
227Ac |
227Th |
228Th |
230Th |
231Pa |
|
230U |
232U |
233U |
234U |
237Np |
238Pu |
239Pu |
240Pu |
241Pu |
242Pu |
|
241Am |
243Am |
242Cm |
243Cm |
244Cm |
245Cm |
246Cm |
249Cm |
250Cf |
252Cf |
|
Group II: High toxicity |
|||||||||
|
22Na |
36Cl |
45Ca |
46Sc |
54Mn |
56Co |
60Co |
89Sr |
90Sr |
91Y |
|
95Zr |
106Ru |
110Agm |
115Cdm |
114Inm |
124Sb |
125Sb |
127Tem |
129Tem |
124I |
|
126I |
131I |
133I |
134Cs |
137Cs |
140Ba |
144Ce |
152Eu (13 y) |
154Eu |
160Tb |
|
170Tm |
181Hf |
210Bi |
182Ta |
192Ir |
204Tl |
207Bi |
230Pa |
211At |
212Pb |
|
224Ra |
228Ac |
234Th |
236U |
249Bk |
|||||
|
Group III: Moderate toxicity |
|||||||||
|
7Be |
14C |
18F |
24Na |
38Cl |
31Si |
32P |
35S |
41A |
42K |
|
43K |
47Sc |
48Sc |
48V |
51Cr |
52Mn |
56Mn |
52Fe |
55Fe |
59Fe |
|
57Co |
53Ni |
65Ni |
64Cu |
65Zn |
69Znm |
72Ga |
73As |
74As |
76As |
|
77As |
82Br |
85Krm |
87Kr |
86Rb |
85Sr |
91Sr |
90Y |
92Y |
93Y |
|
97Zr |
95Nb |
99Mo |
96Tc |
97Tcm |
97Tc |
99Tc |
97Ru |
103Ru |
105Ru |
|
105Rh |
109Pd |
105Ag |
111Ag |
109Cd |
115Cd |
115Inm |
113Sn |
125Sn |
122Sb |
|
125Tem |
129Te |
131Tem |
132Te |
130I |
132I |
134I |
135I |
135Xe |
131Cs |
|
136Cs |
140La |
141Ce |
143Ce |
142Pr |
143Pr |
147Nd |
149Nd |
147Pm |
149Pm |
|
151Sm |
152Eu (9.2 h) |
155Eu |
153Gd |
159Gd |
165Dy |
166Dy |
166Ho |
169Er |
171Er |
|
171Tm |
177Lu |
181W |
185W |
187W |
183Re |
186Re |
188Re |
185Os |
191Os |
|
193Os |
190Ir |
195Ir |
191Pt |
193Pt |
197Pt |
196Au |
198Au |
199Au |
197Hg |
|
197Hgm |
203Hg |
200Tl |
201Tl |
202Tl |
203Pb |
206Bi |
212Bi |
220Rn |
222Rn |
|
231Th |
233Pa |
239Np |
|||||||
|
Group IV: Low toxicity |
|||||||||
|
3H |
15O |
37A |
58Com |
59Ni |
69Zn |
71Ge |
85Kr |
85Srm |
87Rb |
|
91Ym |
93Zr |
97Nb |
96Tcm |
99Tcm |
103Rhm |
133Inm |
129I |
131Xem |
133Xe |
|
134Csm |
135Cs |
147Sm |
187Re |
191Osm |
193Ptm |
197Ptm |
natTh |
232Th |
235U |
|
238U |
natU |
||||||||
(IAEA 1973)
Three broad types of laboratories can be envisaged on the basis of radiotoxicity considerations, the amounts or quantities of radioactive materials that will be handled in the work area and the type of operations involved.
Table 2 describes laboratories by type and provides examples for each type. Table 3 shows the types of laboratories along with the work area classification and access control (IAEA 1973).
Table 2. Classification of working areas
|
Type |
Definition |
Access control |
Typical operations |
|
1 |
Areas in which the external radiation absorbed dose levels or radioactive contamination levels could be high |
Access controlled to radiation workers only, under strictly controlled working conditions and with appropriate protective equipment |
Hot laboratories, highly contaminated areas |
|
2 |
Areas in which external radiation levels could exist and in which the possibility of contamination necessitates operating instructions |
Access limited to radiation workers with |
Luminizing factories and other equivalent |
|
3 |
Areas in which the average external radiation level is less than 1 mGy·wk-1 and in which the possibility of radioactive contamination necessitates special operating instructions |
Access limited to radiation workers, no |
Working areas in the immediate vicinity of |
|
4 |
Areas within the confines of a radiation facility where the external radiation levels are less than 0.1 mGy•wk-1 and where |
Access uncontrolled |
Administration and patient waiting areas |
(ICRP 1977, IAEA 1973)
Table 3. Classification of laboratories for handling radioactive materials
|
Group of |
Type of laboratory required for the activity specified below |
||
|
Type 1 |
Type 2 |
Type 3 |
|
|
I |
<370 kBq |
70 kBq to |
>37 MBq |
|
II |
<37 MBq |
37 MBq to |
>37 GBq |
|
III |
<37 GBq |
37 GBq to |
>370 GBq |
|
IV |
<370 GBq |
370 GBq to |
>37 Tbq |
|
Operational factors for laboratory use of radioactive material |
Multiplication factors for the activity levels |
|
Simple storage |
×100 |
|
Simple wet operations (for example, preparation of aliquots of stock solution) |
×10 |
|
Normal chemical operations (for example, simple chemical preparation and analysis) |
×1 |
|
Complex wet operations (for example, multiple operations or operations with complex glass ware) |
×0.1 |
|
Simple dry operations (for example, manipulations of powders of volatile radioactive compounds) |
×0.1 |
|
Dry and dusty operations (for example, grinding) |
×0.01 |
(ICRP 1977, IAEA 1973)
The hazards involved in working with radioactive material depend not only on the level of radiotoxicity or chemical toxicity and the activity of the radionuclides, but also on the radioactive material’s physical and chemical form and on the nature and complexity of the operation or procedure being performed.
Location of a radiation facility in a building
When a radiation facility is part of a large building, the following should be kept in mind when deciding on the location of such a facility:
- The radiation facility should be located in a relatively unfrequented part of the building, so that access to the area can be easily controlled.
- The potential for fires should be minimal in the area chosen.
- The location of the radiation facility and the heating and ventilation provided should be such that possibilities for the spread of both surface and airborne radioactive contamination are minimal.
- The location of the radiation facility should be chosen judiciously, so that with a minimum expenditure for shielding, radiation levels can be effectively maintained within established limits in the immediate vicinity.
Planning of radiation facilities
Where a gradation of levels of activity is envisioned, the laboratory should be located so that access to areas where high radiation or radioactive contamination levels exist is gradual; that is, one first enters a non-radiation area, then a low activity area, then a medium activity area and so on.
The need for elaborate control of ventilation in small laboratories can be avoided by the use of hoods or glove boxes for handling unsealed sources of radioactive material. However, the ventilation system should be designed to permit air flow in a direction such that any radioactive material that becomes airborne will flow away from the radiation worker. The air flow should always be from an uncontaminated area toward a contaminated or potentially contaminated area.
For the handling of unsealed sources of low to medium radioactivity, the average air speed through the opening in the hood must be about 0.5 ms–1. For highly radiotoxic or high-level radioactivity, the air velocity through the opening should be raised to an average of 0.6 to
1.0 ms–1. However, excessively high air speeds can draw out radioactive materials from open containers and contaminate the entire hood area.
The placement of the hood in the laboratory is important with respect to cross-drafts. In general, a hood should be located well away from doorways where supply or make-up air must enter. Dual-speed fans will permit operation at a higher air velocity while the hood is in use and a lower velocity when it is closed.
The aim of any ventilating system should be to:
- provide comfortable working conditions
- provide continuous air changes (three to five changes per hour) for the purposes of removing and diluting undesirable air contaminants
- minimize the contamination of other areas of the building and the environment.
In the design of radiation facilities, heavy shielding requirements can be minimized by the adoption of certain simple measures. For example, for radiation therapy, accelerators, neutron generators or panoramic radiation sources, a maze can reduce the need for a heavy lead-lined door. Tapering of the primary protective barrier in areas that are not directly in the useful beam or locating the facility partially or completely underground can significantly reduce the amount of required shielding.
Careful attention must be paid to the proper positioning of viewing windows, underground conduit cables and ventilation system baffles. The viewing window should intercept scattered radiation only. Even better is a closed circuit television, which can also improve efficiency.
Surface finishes within a work area
All raw surfaces, such as plaster, concrete, wood and so on, should be permanently sealed with a suitable material. The choice of material should be made with the following considerations in mind:
- the provision of a smooth, chemically inert surface
- the environmental conditions of temperature, humidity and mechanical wear and tear to which the surfaces may be exposed
- compatibility with radiation fields to which the surface is exposed
- the need for ease of repair in the event of damage.
Ordinary paints, varnishes and lacquers are not recommended for covering wear surfaces. The application of a surfacing material that can be easily removed may be helpful if contamination occurs and decontamination is required. However, the removal of such materials sometimes can be difficult and messy.
Plumbing
Sinks, wash basins and floor drains should be properly marked. Wash basins where contaminated hands may be washed should have knee- or foot-operated faucets. It may be economical to reduce maintenance by using piping which can be easily decontaminated or replaced if required. In some cases it may be advisable to install underground holding or storage tanks to control the disposal of liquid radioactive materials.
Radiation Shielding Design
Shielding is important for reducing radiation exposure of facility workers and members of the general public. Shielding requirements depend on a number of factors, including the time that radiation workers or members of the public are exposed to the radiation sources and the type and energy of the radiation sources and radiation fields.
In the design of radiation shields, the shielding material should be placed near the radiation source if possible. Separate shielding considerations must be made for each type of radiation concerned.
Shielding design can be a complex task. For example, the use of computers to model shielding for accelerators, reactors and other high-level radiation sources is beyond the scope of this article. Qualified experts always should be consulted for complex shielding design.
Gamma source shielding
The attenuation of gamma radiation is qualitatively different from that of either alpha or beta radiation. Both of those types of radiation have a definite range in matter and are completely absorbed. Gamma radiation, on the other hand, can be reduced in intensity by increasingly thicker absorbers but it cannot be completely absorbed. If the attenuation of monoenergetic gamma rays is measured under conditions of good geometry (that is, the radiation is well collimated in a narrow beam) the intensity data, when plotted on a semi-log graph versus absorber thickness, will lie on a straight line with the slope equal to the attenuation
coefficient, μ.
The intensity or absorbed dose rate transmitted through an absorber can be calculated as follows:
I(t) = I(0)e– μ t
where I(t) is the gamma-ray intensity or absorbed dose rate transmitted through an absorber of thickness t.
The units of μ and t are the reciprocal of each other. If the absorber thickness t is measured in cm, then μ is the linear attenuation coefficient and has units of cm–1. If t has units of areal density (g/cm2), then μ is the mass attenuation coefficient μm and has units of cm2/g.
As a first-order approximation using areal density, all materials have about the same photon attenuation properties for photons with energies between about 0.75 and 5.0 MeV (mega-electron volts). Within this energy range, gamma shielding properties are approximately proportional to the density of the shielding material. For lower or higher photon energies, absorbers of higher atomic number provide more effective shielding than those of lower atomic number, for a given areal density.
Under conditions of poor geometry (for example, for a broad beam or for a thick shield), the above equation will significantly underestimate the required shield thickness because it assumes that every photon that interacts with the shield will be removed from the beam and not be detected. A significant number of photons may be scattered by the shield into the detector, or photons that had been scattered out of the beam may be scattered back into it after a second interaction.
A shield thickness for conditions of poor geometry may be estimated through the use of the build-up factor B that may be estimated as follows:
I(t) = I(0)Be– μ t
The build-up factor is always greater than one, and may be defined as the ratio of the intensity of the photon radiation, including both the primary and scattered radiation, at any point in the beam, to the intensity of the primary beam only at that point. The build-up factor may apply either to radiation flux or to absorbed dose rate.
Build-up factors have been calculated for various photon energies and various absorbers. Many of the graphs or tables give the shield thickness in terms of relaxation lengths. A relaxation length is the thickness of a shield that will attenuate a narrow beam to 1/e (about 37%) of its original intensity. One relaxation length, therefore, is numerically equal to the reciprocal of the linear attenuation coefficient (that is, 1/μ).
The thickness of an absorber that, when introduced into the primary photon beam, reduces the absorbed dose rate by one-half is called the half-value layer (HVL) or half-value thickness (HVT). The HVL may be calculated as follows:
HVL = ln2 / μ
The required photon shield thickness can be estimated by assuming narrow-beam or good geometry while calculating the required shielding, and then increasing the value thus found by one HVL to account for build-up.
The thickness of an absorber that, when introduced into the primary photon beam, reduces the absorbed dose rate by one-tenth is the tenth-value layer (TVL). One TVL is equal to about 3.32 HVLs, since:
ln10 / ln2 ≈ 3.32
Values for both TVLs and HVLs have been tabulated for various photon energies and several common shielding materials (e.g., lead, steel and concrete) (Schaeffer 1973).
The intensity or absorbed dose rate for a point source obeys the inverse square law and may be calculated as follows:

where Ii is the photon intensity or absorbed dose rate at distance di from the source.
Medical and non-medical x-ray equipment shielding
Shielding for x-ray equipment is considered under the two categories, source shielding and structural shielding. Source shielding is usually provided by the manufacturer of the x-ray tube housing.
Safety regulations specify one type of protective tube housing for medical diagnostic x-ray facilities and another type for medical therapeutic x-ray facilities. For non-medical x-ray equipment, the tube housing and other parts of the x-ray apparatus, such as the transformer, are shielded to reduce the leakage x-ray radiation to acceptable levels.
All x-ray machines, both medical and non-medical, have protective tube housings designed to limit the amount of leakage radiation. Leakage radiation, as used in these specifications for tube housings, means all radiation coming from the tube housing except for the useful beam.
Structural shielding for an x-ray facility provides protection from the useful or primary x-ray beam, from leakage radiation and from scatter radiation. It encloses both the x-ray equipment and the object being irradiated.
The amount of scatter radiation depends on the x-ray field size, energy of the useful beam, the effective atomic number of the scattering media and the angle between the incoming useful beam and the direction of scatter.
A key design parameter is the facility workload (W):
![]()
where W is the weekly workload, usually given in mA-min per week; E is the tube current multiplied by the exposure time per view, usually given in mA s; Nv is the number of views per patient or object irradiated; Np is the number of patients or objects per week and k is a conversion factor (1 min divided by 60 s).
Another key design parameter is the use factor Un for a wall (or floor or ceiling) n. The wall may be protecting any occupied area such as a control room, office or waiting room. The use factor is given by:
![]()
where, Nv,n is the number of views for which the primary x ray beam is directed toward wall n.
The structural shielding requirements for a given x ray facility are determined by the following:
- the maximum tube potential, in kilovolts-peak (kVp), at which the x ray tube is operated
- the maximum beam current, in mA, at which the x ray system is operated
- the workload (W), which is a measure, in suitable units (usually mA-min per week), of the amount of use of the x ray system
- the use factor (U), which is the fraction of the workload during which the useful beam is pointed in the direction of interest
- the occupancy factor (T), which is the factor by which the workload should be multiplied to correct for the degree or type of occupancy of the area to be protected
- the maximum permissible dose equivalent rate (P) to a person for controlled and non-controlled areas (typical absorbed dose limits are 1 mGy for a controlled area in one week and 0.1 mGy for a non-controlled area in one week)
- type of shielding material (for example, lead or concrete)
- the distance (d) from the source to the location being protected.
With these considerations included, the value of the primary beam ratio or transmission factor K in mGy per mA-min at one metre is given by:
![]()
Shielding of the x-ray facility must be constructed so that protection is not impaired by joints; by openings for ducts, pipes and so on, that pass through the barriers; or by conduits, service boxes and so on, embedded in the barriers. The shielding should cover not only the back of the service boxes, but also the sides, or be extended sufficiently to offer equivalent protection. Conduits that pass through barriers should have sufficient bends to reduce the radiation to the required level. Observation windows must have shielding equivalent to that required for the partition (barrier) or door in which they are located.
Radiation therapy facilities may require door interlocks, warning lights, closed circuit television or means for audible (e.g., voice or buzzer) and visual communication between anyone who may be in the facility and the operator.
Protective barriers are of two types:
- primary protective barriers, which are sufficient to attenuate the primary (useful) beam to the required level
- secondary protective barriers, which are sufficient to attenuate leakage, scattered and stray radiation to the required level.
To design the secondary protective barrier, separately calculate the required thickness to protect against each component. If the required thicknesses are about the same, add an additional HVL to the greatest calculated thickness. If the greatest difference between the calculated thicknesses is one TVL or more, the thickest of the calculated values will suffice.
Scattered radiation intensity depends on scattering angle, energy of the useful beam, field size or scattering area, and subject composition.
When designing secondary protective barriers, the following simplifying conservative assumptions are made:
- When x rays are produced at 500 kV or less, the energy of the scattered radiation is equal to the energy of the useful beam.
- After being scattered, the x-ray energy spectrum for beams generated at voltages greater than 500 kV are degraded to that of a 500 kV beam, and the absorbed dose rate at 1 m and 90 degrees from the scatterer is 0.1% of that in the useful beam at the point of scattering.
The transmission relationship for scattered radiation is written in terms of the scattering transmission factor (Kμx) with units of mGy•m2 (mA-min)–1:
![]()
where P is the maximum weekly absorbed dose rate (in mGy), dscat is the distance from the x ray tube’s target and the object (patient), dsec is the distance from the scatterer (object) to the point of interest that the secondary barriers are meant to shield, a is the ratio of scattered radiation to incident radiation, f is the actual scattering field size (in cm2), and F is a factor accounting for the fact that x ray output increases with voltage. Smaller values of Kμx require thicker shields.
The leakage attenuation factor BLX for diagnostic x-ray systems is calculated as follows:
![]()
where d is the distance from the tube target to the point of interest and I is the tube current in mA.
The barrier attenuation relationship for therapeutic x-ray systems operating at 500 kV or less is given by:
![]()
For therapeutic x-ray tubes operating at potentials greater than 500 kV, the leakage is usually limited to 0.1% of the intensity of the useful beam at 1 m. The attenuation factor in this case is:
![]()
where Xn is the absorbed dose rate (in mGy/h) at 1 m from a therapeutic x-ray tube operated at a tube current of 1 mA.
The number n of HVLs required to obtain the desired attenuation BLX is obtained from the relationship:
![]()
or
![]()
Beta particle shielding
Two factors must be considered when designing a shield for a high-energy beta emitter. They are the beta particles themselves and the bremsstrahlung produced by beta particles absorbed in the source and in the shield. Bremsstrahlung consists of x-ray photons produced when high-speed charged particles undergo rapid deceleration.
Therefore, a beta shield often consists of a substance of low atomic number (to minimize bremsstrahlung production) that is thick enough to stop all the beta particles. This is followed by a material of high atomic number that is thick enough to attenuate bremsstrahlung to an acceptable level. (Reversing the order of the shields increases bremsstrahlung production in the first shield to a level so high that the second shield may provide inadequate protection.)
For purposes of estimating bremsstrahlung hazard, the following relationship may be used:
![]()
where f is the fraction of the incident beta energy converted into photons, Z is the atomic number of the absorber, and Eβ is the maximum energy of the beta particle spectrum in MeV. To assure adequate protection, it is normally assumed that all bremsstrahlung photons are of the maximum energy.
The bremsstrahlung flux F at a distance d from the beta source can be estimated as follows:
![]()
`Eβ is the average beta particle energy and can be estimated by:
![]()
The range Rβ of beta particles in units of areal density (mg/cm2) may be estimated as follows for beta particles with energies between 0.01 and 2.5 MeV:
![]()
where Rβ is in mg/cm2 and Eβ is in MeV.
For Eβ>2.5 MeV, the beta particle range Rβ may be estimated as follows:
![]()
where Rβ is in mg/cm2 and Eβ is in MeV.
Alpha particle shielding
Alpha particles are the least penetrating type of ionizing radiation. Because of the random nature of its interactions, the range of an individual alpha particle varies between nominal values as indicated in figure 1. Range in the case of alpha particles may be expressed in different ways: by minimum, mean, extrapolated, or maximum range. The mean range is the most accurately determinable, corresponds to the range of the “average” alpha particle, and is used most often.
Figure 1. Typical range distribution of alpha particles
Air is the most commonly used absorbing medium for specifying the range-energy relationship of alpha particles. For alpha energy Eα less than about 4 MeV, Rα in air is approximately given by:
![]()
where Rα is in cm, Eα in MeV.
For Eα between 4 and 8 MeV, Rα in air is given approximately by:
![]()
where Rα is in cm, Eα in MeV.
The range of alpha particles in any other medium may be estimated from the following relationship:
Rα (in other medium; mg/cm2) » 0.56 A1/3 Rα (in air; cm) where A is the atomic number of the medium.
Neutron shielding
As a general rule of thumb for neutron shielding, neutron energy equilibrium is achieved and then remains constant after one or two relaxation lengths of shielding material. Therefore, for shields thicker than a few relaxation lengths, the dose equivalent outside concrete or iron shielding will be attenuated with relaxation lengths of 120 g/cm2 or 145 g/cm2, respectively.
Neutron energy loss by elastic scattering requires a hydrogenous shield to maximize the energy transfer as the neutrons are moderated or slowed down. For neutron energies above 10 MeV, inelastic processes are effective in attenuating neutrons.
As with nuclear power reactors, high-energy accelerators require heavy shielding to protect workers. Most of the dose equivalents to workers come from exposure to activated radioactive material during maintenance operations. Activation products are produced in the accelerator’s components and support systems.
Monitoring of the Workplace Environment
It is necessary to deal separately with the design of routine and of operational monitoring programs for the workplace environment. Special monitoring programs will be designed to achieve specific objectives. It is not desirable to design programs in general terms.
Routine monitoring for external radiation
An important part in the preparation of a program for routine monitoring for external radiation in the workplace is to conduct a comprehensive survey when a new radiation source or a new facility is put into service, or when any substantial changes have been made or may have been made in an existing installation.
The frequency of routine monitoring is determined by consideration of the expected changes in the radiation environment. If changes to the protective equipment or alterations of the processes conducted in the workplace are minimal or non-substantial, then routine radiation monitoring of the workplace is rarely required for review purposes. If the radiation fields are subject to increase rapidly and unpredictably to potentially hazardous levels, then an area radiation monitoring and warning system is required.
Operational monitoring for external radiation
The design of an operational monitoring program depends greatly on whether the operations to be conducted influence the radiation fields or whether the radiation fields will remain substantially constant throughout normal operations. The detailed design of such a survey depends critically on the form of the operation and on the conditions under which it takes place.
Routine monitoring for surface contamination
The conventional method of routine monitoring for surface contamination is to monitor a representative fraction of the surfaces in an area at a frequency dictated by experience. If operations are such that considerable surface contamination is likely and such that workers could carry significant amounts of radioactive material out of the work area in a single event, routine monitoring should be supplemented by the use of portal contamination monitors.
Operational monitoring for surface contamination
One form of operational monitoring is the surveying of items for contamination when they leave a radiologically controlled area. This monitoring must include workers’ hands and feet.
The principle objectives of a program of monitoring for surface contamination are:
- to assist in preventing the spread of radioactive contamination
- to detect failures of containment or departures from good operating procedures
- to limit surface contamination to levels at which general standards of good housekeeping are adequate to keep radiation exposures as low as reasonably achievable and to avoid excessive exposures caused by contamination of clothing and skin
- to provide information for the planning of optimized programs for individuals, for air monitoring and for defining operational procedures.
Monitoring for airborne contamination
The monitoring of airborne radioactive materials is important because inhalation is usually the most important route of intake of such material by radiation workers.
The monitoring of the workplace for airborne contamination will be needed on a routine basis in the following circumstances:
- when gaseous or volatile materials are handled in quantity
- when the handling of any radioactive material in such operations results in frequent and substantial contamination of the workplace
- during the processing of moderately to highly toxic radioactive materials
- during the handling of unsealed therapeutic radionuclides in hospitals
- during the use of hot cells, reactors and critical assemblies.
When an air monitoring program is required, it must:
- be able to assess the probable upper limit of the inhalation of radioactive material by radiation workers
- be able to draw attention to unexpected airborne contamination so that radiation workers can be protected and remedial measures instituted
- provide information for planning of programs of individual monitoring for internal contamination.
The most common form of monitoring for airborne contamination is the use of air samplers at a number of selected locations selected to be reasonably representative of the breathing zones of radiation workers. It may be necessary to make samples more accurately represent breathing zones by using personal air or lapel samplers.
Detection and measurement of radiation and radioactive contamination
The monitoring or surveying by wipes and instrument surveys of bench tops, floors, clothing, skin, and other surfaces are at best qualitative procedures. It is difficult to make them highly quantitative. The instruments used are usually detecting types rather than measuring devices. Since the amount of radioactivity involved is often small, the sensitivity of the instruments should be high.
The requirement for portability of contamination detectors depends on their intended uses. If the instrument is for general-purpose monitoring of laboratory surfaces, a portable type of instrument is desirable. If the instrument is for a specific use in which the item to be monitored can be brought to the instrument, then portability is not necessary. Clothing monitors and hand and shoe monitors generally are not portable.
Count-rate instruments and monitors usually incorporate meter readouts and aural outputs or earphone jacks. Table 4 identifies instruments that may be used for the detection of radioactive contamination.+
Table 4. Contamination detection instruments
|
Instrument |
Counting rate range and other characteristics1 |
Typical uses |
Remarks |
|
bg surface monitors2 |
|||
|
General |
|||
|
Portable count rate meter (thin-walled or thin window G-M3 counter) |
0-1,000 cpm |
Surfaces, hands, clothing |
Simple, reliable, battery-powered |
|
Thin end-window |
0-1,000 cpm |
Surfaces, hands, clothing |
Line-operated |
|
Personnel |
|||
|
Hand-and-shoe monitor, G-M or |
Between 1½ and 2 times natural |
Rapid monitoring for contamination |
Automatic operation |
|
Special |
|||
|
Laundry monitors, floor monitors, |
Between 1½ and 2 times natural |
Monitoring for contamination |
Convenient and rapid |
|
Alpha surface monitors |
|||
|
General |
|||
|
Portable air proportional counter with probe |
0-100,000 cpm over 100 cm2 |
Surfaces, hands, clothing |
Not for use in high humidity, battery- |
|
Portable gas-flow counter with probe |
0-100,000 cpm over 100 cm2 |
Surfaces, hands, clothing |
Battery-powered, fragile window |
|
Portable scintillation counter with probe |
0-100,000 cpm over 100 cm2 |
Surfaces, hands, clothing |
Battery-powered, fragile window |
|
Personal |
|||
|
Hand-and-shoe proportional counter-type, monitor |
0-2,000 cpm over about 300 cm2 |
Rapid monitoring of hands and shoes for contamination |
Automatic operation |
|
Hand-and-shoe scintillation counter-type, monitor |
0-4,000 cpm over about 300 cm2 |
Rapid monitoring of hands and shoes for contamination |
Rugged |
|
Wound monitors |
Low-energy photon detection |
Plutonium monitoring |
Special design |
|
Air monitors |
|||
|
Particle samplers |
|||
|
Filter paper, high-volume |
1.1 m3/min |
Quick grab samples |
Intermittent use, requires separate |
|
Filter paper, low volume |
0.2-20 m3/h |
Continuous room air monitoring |
Continuous use, requires separate |
|
Lapel |
0.03 m3/min |
Continuous breathing zone air monitoring |
Continuous use, requires separate |
|
Electrostatic precipitator |
0.09 m3/min |
Continuous monitoring |
Sample deposited on cylindrical shell, |
|
Impinger |
0.6-1.1 m3/min |
Alpha contamination |
Special uses, requires separate counter |
|
Tritium air monitors |
|||
|
Flow ionization chambers |
0-370 kBq/m3 min |
Continuous monitoring |
May be sensitive to other ionization |
|
Complete air monitoring systems |
Minimum detectable activity |
|
|
|
Fixed filter paper |
α » 0.04 Bq/m3; βγ » 0.04 Bq/m3 |
Background buildup can mask low-level activity, counter included |
|
|
Moving filter paper |
α » 0.04 Bq/m3; βγ » 0.04 Bq/m3 |
Continuous record of air activity, time of measurement can be adjusted from |
|
1 cpm = counts per minute.
2 Few surface monitors are suitable for detecting tritium (3H). Wipe tests counted by liquid scintillation devices are appropriate for detecting tritium contamination.
3 G-M = Geiger-Muller countrate meter.
Alpha contamination detectors
The sensitivity of an alpha detector is determined by its window area and window thickness. Generally window area is 50 cm2 or greater with a window areal density of 1 mg/cm2 or less. Alpha contamination monitors should be insensitive to beta and gamma radiation in order to minimize background interference. This is generally accomplished by pulse height discrimination in the counting circuit.
Portable alpha monitors can be either gas proportional counters or zinc sulphide scintillation counters.
Beta contamination detectors
Portable beta monitors of several types can be used for the detection of beta-particle contamination. Geiger-Mueller (G-M) count-rate meters generally require a thin window (areal density between 1 and 40 mg/cm2). Scintillation (anthracene or plastic) counters are very sensitive to beta particles and relatively insensitive to photons. Portable beta counters generally cannot be used to monitor for tritium (3H) contamination because tritium beta-particle energy is very low.
All instruments used for beta contamination monitoring also respond to background radiation. This must be taken into account when interpreting instrument readings.
When high background radiation levels exist, portable counters for contamination monitoring are of limited value, since they do not indicate small increases in initially high counting rates. Under these conditions smears or wipe tests are recommended.
Gamma contamination detectors
Since most gamma emitters also emit beta particles, most contamination monitors will detect both beta and gamma radiation. The usual practice is to use a detector that is sensitive to both types of radiation in order to have increased sensitivity, since the detection efficiency is usually greater for beta particles than for gamma rays. Plastic scintillators or sodium iodide (NaI) crystals are more sensitive to photons than are G-M counters, and are therefore recommended for detecting gamma rays.
Air samplers and monitors
Particulates may be sampled by the following methods: sedimentation, filtration, impaction and electrostatic or thermal precipitation. However, particulate contamination in the air is generally monitored by filtration (pumping air through filter media and measuring the radioactivity on the filter). Sampling flow rates generally are greater than 0.03 m3/min. However, most laboratories’ sampling flow rates are no more than 0.3 m3/min. Specific types of air samplers include “grab” samplers and continuous air monitors (CAM). The CAMs are available with either fixed or moving filter paper. A CAM should include an alarm since its principle function is to warn of changes in airborne contamination.
Because alpha particles have very short range, surface-loading filters (for example, membrane filters) must be used for the measurement of alpha-particle contamination. The sample collected must be thin. The time between collection and measurement must be considered to allow for the decay of radon (Rn) progeny.
Radioiodines such as 123I, 125I and 131I can be detected with filter paper (particularly if the paper is loaded with charcoal or silver nitrate) because some of the iodine will deposit on the filter paper. However, quantitative measurements require activated charcoal or silver zeolite traps or canisters to provide efficient absorption.
Tritiated water and tritium gas are the primary forms of tritium contamination. Although tritiated water has some affinity for most filter papers, filter paper techniques are not very effective for tritiated water sampling. The most sensitive and accurate measurement methods involve the absorption of tritiated water vapour condensate. Tritium in the air (for example, as hydrogen, hydrocarbons or water vapour) can be measured effectively with Kanne chambers (flow-through ionization chambers). Absorption of tritiated water vapour from an air sample can be accomplished by passing the sample through a trap containing a silica-gel molecular sieve or by bubbling the sample through distilled water.
Depending on the operation or process it may be necessary to monitor for radioactive gases. This can be accomplished with Kanne chambers. The most commonly used devices for sampling by absorption are fretted gas scrubbers and impingers. Many gases may also be collected by cooling the air below the freezing point of the gas and collecting the condensate. This method of collection is most often used for tritium oxide and noble gases.
There are a number of ways to obtain grab samples. The method selected should be appropriate for the gas to be sampled and the required method of analysis or measurement.
Monitoring of effluent
Effluent monitoring refers to the measurement of radioactivity at its point of release to the environment. It is relatively easy to accomplish because of the controlled nature of the sampling location, which is usually in a waste stream that is being discharged through a stack or liquid discharge line.
Continuous monitoring of airborne radioactivity may be necessary. In addition to the sample collection device, usually a filter, a typical sampling arrangement for particulates in air includes an air-moving device, a flowmeter and associated ducting. The air-moving device is located downstream from the sample collector; that is, the air is first passed through the sample collector, then through the remainder of the sampling system. Sampling lines, particularly those ahead of the sample collector system, should be kept as short as possible and free of sharp bends, areas of turbulence, or resistance to the air flow. Constant volume over a suitable range of pressure drops should be used for air sampling. Continuous sampling for radioactive xenon (Xe) or krypton (Kr) isotopes is accomplished by adsorption on activated charcoal or by cryogenic means. The Lucas cell is one of the oldest techniques and still the most popular method for the measurement of Rn concentrations.
Continuous monitoring of liquids and waste lines for radioactive materials is sometimes necessary. Waste lines from hot laboratories, nuclear medicine laboratories and reactor coolant lines are examples. Continuous monitoring can be performed, however, by routine laboratory analysis of a small sample proportional to the effluent flow rate. Samplers that take periodic aliquots or that continuously extract a small amount of liquid are available.
Grab sampling is the usual method used to determine the concentration of radioactive material in a hold-up tank. The sample must be taken after recirculation in order to compare the result of the measurement with allowable discharge rates.
Ideally, results of effluent monitoring and environmental monitoring will be in good agreement, with the latter calculable from the former with the aid of various pathway models. However, it must be recognized and emphasized that effluent monitoring, no matter how good or extensive, cannot substitute for actual measurement of radiological conditions in the environment.
Radiation Safety
This article describes aspects of radiation safety programmes. The objective of radiation safety is to eliminate or minimize harmful effects of ionizing radiation and radioactive material on workers, the public and the environment while allowing their beneficial uses.
Most radiation safety programmes will not have to implement every one of the elements described below. The design of a radiation safety programme depends on the types of ionizing radiation sources involved and how they are used.
Radiation Safety Principles
The International Commission on Radiological Protection (ICRP) has proposed that the following principles should guide the use of ionizing radiation and the application of radiation safety standards:
- No practice involving exposures to radiation should be adopted unless it produces sufficient benefit to the exposed individuals or to society to offset the radiation detriment it causes (the justification of a practice).
- In relation to any particular source within a practice, the magnitude of individual doses, the number of people exposed, and the likelihood of incurring exposures where these are not certain to be received should all be kept as low as reasonably achievable (ALARA), economic and social factors being taken into account. This procedure should be constrained by restrictions on the doses to individuals (dose constraints), so as to limit the inequity likely to result from the inherent economic and social judgements (the optimization of protection).
- The exposure of individuals resulting from the combination of all the relevant practices should be subject to dose limits, or to some control of risk in the case of potential exposures. These are aimed at ensuring that no individual is exposed to radiation risks that are judged to be unacceptable from these practices in any normal circumstances. Not all sources are susceptible of control by action at the source and it is necessary to specify the sources to be included as relevant before selecting a dose limit (individual dose and risk limits).
Radiation Safety Standards
Standards exist for radiation exposure of workers and the general public and for annual limits on intake (ALI) of radionuclides. Standards for concentrations of radionuclides in air and in water can be derived from the ALIs.
The ICRP has published extensive tabulations of ALIs and derived air and water concentrations. A summary of its recommended dose limits is in table 1.
Table 1. Recommended dose limits of the International Commission on Radiological Protection1
|
Application |
Dose limit |
|
|
Occupational |
Public |
|
|
Effective dose |
20 mSv per year averaged over |
1 mSv in a year3 |
|
Annual equivalent dose in: |
||
|
Lens of the eye |
150 mSv |
15 mSv |
|
Skin4 |
500 mSv |
50 mSv |
|
Hands and feet |
500 mSv |
- |
1 The limits apply to the sum of the relevant doses from external exposure in the specified period and the 50-year committed dose (to age 70 years for children) from intakes in the same period.
2 With the further provision that the effective dose should not exceed 50 mSv in any single year. Additional restrictions apply to the occupational exposure of pregnant women.
3 In special circumstances, a higher value of effective dose could be allowed in a single year, provided that the average over 5 years does not exceed 1 mSv per year.
4 The limitation on the effective dose provides sufficient protection for the skin against stochastic effects. An additional limit is needed for localized exposures in order to prevent deterministic effects.
Dosimetry
Dosimetry is used to indicate dose equivalents that workers receive from external radiation fields to which they may be exposed. Dosimeters are characterized by the type of device, the type of radiation they measure and the portion of the body for which the absorbed dose is to be indicated.
Three main types of dosimeters are most commonly employed. They are thermoluminescent dosimeters, film dosimeters and ionization chambers. Other types of dosimeters (not discussed here) include fission foils, track-etch devices and plastic “bubble” dosimeters.
Thermoluminescent dosimeters are the most commonly used type of personnel dosimeter. They take advantage of the principle that when some materials absorb energy from ionizing radiation, they store it such that later it can be recovered in the form of light when the materials are heated. To a high degree, the amount of light released is directly proportional to the energy absorbed from the ionizing radiation and hence to the absorbed dose the material received. This proportionality is valid over a very wide range of ionizing radiation energy and absorbed dose rates.
Special equipment is necessary to process thermoluminescent dosimeters accurately. Reading the thermoluminescent dosimeter destroys the dose information contained in it. However, after appropriate processing, thermoluminescent dosimeters are reusable.
The material used for thermoluminescent dosimeters must be transparent to the light it emits. The most common materials used for thermoluminescent dosimeters are lithium fluoride (LiF) and calcium fluoride (CaF2). The materials may be doped with other materials or made with a specific isotopic composition for specialized purposes such as neutron dosimetry.
Many dosimeters contain several thermoluminescent chips with different filters in front of them to allow discrimination between energies and types of radiation.
Film was the most popular material for personnel dosimetry before thermoluminescent dosimetry became common. The degree of film darkening depends on the energy absorbed from the ionizing radiation, but the relationship is not linear. Dependence of film response on total absorbed dose, absorbed dose rate and radiation energy is greater than that for thermoluminescent dosimeters and can limit film’s range of applicability. However, film has the advantage of providing a permanent record of the absorbed dose to which it was exposed.
Various film formulations and filter arrangements may be used for special purposes, such as neutron dosimetry. As with thermoluminescent dosimeters, special equipment is needed for proper analysis.
Film is generally much more sensitive to ambient humidity and temperature than thermoluminescent materials, and can give falsely high readings under adverse conditions. On the other hand, dose equivalents indicated by thermoluminescent dosimeters may be affected by the shock of dropping them on a hard surface.
Only the largest of organizations operate their own dosimetry services. Most obtain such services from companies specializing in providing them. It is important that such companies be licensed or accredited by appropriate independent authorities so that accurate dosimetry results are assured.
Self-reading, small ionization chambers, also called pocket chambers, are used to obtain immediate dosimetry information. Their use is often required when personnel must enter high or very high radiation areas, where personnel could receive a large absorbed dose in a short period of time. Pocket chambers often are calibrated locally, and they are very sensitive to shock. Consequently, they should always be supplemented by thermoluminescent or film dosimeters, which are more accurate and dependable but do not provide immediate results.
Dosimetry is required for a worker when he or she has a reasonable probability of accumulating a certain percentage, usually 5 or 10%, of the maximum permissible dose equivalent for the whole-body or certain parts of the body.
A whole-body dosimeter should be worn somewhere between the shoulders and the waist, at a point where the highest exposure is anticipated. When conditions of exposure warrant, other dosimeters may be worn on fingers or wrists, at the abdomen, on a band or hat at the forehead, or on a collar, to assess localized exposure to extremities, a foetus or embryo, the thyroid or the lenses of the eyes. Refer to appropriate regulatory guidelines about whether dosimeters should be worn inside or outside protective garments such as lead aprons, gloves and collars.
Personnel dosimeters indicate only the radiation to which the dosimeter was exposed. Assigning the dosimeter dose equivalent to the person or organs of the person is acceptable for small, trivial doses, but large dosimeter doses, especially those greatly exceeding regulatory standards, should be analysed carefully with respect to dosimeter placement and the actual radiation fields to which the worker was exposed when estimating the dose that the worker actually received. A statement should be obtained from the worker as part of the investigation and included in the record. However, much more often than not, very large dosimeter doses are the result of deliberate radiation exposure of the dosimeter while it was not being worn.
Bioassay
Bioassay (also called radiobioassay) means the determination of kinds, quantities or concentrations, and, in some cases, the locations of radioactive material in the human body, whether by direct measurement (in vivo counting) or by analysis and evaluation of materials excreted or removed from the human body.
Bioassay is usually used to assess worker dose equivalent due to radioactive material taken into the body. It also can provide an indication of the effectiveness of active measures taken to prevent such intake. More rarely it may be used to estimate the dose a worker received from a massive external radiation exposure (for example, by counting white blood cells or chromosomal defects).
Bioassay must be performed when a reasonable possibility exists that a worker may take or has taken into his or her body more than a certain percentage (usually 5 or 10%) of the ALI for a radionuclide. The chemical and physical form of the radionuclide sought in the body determines the type of bioassay necessary to detect it.
Bioassay can consist of analysing samples taken from the body (for example, urine, faeces, blood or hair) for radioactive isotopes. In this case, the amount of radioactivity in the sample can be related to the radioactivity in the person’s body and subsequently to the radiation dose that the person’s body or certain organs have received or are committed to receive. Urine bioassay for tritium is an example of this type of bioassay.
Whole or partial body scanning can be used to detect radionuclides that emit x or gamma rays of energy reasonably detectable outside the body. Thyroid bioassay for iodine-131 (131I) is an example of this type of bioassay.
Bioassay can be performed in-house or samples or personnel can be sent to a facility or organization that specializes in the bioassay to be performed. In either case, proper calibration of equipment and accreditation of laboratory procedures is essential to ensure accurate, precise, and defensible bioassay results.
Protective Clothing
Protective clothing is supplied by the employer to the worker to reduce the possibility of radioactive contamination of the worker or his or her clothing or to partially shield the worker from beta, x, or gamma radiation. Examples of the former are anti-contamination clothing, gloves, hoods and boots. Examples of the latter are leaded aprons, gloves and eyeglasses.
Respiratory Protection
A respiratory protection device is an apparatus, such as a respirator, used to reduce a worker’s intake of airborne radioactive materials.
Employers must use, to the extent practical, process or other engineering controls (for example, containment or ventilation) to limit the concentrations of the radioactive materials in air. When this is not possible for controlling the concentrations of radioactive material in air to values below those that define an airborne radioactivity area, the employer, consistent with maintaining the total effective dose equivalent ALARA, must increase monitoring and limit intakes by one or more of the following means:
- control of access
- limitation of exposure times
- use of respiratory protection equipment
- other controls.
Respiratory protection equipment issued to workers must comply with applicable national standards for such equipment.
The employer must implement and maintain a respiratory protection programme that includes:
- air sampling sufficient to identify the potential hazard, permit proper equipment selection and estimate exposures
- surveys and bioassays, as appropriate, to evaluate actual intakes
- testing of respirators for operability immediately prior to each use
- written procedures regarding selection, fitting, issuance, maintenance and testing of respirators, including testing for operability immediately prior to each use; supervision and training of personnel; monitoring, including air sampling and bioassays; and record-keeping
- determination by a physician prior to the initial fitting of respirators, and periodically at a frequency determined by a physician, that the individual user is medically fit to use the respiratory protection equipment.
The employer must advise each respirator user that the user may leave the work area at any time for relief from respirator use in the event of equipment malfunction, physical or psychological distress, procedural or communication failure, significant deterioration of operating conditions, or any other conditions that might require such relief.
Even though circumstances may not require routine use of respirators, credible emergency conditions may mandate their availability. In such cases, the respirators also must be certified for such use by an appropriate accrediting organization and maintained in a condition ready for use.
Occupational Health Surveillance
Workers exposed to ionizing radiation should receive occupational health services to the same extent as workers exposed to other occupational hazards.
General preplacement examinations assess the overall health of the prospective employee and establish baseline data. Previous medical and exposure history should always be obtained. Specialized examinations, such as of lens of the eye and blood cell counts, may be necessary depending on the nature of the expected radiation exposure. This should be left to the discretion of the attending physician.
Contamination Surveys
A contamination survey is an evaluation of the radiological conditions incident to the production, use, release, disposal or presence of radioactive materials or other sources of radiation. When appropriate, such an evaluation includes a physical survey of the location of radioactive material and measurements or calculations of levels of radiation, or concentrations or quantities of radioactive material present.
Contamination surveys are performed to demonstrate compliance with national regulations and to evaluate the extent of radiation levels, concentrations or quantities of radioactive material, and the potential radiological hazards that could be present.
The frequency of contamination surveys is determined by the degree of potential hazard present. Weekly surveys should be performed in radioactive waste storage areas and in laboratories and clinics where relatively large amounts of unsealed radioactive sources are used. Monthly surveys suffice for laboratories that work with small amounts of radioactive sources, such as laboratories that perform in vitro testing using isotopes such as tritium, carbon-14 (14C), and iodine-125 (125I) with activities less than a few kBq.
Radiation safety equipment and survey meters must be appropriate for the types of radioactive material and radiations involved, and must be properly calibrated.
Contamination surveys consist of measurements of ambient radiation levels with a Geiger-Mueller (G-M) counter, ionization chamber or scintillation counter; measurements of possible α or βγ surface contamination with appropriate thin-window G-M or zinc sulphide (ZnS) scintillation counters; and wipe tests of surfaces to be later counted in a scintillation (sodium iodide (NaI)) well counter, a germanium (Ge) counter or a liquid scintillation counter, as appropriate.
Appropriate action levels must be established for ambient radiation and contamination measurement results. When an action level is exceeded, steps must be taken immediately to mitigate the detected levels, restore them to acceptable conditions and prevent unnecessary personnel exposure to radiation and the uptake and spread of radioactive material.
Environmental Monitoring
Environmental monitoring refers to collecting and measuring environmental samples for radioactive materials and monitoring areas outside the environs of the workplace for radiation levels. Purposes of environmental monitoring include estimating consequences to humans resulting from the release of radionuclides to the biosphere, detecting releases of radioactive material to the environment before they become serious and demonstrating compliance with regulations.
A complete description of environmental monitoring techniques is beyond the scope of this article. However, general principles will be discussed.
Environmental samples must be taken that monitor the most likely pathway for radionuclides from the environment to man. For example, soil, water, grass and milk samples in agricultural regions around a nuclear power plant should be taken routinely and analysed for iodine-131 (131I) and strontium-90 (90Sr) content.
Environmental monitoring can include taking samples of air, ground water, surface water, soil, foliage, fish, milk, game animals and so on. The choices of which samples to take and how often to take them should be based on the purposes of the monitoring, although a small number of random samples may sometimes identify a previously unknown problem.
The first step in designing an environmental monitoring programme is to characterize the radionuclides being released or having the potential for being accidentally released, with respect to type and quantity and physical and chemical form.
The possibility of transport of these radionuclides through the air, ground water and surface water is the next consideration. The objective is to predict the concentrations of radionuclides reaching humans directly through air and water or indirectly through food.
The bioaccumulation of radionuclides resulting from deposition in aquatic and terrestrial environments is the next item of concern. The goal is to predict the concentration of radionuclides once they enter the food chain.
Finally, the rate of human consumption of these potentially contaminated foodstuffs and how this consumption contributes to human radiation dose and resultant health risk are examined. The results of this analysis are used to determine the best approach to environmental sampling and to ensure that the goals of the environmental monitoring programme are met.
Leak Tests of Sealed Sources
A sealed source means radioactive material that is encased in a capsule designed to prevent leakage or escape of the material. Such sources must be tested periodically to verify that the source is not leaking radioactive material.
Each sealed source must be tested for leakage before its first use unless the supplier has provided a certificate indicating that the source was tested within six months (three months for α emitters) before transfer to the present owner. Each sealed source must be tested for leakage at least once every six months (three months for α emitters) or at an interval specified by the regulatory authority.
Generally, leak tests on the following sources are not required:
- sources containing only radioactive material with a half-life of less than 30 days
- sources containing only radioactive material as a gas
- sources containing 4 MBq or less of βγ-emitting material or 0.4 MBq or less of α-emitting material
- sources stored and not being used; however, each such source must be tested for leakage before any use or transfer unless it has been leakage-tested within six months before the date of use or transfer
- seeds of iridium-192 (192Ir) encased in nylon ribbon.
A leak test is performed by taking a wipe sample from the sealed source or from the surfaces of the device in which the sealed source is mounted or stored on which radioactive contamination might be expected to accumulate or by washing the source in a small volume of detergent solution and treating the entire volume as the sample.
The sample should be measured so that the leakage test can detect the presence of at least 200 Bq of radioactive material on the sample.
Sealed radium sources require special leak test procedures to detect leaking radon (Rn) gas. For example, one procedure involves keeping the sealed source in a jar with cotton fibres for at least 24 hours. At the end of the period, the cotton fibres are analysed for the presence of Rn progeny.
A sealed source found to be leaking in excess of allowable limits must be removed from service. If the source is not repairable, it should be handled as radioactive waste. The regulatory authority may require that leaking sources be reported in case the leakage is a result of a manufacturing defect worthy of further investigation.
Inventory
Radiation safety personnel must maintain an up-to-date inventory of all radioactive material and other sources of ionizing radiation for which the employer is responsible. The organization’s procedures must ensure that radiation safety personnel are aware of the receipt, use, transfer and disposal of all such material and sources so that the inventory can be kept current. A physical inventory of all sealed sources should be done at least once every three months. The complete inventory of ionizing radiation sources should be verified during the annual audit of the radiation safety programme.
Posting of Areas
Figure 1 shows the international standard radiation symbol. This must appear prominently on all signs denoting areas controlled for the purposes of radiation safety and on container labels indicating the presence of radioactive materials.
Figure 1. Radiation symbol
Areas controlled for the purposes of radiation safety are often designated in terms of increasing dose rate levels. Such areas must be conspicuously posted with a sign or signs bearing the radiation symbol and the words “CAUTION, RADIATION AREA,” “CAUTION (or DANGER), HIGH RADIATION AREA,” or “GRAVE DANGER, VERY HIGH RADIATION AREA,” as appropriate.
- A radiation area is an area, accessible to personnel, in which radiation levels could result in an individual receiving a dose equivalent in excess of 0.05 mSv in 1 h at 30 cm from the radiation source or from any surface that the radiation penetrates.
- A high radiation area is an area, accessible to personnel, in which radiation levels could result in an individual receiving a dose equivalent in excess of 1 mSv in 1 h at 30 cm from the radiation source or from any surface that the radiation penetrates.
- A very high radiation area is an area, accessible to personnel, in which radiation levels could result in an individual receiving an absorbed dose in excess of 5 Gy in 1 h at 1 m from a radiation source or from any surface that the radiation penetrates.
If an area or room contains a significant amount of radioactive material (as defined by the regulatory authority), the entrance to such area or room must be conspicuously posted with a sign bearing the radiation symbol and the words “CAUTION (or DANGER), RADIOACTIVE MATERIALS”.
An airborne radioactivity area is a room or area in which airborne radioactivity exceeds certain levels defined by the regulatory authority. Each airborne radioactivity area must be posted with a conspicuous sign or signs bearing the radiation symbol and the words “CAUTION, AIRBORNE RADIOACTIVITY AREA” or “DANGER, AIRBORNE RADIOACTIVITY AREA”.
Exceptions for these posting requirements may be granted for patients’ rooms in hospitals where such rooms are otherwise under adequate control. Areas or rooms in which the sources of radiation are to be located for periods of eight hours or less and are otherwise constantly attended under adequate control by qualified personnel need not be posted.
Access Control
The degree to which access to an area must be controlled is determined by the degree of the potential radiation hazard in the area.
Control of access to high radiation areas
Each entrance or access point to a high radiation area must have one or more of the following features:
- a control device that, upon entry into the area, causes the level of radiation to be reduced below that level at which an individual might receive a dose of 1 mSv in 1 h at 30 cm from the radiation source or from any surface that the radiation penetrates
- a control device that energizes a conspicuous visible or audible alarm signal so that the individual entering the high radiation area and the supervisor of the activity are made aware of the entry
- entryways that are locked, except during periods when access to the area is required, with positive control over each individual entry.
In place of the controls required for a high radiation area, continuous direct or electronic surveillance that is capable of preventing unauthorized entry may be substituted.
The controls must be established in a way that does not prevent individuals from leaving the high radiation area.
Control of access to very high radiation areas
In addition to the requirements for a high radiation area, additional measures must be instituted to ensure that an individual is not able to gain unauthorized or inadvertent access to areas in which radiation levels could be encountered at 5 Gy or more in 1 h at 1 m from a radiation source or any surface through which the radiation penetrates.
Markings on Containers and Equipment
Each container of radioactive material above an amount determined by the regulatory authority must bear a durable, clearly visible label bearing the radiation symbol and the words “CAUTION, RADIOACTIVE MATERIAL” or “DANGER, RADIOACTIVE MATERIAL”. The label must also provide sufficient information - such as the radionuclide(s) present, an estimate of the quantity of radioactivity, the date for which the activity is estimated, radiation levels, kinds of materials and mass enrichment - to permit individuals handling or using the containers, or working in the vicinity of the containers, to take precautions to avoid or minimize exposures.
Prior to removal or disposal of empty uncontaminated containers to unrestricted areas, the radioactive material label must be removed or defaced, or it must be clearly indicated that the container no longer contains radioactive materials.
Containers need not be labelled if:
- the containers are attended by an individual who takes the precautions necessary to prevent the exposure of individuals in excess of the regulatory limits
- containers, when they are in transport, are packaged and labelled in accordance with appropriate transportation regulations
- containers are accessible only to individuals authorized to handle or use them, or to work in the vicinity of the containers, if the contents are identified to these individuals by a readily available written record (examples of containers of this type are containers in locations such as water-filled canals, storage vaults or hot cells); the record must be retained as long as the containers are in use for the purpose indicated on the record; or
- the containers are installed in manufacturing or process equipment, such as reactor components, piping and tanks.
Warning Devices and Alarms
High radiation areas and very high radiation areas must be equipped with warning devices and alarms as discussed above. These devices and alarms can be visible or audible or both. Devices and alarms for systems such as particle accelerators should be automatically energized as part of the start-up procedure so that personnel will have time to vacate the area or turn off the system with a “scram” button before radiation is produced. “Scram” buttons (buttons in the controlled area that, when pressed, cause radiation levels to drop immediately to safe levels) must be easily accessible and prominently marked and displayed.
Monitor devices, such as continuous air monitors (CAMs), can be preset to emit audible and visible alarms or to turn off a system when certain action levels are exceeded.
Instrumentation
The employer must make available instrumentation appropriate for the degree and kinds of radiation and radioactive material present in the workplace. This instrumentation may be used to detect, monitor or measure the levels of radiation or radioactivity.
The instrumentation must be calibrated at appropriate intervals using accredited methods and calibration sources. The calibration sources should be as much as possible like the sources to be detected or measured.
Types of instrumentation include hand-held survey instruments, continuous air monitors, hand-and-feet portal monitors, liquid scintillation counters, detectors containing Ge or NaI crystals and so on.
Radioactive Material Transportation
The International Atomic Energy Agency (IAEA) has established regulations for the transportation of radioactive material. Most countries have adopted regulations compatible with IAEA radioactive shipment regulations.
Figure 2. Category I - WHITE label
Figure 2, figure 3 and figure 4 are examples of shipping labels that IAEA regulations require on the exterior of packages presented for shipment that contain radioactive materials. The transport index on the labels shown in figure 3 and figure 4 refer to the highest effective dose rate at 1 m from any surface of the package in mSv/h multiplied by 100, then rounded up to the nearest tenth. (For example, if the highest effective dose rate at 1 m from any surface of a package is 0.0233 mSv/h, then the transport index is 2.4.)
Figure 3. Category II - YELLOW label
Figure 5 shows an example of a placard that ground vehicles must prominently display when carrying packages containing radioactive materials above certain amounts.
Figure 5. Vehicle placard
Packaging intended for use in shipping radioactive materials must comply with stringent testing and documentation requirements. The type and quantity of radioactive material being shipped determines what specifications the packaging must meet.
Radioactive material transportation regulations are complicated. Persons who do not routinely ship radioactive materials should always consult experts experienced with such shipments.
Radioactive Waste
Various radioactive waste disposal methods are available, but all are controlled by regulatory authorities. Therefore, an organization must always confer with its regulatory authority to ensure that a disposal method is permissible. Radioactive waste disposal methods include holding the material for radioactive decay and subsequent disposal without regard to radioactivity, incineration, disposal in the sanitary sewerage system, land burial and burial at sea. Burial at sea is often not permitted by national policy or international treaty and will not be discussed further.
Radioactive waste from reactor cores (high-level radioactive waste) presents special problems with regard to disposal. Handling and disposal of such wastes is controlled by national and international regulatory authorities.
Often radioactive waste may have a property other than radioactivity that by itself would make the waste hazardous. Such wastes are called mixed wastes. Examples include radioactive waste that is also a biohazard or is toxic. Mixed wastes require special handling. Refer to regulatory authorities for proper disposition of such wastes.
Holding for radioactive decay
If the half-life of the radioactive material is short (generally less than 65 days) and if the organization has enough storage space, the radioactive waste can be held for decay with subsequent disposal without regard to its radioactivity. A holding period of at least ten half-lives usually is sufficient to make radiation levels indistinguishable from background.
The waste must be surveyed before it may be disposed of. The survey should employ instrumentation appropriate for the radiation to be detected and demonstrate that radiation levels are indistinguishable from background.
Incineration
If the regulatory authority allows incineration, then usually it must be demonstrated that such incineration does not cause the concentration of radionuclides in air to exceed permissible levels. The ash must be surveyed periodically to verify that it is not radioactive. In some circumstances it may be necessary to monitor the stack to ensure that permissible air concentrations are not being exceeded.
Disposal in the sanitary sewerage system
If the regulatory authority allows such disposal, then usually it must be demonstrated that such disposal does not cause the concentration of radionuclides in water to exceed permissible levels. Material to be disposed of must be soluble or otherwise readily dispersible in water. The regulatory authority often sets specific annual limits to such disposal by radionuclide.
Land burial
Radioactive waste not disposable by any other means will be disposed of by land burial at sites licensed by national or local regulatory authorities. Regulatory authorities control such disposal tightly. Waste generators usually are not allowed to dispose of radioactive waste on their own land. Costs associated with land burial include packaging, shipping and storage expenses. These costs are in addition to the cost of the burial space itself and can often be reduced by compacting the waste. Land burial costs for radioactive waste disposal are rapidly escalating.
Programme Audits
Radiation safety programmes should be audited periodically for effectiveness, completeness and compliance with regulatory authority. The audit should be done at least once a year and be comprehensive. Self-audits are usually permissible but audits by independent outside agencies are desirable. Outside agency audits tend to be more objective and have a more global point of view than local audits. An auditing agency not associated with day-to-day operations of a radiation safety programme often can identify problems not seen by the local operators, who may have become accustomed to overlooking them.
Training
Employers must provide radiation safety training to all workers exposed or potentially exposed to ionizing radiation or radioactive materials. They must provide initial training before a worker begins work and annual refresher training. In addition, each female worker of child-bearing age must be provided special training and information about the effects of ionizing radiation on the unborn child and about appropriate precautions she should take. This special training must be given when she is first employed, at annual refresher training, and if she notifies her employer that she is pregnant.
All individuals working in or frequenting any portion of an area access to which is restricted for the purposes of radiation safety:
- must be kept informed of the storage, transfer or use of radioactive materials or of radiation in such portions of the restricted area
- must be instructed in the health protection problems associated with exposure to such radioactive materials or radiation, in precautions or procedures to minimize exposure, and in the purposes and functions of protective devices employed
- must be instructed in, and instructed to observe, to the extent within the worker’s control, the applicable provisions of national and employer regulations for the protection of personnel from exposures to radiation or radioactive materials occurring in such areas
- must be instructed of their responsibility to report promptly to the employer any condition which may lead to or cause a violation of national or employer regulations or unnecessary exposure to radiation or to radioactive material
- must be instructed in the appropriate response to warnings made in the event of any unusual occurrence or malfunction that may involve exposure to radiation or radioactive material
- must be advised as to the radiation exposure reports that workers may request.
The extent of radiation safety instructions must be commensurate with potential radiological health protection problems in the controlled area. Instructions must be extended as appropriate to ancillary personnel, such as nurses who attend radioactive patients in hospitals and fire-fighters and police officers who might respond to emergencies.
Worker Qualifications
Employers must ensure that workers using ionizing radiation are qualified to perform the work for which they are employed. The workers must have the background and experience to perform their jobs safely, particularly with reference to exposure to and use of ionizing radiation and radioactive materials.
Radiation safety personnel must have the appropriate knowledge and qualifications to implement and operate a good radiation safety programme. Their knowledge and qualifications must be at least commensurate with the potential radiological health protection problems that they and the workers are reasonably likely to encounter.
Emergency Planning
All but the smallest operations that use ionizing radiation or radioactive materials must have emergency plans in place. These plans must be kept current and exercised on a periodic basis.
Emergency plans should address all credible emergency situations. The plans for a large nuclear power plant will be much more extensive and involve a much larger area and number of people than the plans for a small radioisotope laboratory.
All hospitals, especially in large metropolitan areas, should have plans for receiving and caring for radioactively contaminated patients. Police and fire-fighting organizations should have plans for dealing with transportation accidents involving radioactive material.
Record Keeping
The radiation safety activities of an organization must be fully documented and appropriately retained. Such records are essential if the need arises for past radiation exposures or radioactivity releases and for demonstrating compliance with regulatory authority requirements. Consistent, accurate and comprehensive record keeping must receive high priority.
Organizational Considerations
The position of the person primarily responsible for radiation safety must be placed in the organization so that he or she has immediate access to all echelons of workers and management. He or she must have free access to areas to which access is restricted for purposes of radiation safety and the authority to halt unsafe or illegal practices immediately.
Planning for and Management of Radiation Accidents
This article describes several significant radiation accidents, their causes and the responses to them. A review of the events leading up to, during and following these accidents can provide planners with information to preclude future occurrences of such accidents and to enhance an appropriate, rapid response in the event a similar accident occurs again.
Acute Radiation Death Resulting from an Accidental Nuclear Critical Excursion on 30 December 1958
This report is noteworthy because it involved the largest accidental dose of radiation received by humans (to date) and because of the extremely professional and thorough work-up of the case. This represents one of the best, if not the best, documented acute radiation syndrome descriptions that exists (JOM 1961).
At 4:35 p.m. on 30 December 1958, an accidental critical excursion resulting in fatal radiation injury to an employee (K) took place in the plutonium recovery plant at the Los Alamos National Laboratory (New Mexico, United States).
The time of the accident is important because six other workers had been in the same room with K thirty minutes earlier. The date of the accident is important because the normal flow of fissionable material into the system was interrupted for year-end physical inventory. This interruption caused a routine procedure to become non-routine and led to an accidental “criticality” of the plutonium-rich solids that were accidentally introduced into the system.
Summary of estimates of K’s radiation exposure
The best estimate of K’s average total-body exposure was between 39 and 49 Gy, of which about 9 Gy was due to fission neutrons. A considerably greater portion of the dose was delivered to the upper half of the body than to the lower half. Table 1 shows an estimate of K’s radiation exposure.
Table 1. Estimates of K’s radiation exposure
|
Region and conditions |
Fast neutron |
Gamma |
Total |
|
Head (incident) |
26 |
78 |
104 |
|
Upper abdomen |
30 |
90 |
124 |
|
Total body (average) |
9 |
30-40 |
39-49 |
Clinical course of patient
In retrospect, the clinical course of patient K can be divided into four distinct periods. These periods differed in duration, symptoms and response to supportive therapy.
The first period, lasting from 20 to 30 minutes, was characterized by his immediate physical collapse and mental incapacitation. His condition progressed to semi-consciousness and severe prostration.
The second period lasted about 1.5 hours and began with his arrival by stretcher at the emergency room of the hospital and ended with his transfer from the emergency room to the ward for further supportive therapy. This interval was characterized by such severe cardiovascular shock that death seemed imminent during the whole time. He seemed to be suffering severe abdominal pain.
The third period was about 28 hours long and was characterized by enough subjective improvement to encourage continued attempts to alleviate his anoxia, hypotension and circulatory failure.
The fourth period began with the unheralded onset of rapidly increasing irritability and antagonism, bordering on mania, followed by coma and death in approximately 2 hours. The entire clinical course lasted 35 hours from the time of radiation exposure to death.
The most dramatic clinicopathological changes were observed in the haemopoietic and urinary systems. Lymphocytes were not found in the circulating blood after the eighth hour, and there was virtually complete urinary shutdown despite administration of large amount of fluids.
K’s rectal temperature varied between 39.4 and 39.7°C for the first 6 hours and then fell precipitously to normal, where it remained for the duration of his life. This high initial temperature and its maintenance for 6 hours were considered in keeping with his suspected massive dose of radiation. His prognosis was grave.
Of all the various determinations made during the course of the illness, changes in white cell count were found to be the simplest and best prognostic indicator of severe irradiation. The virtual disappearance of lymphocytes from the peripheral circulation within 6 hours of exposure was considered a grave sign.
Sixteen different therapeutic agents were employed in the symptomatic treatment of K over about a 30-hour period. In spite of this and continued oxygen administration, his heart tones became very distant, slow and irregular about 32 hours after irradiation. His heart then became progressively weaker and suddenly stopped 34 hours 45 minutes after irradiation.
Windscale Reactor No. 1 Accident of 9-12 October 1957
Windscale reactor No. 1 was an air-cooled, graphite-moderated natural uranium-fuelled plutonium production reactor. The core was partially ruined by fire on 15 October 1957. This fire resulted in a release of approximately 0.74 PBq (10+15 Bq) of iodine-131 (131I) to the downwind environment.
According to a US Atomic Energy Commission accident information report about the Windscale incident, the accident was caused by operator judgement errors concerning thermocouple data and was made worse by faulty handling of the reactor that permitted the graphite temperature to rise too rapidly. Also contributory was the fact that fuel temperature thermocouples were located in the hottest part of the reactor (that is, where the highest dose rates occurred) during normal operations rather than in parts of the reactor which were hottest during an abnormal release. A second equipment deficiency was the reactor power meter, which was calibrated for normal operations and read low during the annealing. As a result of the second heating cycle, the graphite temperature rose on 9 October, especially in the lower front part of the reactor where some cladding had failed because of the earlier rapid temperature rise. Although there were a number of small iodine releases on 9 October, the releases were not recognized until 10 October when the stack activity meter showed a significant increase (which was not regarded as highly significant). Finally, on the afternoon of 10 October, other monitoring (Calder site) indicated the release of radioactivity. Efforts to cool the reactor by forcing air through it not only failed but actually increased the magnitude of the radioactivity released.
The estimated releases from the Windscale accident were 0.74 PBq of 131I, 0.22 PBq of caesium-137 (137Cs), 3.0 TBq (1012Bq) of strontium-89 (89Sr), and 0.33 TBq of strontium-90
(90Sr). The highest offsite gamma absorbed dose rate was about 35 μGy/h due to airborne activity. Air activity readings around the Windscale and Calder plants often were 5 to 10 times the maximum permissible levels, with occasional peaks of 150 times permissible levels. A milk ban extended over a radius of approximately 420 km.
During operations to bring the reactor under control, 14 workers received dose equivalents greater than 30 mSv per calendar quarter, with the maximum dose equivalent at 46 mSv per calendar quarter.
Lessons learned
There were many lessons learned concerning natural uranium reactor design and operation. The inadequacies concerning reactor instrumentation and reactor operator training also bring up points analogous to the Three Mile Island accident (see below).
No guidelines existed for short-term permissible exposure to radioiodine in food. The British Medical Research Council performed a prompt and thorough investigation and analysis. Much ingenuity was used in promptly deriving maximum permissible concentrations for 131I in food. The study Emergency Reference Levels that resulted from this accident serves as a basis for emergency planning guides now used worldwide (Bryant 1969).
A useful correlation was derived for predicting significant radioiodine contamination in milk. It was found that gamma radiation levels in pastures which exceeded 0.3 μGy/h yielded milk which exceeded 3.7 MBq/m3.
Absorbed dose from inhalation of external exposure to radioiodines is negligible compared to that from drinking milk or eating dairy products. In an emergency, rapid gamma spectroscopy is preferable to slower laboratory procedures.
Fifteen two-person teams performed radiation surveys and obtained samples. Twenty persons were used for sample coordination and data reporting. About 150 radiochemists were involved in sampling analysis.
Glass wool stack filters are not satisfactory under accident conditions.
Gulf Oil Accelerator Accident of 4 October 1967
Gulf Oil Company technicians were using a 3 MeV Van de Graaff accelerator for the activation of soil samples on 4 October 1967. The combination of an interlock failure on the power key of the accelerator console and the taping of several of the interlocks on the safety tunnel door and the target room inside door produced serious accidental exposures to three individuals. One individual received approximately 1 Gy whole-body dose equivalent, the second received close to 3 Gy whole-body dose equivalent and the third received approximately 6 Gy whole-body dose equivalent, in addition to approximately 60 Gy to the hands and 30 Gy to the feet.
One of the accident victims reported to the medical department, complaining of nausea, vomiting and generalized muscular aches. His symptoms initially were misdiagnosed as flu symptoms. When the second patient came in with approximately the same symptoms, it was decided that they may possibly have received significant radiation exposures. Film badges verified this. Dr. Niel Wald, University of Pittsburgh Radiological Health Division, supervised the dosimetry tests and also acted as coordinating physician in the work-up and treatment of the patients.
Dr. Wald very quickly had absolute filter units flown in to the western Pennsylvania hospital in Pittsburgh where the three patients had been admitted. He set up these absolute filter/laminar flow filters to clean the patients’ environment of all biological contaminants. These “reverse isolation” units were used on the 1 Gy exposure patient for about 16 days, and on the 3 and 6 Gy exposure patients for about a month and half.
Dr. E. Donnal Thomas from the University of Washington arrived to perform a bone marrow transplant on the 6 Gy patient on the eighth day after exposure. The patient’s twin brother served as the bone marrow donor. Although this heroic medical treatment saved the 6 Gy patient’s life, nothing could be done to save his arms and legs, each of which received tens-of-gray absorbed dose.
Lessons learned
If the simple operating procedure of always using a survey meter when entering the exposure room had been followed, this tragic accident would have been avoided.
At least two interlocks had been taped closed for long periods of time prior to this accident. Defeating of protective interlocks is intolerable.
Regular maintenance checks should have been made on the key-operated power interlocks for the accelerator.
Timely medical attention saved the life of the person with the highest exposure. The heroic procedure of a complete bone marrow transplant together with the use of reverse isolation and quality medical care were all major factors in saving this person’s life.
Reverse isolation filters can be obtained in a matter of hours to be set up in any hospital to care for highly exposed patients.
In retrospect, medical authorities involved with these patients would have recommended amputation earlier and at a definitive level within two or three months after the exposure. Earlier amputation decreases the likelihood of infection, gives a shorter period of severe pain, reduces pain medication required for the patient, possibly reduces the patient’s hospital stay, and possibly contributes to earlier rehabilitation. Earlier amputation should, of course, be done while correlating dosimetry information with clinical observations.
The SL–1 Prototype Reactor Accident (Idaho, USA, 3 January 1961)
This is the first (and to date the only) fatal accident in the history of US reactor operations. The SL-1 is a prototype of a small Army Package Power Reactor (APPR) designed for air transportation to remote areas for production of electrical power. This reactor was used for fuel testing, and for reactor crew training. It was operated in the remote desert location of the National Reactor Testing Station in Idaho Falls, Idaho, by Combustion Engineering for the US Army. The SL-1 was not a commercial power reactor (AEC 1961; American Nuclear Society 1961).
At the time of the accident, the SL-1 was loaded with 40 fuel elements and 5 control rod blades. It could produce a power level of 3 MW (thermal) and was a boiling water–cooled and –moderated reactor.
The accident resulted in the deaths of three military personnel. The accident was caused by the withdrawal of a single control rod for a distance of more than 1 m. This caused the reactor to go into prompt criticality. The reason why a skilled, licensed reactor operator with much refuelling operation experience withdrew the control rod past its normal stop point is unknown.
One of the three accident victims was still alive when initial response personnel first reached the scene of the accident. High activity fission products covered his body and were embedded in his skin. Portions of the victim’s skin registered in excess of 4.4 Gy/h at 15 cm and hampered rescue and medical treatment.
Lessons learned
No reactor designed since the SL-1 accident can be brought to “prompt-critical” state with a single control rod.
All reactors must have portable survey meters onsite that have ranges greater than 20 mGy/h. Survey meters of 10 Gy/h maximum range are recommended.
Note: The Three Mile Island accident showed that 100 Gy/h is the required range for both gamma and beta measurements.
Treatment facilities are required where a highly contaminated patient can receive definitive medical treatment with reasonable safeguards for attendant personnel. Since most of these facilities will be in clinics with other ongoing missions, control of airborne and waterborne radioactive contaminants may require special provisions.
X-ray Machines, Industrial and Analytical
Accidental exposures from x-ray systems are numerous and often involve extremely high exposures to small portions of the body. It is not unusual for x-ray diffraction systems to produce absorbed dose rates of 5 Gy/s at 10 cm from the tube focus. At shorter distances, 100 Gy/s rates have often been measured. The beam is usually narrow, but even a few seconds’ exposure can result in severe local injury (Lubenau et al. 1967; Lindell 1968; Haynie and Olsher 1981; ANSI 1977).
Because these systems are often used in “non-routine” circumstances, they lend themselves to the production of accidental exposures. X-ray systems commonly used in normal operations appear to be reasonably safe. Equipment failure has not caused severe exposures.
Lessons learned from accidental x-ray exposures
Most accidental exposures occurred during non-routine uses when equipment was partially disassembled or shield covers had been removed.
In most serious exposures, adequate instruction for the staff and maintenance personnel had been lacking.
If simple and fail-safe methods had been used to ensure that x-ray tubes were turned off during repairs and maintenance, many accidental exposures would have been avoided.
Finger or wrist personnel dosimeters should be used for operators and maintenance personnel working with these machines.
If interlocks had been required, many accidental exposures would have been avoided.
Operator error was a contributing cause in most of the accidents. Lack of adequate enclosures or poor shielding design often worsened the situation.
Industrial radiography accidents
From the 1950s through the 1970s, the highest radiation accident rate for a single activity has consistently been for industrial radiographic operations (IAEA 1969, 1977). National regulatory bodies continue to struggle to reduce the rate by a combination of improved regulations, strict training requirements and ever tougher inspection and enforcement policies (USCFR 1990). These regulatory efforts have generally succeeded, but many accidents associated with industrial radiography still occur. Legislation allowing huge monetary fines may be the most effective tool in keeping radiation safety focused in the minds of industrial radiography management (and also, therefore, in workers’ minds).
Causes of industrial radiography accidents
Worker training. Industrial radiography probably has lower education and training requirements than any other type of radiation employment. Therefore, existing training requirements must be strictly enforced.
Worker production incentive. For years, major emphasis for industrial radiographers was placed on the amount of successful radiographs produced per day. This practice can lead to unsafe acts as well as to occasional non-use of personnel dosimetry so that exceeding dose equivalent limits would not be detected.
Lack of proper surveys. Thorough surveying of source pigs (storage containers) (figure 1) after every exposure is most important. Not performing these surveys is the single most probable cause of unnecessary exposures, many of which are unrecorded, since industrial radiographers rarely use hand or finger dosimeters (figure 1).
Figure 1. Industrial radiography camera
Equipment problems. Because of heavy use of industrial radiographic cameras, source winding mechanisms can loosen and cause the source to not completely retract into its safe storage position (point A in figure 1). There are also many instances of closet-source interlock failures that cause accidental exposures of personnel.
Design of Emergency Plans
Many excellent guidelines, both general and specific, exist for the design of emergency plans. Some references are particularly helpful. These are given in the suggested readings at the end of this chapter.
Initial drafting of emergency plan and procedures
First, one must assess the entire radioactive material inventory for the subject facility. Then credible accidents must be analysed so that one can determine the probable maximum source release terms. Next, the plan and its procedures must enable the facility operators to:
- recognize an accident situation
- classify the accident according to severity
- take steps to mitigate the accident
- make timely notifications
- call for help efficiently and quickly
- quantify releases
- keep track of exposures both on- and offsite, as well as keep emergency exposures ALARA
- recover the facility as quickly as practical
- keep accurate and detailed records.
Types of accidents associated with nuclear reactors
A list, from most likely to least likely, of types of accidents associated with nuclear reactors follows. (The non-nuclear reactor, general-industrial type accident is by far the most likely.)
- Low level unexpected release of radioactive material with little or no external radiation exposure to personnel. Usually occurs during major overhauls or in shipment of spent resin or spent fuel. Coolant system leakage and coolant-sample sink spills are often causes of spread of radioactive contamination.
- Unexpected external exposure of personnel. This usually occurs during major overhauls or routine maintenance.
- A combination of contamination spread, contamination of personnel, and low-level personnel external radiation exposure is the next most likely accident. These accidents occur under the same conditions as 1 and 2 above.
- Gross surface contamination due to a major reactor coolant system leak or a leak of spent fuel coolant.
- Chips or large particles of activated CRUD (see definition below) in or on skin, ears or eyes.
- High-level radiation exposure of plant personnel. This is usually caused by carelessness.
- Release of small but greater than permissible quantities of radioactive wastes to outside the plant boundary. This is usually associated with human failures.
- Meltdown of reactor. Gross contamination offsite plus high personnel exposure would probably occur.
- Reactor excursion (SL–1 type of accident).
Radionuclides expected from water-cooled reactor accidents:
- activated corrosion and erosion products (commonly known as CRUD) in the coolant; for example, cobalt-60 or -58 (60Co, 58Co), iron-59 (59Fe), manganese-58 (58Mn) and tantalum-183 (183Ta)
- low level fission products usually present in the coolant; for example, iodine-131 (131I) and caesium-137 (137Cs)
- in boiling water reactors, 1 and 2 above plus continuous off-gassing of low levels of tritium
- (3H) and noble radioactive gases such as xenon-133 and -135 (133Xe, 135Xe), argon-41 (41Ar), and krypton-85 (85Kr)
- tritium (3H) manufactured inside the core at the rate of 1.3 × 10–4 atoms of 3H per fission (only a fraction of this leaves the fuel).
Figure 2. Example of a nuclear power plant emergency plan, table of contents
Typical Nuclear Power Plant Emergency Plan, Table of Contents
Figure 2 is an example of a table of contents for a nuclear power plant emergency plan. Such a plan should include each chapter shown and be tailored to meet local requirements. A list of typical power reactor implementation procedures is given in figure 3.
Figure 3. Typical power reactor implementation procedures
Radiological Environmental Monitoring during Accidents
This task is often called EREMP (Emergency Radiological Environmental Monitoring Programme) at large facilities.
One of the most important lessons learned for the US Nuclear Regulatory Commission and other government agencies from the Three Mile Island accident was that one cannot successfully implement EREMP in one or two days without extensive prior planning. Although the US government spent many millions of dollars monitoring the environment around the Three Mile Island nuclear station during the accident, less then 5% of the total releases were measured. This was due to poor and inadequate prior planning.
Designing Emergency Radiological Environmental Monitoring Programmes
Experience has shown that the only successful EREMP is one that is designed into the routine radiological environmental monitoring programme. During the early days of the Three Mile Island accident, it was learned that an effective EREMP cannot be established successfully in a day or two, no matter how much manpower and money are applied to the programme.
Sampling locations
All routine radiological environmental monitoring programme locations will be used during long-term accident monitoring. In addition, a number of new locations must be set up so that motorized survey teams have pre-determined locations in each portion of each 22½° sector (see figure 3). Generally, sampling locations will be in areas with roads. However, exceptions must be made for normally inaccessible but potentially occupied sites such as camp grounds and hiking trails within about 16 km downwind of the accident.
Figure 3. Sector and zone designations for radiological sampling and monitoring points within emergency planning zones
Figure 3 shows the sector and zone designation for radiation and environmental monitoring points. One may designate 22½° sectors by cardinal directions (for example, N, NNE, and NE) or by simple letters (for example, A through R). However, use of letters is not recommended because they are easily confused with directional notation. For example, it is less confusing to use the directional W for west rather than the letter N.
Each designated sample location should be visited during a practice drill so that people responsible for monitoring and sampling will be familiar with the location of each point and will be aware of radio “dead spaces,” poor roads, problems with finding the locations in the dark and so on. Since no drill will cover all the pre-designated locations within the 16 km emergency protection zone, drills must be designed so that all sample points will be visited eventually. It is often worthwhile to predetermine the ability of survey team vehicles to communicate with each pre-designated point. The actual locations of the sample points are chosen utilizing the same criteria as in the REMP (NRC 1980); for example, line of site, minimum exclusion area, closest individual, closest community, closest school, hospital, nursing home, milch animal herd, garden, farm and so on.
Radiological monitoring survey team
During an accident involving significant releases of radioactive materials, radiological monitoring teams should be continuously monitoring in the field. They also should continuously monitor onsite if conditions allow. Normally, these teams will monitor for ambient gamma and beta radiation and sample air for the presence of radioactive particulates and halogens.
These teams must be well trained in all monitoring procedures, including monitoring their own exposures, and be able to accurately relay these data to the base station. Details such as survey-meter type, serial number, and open-or closed-window status must be carefully reported on well-designed log sheets.
At the beginning of an emergency, an emergency monitoring team may have to monitor for 12 hours without a break. After the initial period, however, field time for the survey team should be decreased to eight hours with at least one 30 minute break.
Since continuous surveillance may be needed, procedures must be in place to supply the survey teams with food and drink, replacement instruments and batteries, and for back-and-forth transfer of air filters.
Even though survey teams will probably be working 12 hours per shift, three shifts a day are needed to provide continuous surveillance. During the Three Mile Island accident, a minimum of five monitoring teams was deployed at any one time for the first two weeks. The logistics for supporting such an effort must be carefully planned in advance.
Radiological environmental sampling team
The types of environmental samples taken during an accident depend on the type of releases (airborne versus water), direction of the wind and time of year. Soil and drinking water samples must be taken even in winter. Although radio-halogen releases may not be detected, milk samples should be taken because of the large bioaccumulation factor.
Many food and environmental samples must be taken to reassure the public even though technical reasons may not justify the effort. In addition, these data may be invaluable during any subsequent legal proceedings.
Pre-planned log sheets using carefully thought out offsite data procedures are essential for environmental samples. All persons taking environmental samples should have demonstrated a clear understanding of procedures and have documented field training.
If possible, offsite environmental sample data collection should be done by an independent offsite group. It is also preferable that routine environmental samples be taken by the same offsite group, so that the valuable onsite group may be used for other data collection during an accident.
It is notable that during the Three Mile Island accident every single environmental sample that should have been taken was collected, and not one environmental sample was lost. This occurred even though the sampling rate increased by a factor of more than ten over pre-accident sampling rates.
Emergency monitoring equipment
The inventory of emergency monitoring equipment should be at least double that needed at any given time. Lockers should be placed around nuclear complexes in various places so that no one accident will deny access to all of these lockers. To ensure readiness, equipment should be inventoried and its calibration checked at least twice a year and after each drill. Vans and trucks at large nuclear facilities should be completely outfitted for both on and offsite emergency surveillance.
Onsite counting laboratories may be unusable during an emergency. Therefore, prior arrangements must be made for an alternate or a mobile counting laboratory. This is now a requirement for US nuclear power plants (USNRC 1983).
The type and sophistication of environmental monitoring equipment should meet the requirements of attending the nuclear facility’s worst credible accident. Following is a list of typical environmental monitoring equipment required for nuclear power plants:
- Air sampling equipment should include units which are battery operated for short-term sampling and AC operable with strip chart recorders and alarm capabilities for longer-term surveillance.
- Liquid sampling equipment should contain continuous samplers. The samplers must be operable in the local environment, no matter how harsh it is.
- Portable gamma survey meters for implant work should have a maximum range of 100 Gy/h, and separate survey equipment should be able to measure beta radiation up to 100 Gy/h.
- Onsite, personnel dosimetry must include beta measurement capability, as well as finger thermoluminescent dosimeters (TLDs) (figure 4). Other extremity dosimetry also may be needed. Extra sets of control dosimeters are always needed in emergencies. A portable TLD reader may be needed to link with the station computer via telephone modem in emergency locations. In-house survey teams, such as rescue and repair teams, should have low- and high-range pocket dosimeters as well as pre-set alarm dosimeters. Careful thought must be given to pre-established dose levels for teams that may be in high radiation areas.
- Supplies of protective clothing should be supplied in emergency locations and in emergency vehicles. Extra back-up protective clothing should be available in case of accidents lasting for an extended period of time.
- Respiratory protection equipment should be in all emergency lockers and vehicles. Up-to-date lists of respiratory trained personnel should be kept in each of the major emergency equipment storage areas.
- Mobile vehicles equipped with radios are essential for emergency radiation monitoring survey teams. The location and availability of back-up vehicles must be known.
- Environmental survey team equipment should be stored in a convenient place, preferably offsite, so that it is always available.
- Emergency kits should be placed in the Technical Support Center and the Emergency Offsite Facility so that replacement survey teams need not go onsite in order to receive equipment and be deployed.
- For a severe accident involving the release of radioactive materials into the air, preparations must be in place for the use of helicopters and single-engine airplanes for airborne surveillance.
Figure 4. An industrial radiographer wearing a TLD badge and a ring thermoluminescent dosimeter (optional in the US)
Data analysis
Environmental data analysis during a serious accident should be shifted as soon as possible to an offsite location such as the Emergency Offsite Facility.
Pre-set guidelines about when environmental sample data are to be reported to management must be established. The method and frequency for transfer of environmental sample data to governmental agencies should be agreed upon early in the accident.
Health Physics and Radiochemistry Lessons Learned from the Three Mile Island Accident
Outside consultants were needed to perform the following activities because plant health physicists were fully occupied by other duties during the early hours of the 28 March 1979 Three Mile Island accident:
- radioactive effluent release assessment (gaseous and liquid), including sample collection, coordination of laboratories for sample counting, quality control of laboratories, data collection, data analysis, report generation, distribution of data to government agencies and power plant owner
- dose assessment, including suspected and actual overexposure investigations, skin contamination and internal deposition investigations, significant exposure mock-ups, and dose calculations
- radiological environmental monitoring programme, including complete coordination of sample taking, data analysis, report generation and distribution, action-point notifications, expansion of programme for the accident situation and then contraction of the programme for up to one year after the accident
- special beta dosimetry studies, including studies of the state of the art in beta personnel monitoring, modelling of the beta dose to skin from radioactive contaminants, inter-comparisons of all commercially available beta-gamma TLD personnel dosimetry systems.
The above list includes examples of activities that the typical utility health physics staff cannot adequately accomplish during a serious accident. The Three Mile Island health physics staff was very experienced, knowledgeable and competent. They worked 15 to 20 hours per day for the first two weeks of the accident without a break. Yet, additional requirements caused by the accident were so numerous that they were unable to perform many important routine tasks that ordinarily would be performed easily.
Lessons learned from the Three Mile Island accident include:
Auxiliary building entry during accident
- All entries must be on a new radiation work permit reviewed by the senior health physicist onsite and signed by the unit superintendent or designated alternate.
- The appropriate control room should have absolute control over all Auxiliary and Fuel Handling Building entries. No entries must be allowed unless a health physicist is at the control point during the entry.
- No entries without a properly operating survey meter of appropriate range should be allowed. A spot check of meter response should be performed immediately prior to entry.
- Exposure history for all persons prior to their entry into a high radiation area must be obtained.
- Allowable exposures during entry, no matter how important the task should be designated.
Primary coolant sampling during accident
- All samples to be taken on a new radiation work permit should be reviewed by the senior health physicist onsite and signed by the unit superintendent or alternate.
- No coolant samples should be taken unless an extremity dosimeter is worn.
- No coolant samples should be taken without the availability of shielded gloves and tongs at least 60 cm long in case a sample is more radioactive than expected.
- No coolant samples should be taken without a leaded-glass personnel shield in place in case a sample is more radioactive than expected.
- Sample-taking should be discontinued if the exposure to an extremity or to the whole-body is likely to exceed pre-set levels stated on the radiation work permit.
- Significant exposures should be distributed among a number of workers if possible.
- All cases of skin contamination in excess of action levels within 24 hours should be reviewed.
Make-up valve room entry
- Beta and gamma area surveys using remote detectors with appropriate maximum range must be performed.
- Initial entry in an area with an absorbed dose rate of more than 20 mGy/h must have prior review to verify that exposure to radiation will be kept as low as reasonably achievable.
- When water leaks are suspected, possible floor contamination should be detected.
- A consistent programme for type and placement of personnel dosimetry must be put into operation.
- With persons entering an area with an absorbed dose rate of more than 20 mGy/h, TLDs must be assessed immediately after exit.
- It should be verified that all radiation work permit requirements are being carried out prior to entry into an area with an absorbed dose rate of more than 20 mGy/h.
- Controlled-time entries into hazardous areas must be timed by a health physicist.
Protective actions and offsite environmental surveillance from the local government’s perspective
- Before beginning a sampling protocol, criteria for stopping it should be established.
- Outside interference should not be allowed.
- Several confidential telephone lines should be in place. The numbers should be changed after each crisis.
- The capabilities of aerial measuring systems are better than most people think they are.
- A tape recorder should be in hand and data recorded regularly.
- While the acute episode is in progress, the reading of newspapers, watching television and listening to the radio should be abandoned as these activities only add to existing tensions.
- Food delivery and other comforts such as sleeping facilities should be planned for as it may be impossible to go home for a while.
- Alternate analytic capabilities should be planned for. Even a small accident can alter laboratory background radiation levels significantly.
- It should be noted that more energy will be expended in heading off unsound decisions than in dealing with real problems.
- It should be understood that emergencies cannot be managed from remote locations.
- It should be noted that protective action recommendations are not amenable to committee vote.
- All non-essential calls should be put on hold, time-wasters are to be hung up on.
The Goiânia Radiological Accident of 1985
A 51 TBq 137Cs teletherapy unit was stolen from an abandoned clinic in Goiânia, Brazil, on or around 13 September 1985. Two people looking for scrap metal took home the source assembly of the teletherapy unit and attempted to disassemble the parts. The absorbed dose rate from the source assembly was about 46 Gy/h at 1 m. They did not understand the meaning of the three-bladed radiation symbol on the source capsule.
The source capsule ruptured during disassembly. Highly soluble caesium-137 chloride (137CsCl) powder was disbursed throughout a part of this city of 1,000,000 people and caused one of the most serious sealed source accidents in history.
After the disassembly, remnants of the source assembly were sold to a junk dealer. He discovered that the 137CsCl powder glowed in the dark with a blue colour (presumably, this was Cerenkov radiation). He thought that the powder could be a gemstone or even supernatural. Many friends and relatives came to see the “wonderful” glow. Portions of the source were given to a number of families. This process continued for about five days. By this time a number of people had developed gastro-intestinal syndrome symptoms from radiation exposure.
Patients who went to the hospital with severe gastro-intestinal disorders were misdiagnosed as having allergic reactions to something they ate. A patient who had severe skin effects from handling the source was suspected of having some tropical skin disease and was sent to the Tropical Disease Hospital.
This tragic sequence of events continued undetected by knowledgeable personnel for about two weeks. Many people rubbed the 137CsCl powder on their skins so that they could glow blue. The sequence might have continued much longer except that one of the irradiated persons finally connected the illnesses with the source capsule. She took the remnants of the 137CsCl source on a bus to the Public Health Department in Goiânia where she left it. A visiting medical physicist surveyed the source the next day. He took actions on his own initiative to evacuate two junkyard areas and to inform authorities. The speed and overall size of response of the Brazilian government, once it became aware of the accident, were impressive.
About 249 people were contaminated. Fifty-four were hospitalized. Four people died, one of whom was a six-year-old girl who received an internal dose of about 4 Gy from ingesting about 1 GBq (109 Bq) of 137Cs.
Response to the accident
The objectives of the initial response phase were to:
- identify the main sites of contamination
- evacuate residences where levels of radioactivity exceeded the intervention levels adopted
- establish health physics controls around these areas, preventing access where necessary
- identify persons who had incurred significant doses or were contaminated.
The medical team initially:
- upon its arrival in Goiânia, took histories and triaged according to acute radiation syndrome symptoms
- sent all acute radiation patients to Goiânia Hospital (which was set up in advance for contamination and exposure control)
- transferred by air the next day the six most critical patients to the tertiary care center at a naval hospital in Rio de Janeiro (later eight more patients were transferred to this hospital)
- made arrangements for cytogenetic radiation dosimetry
- based medical management on each patient on that patient’s clinical course
- gave informal instruction to clinical laboratory staff to diminish their fears (the Goiânia medical community was reluctant to help).
Health physicists:
- assisted physicians in radiation dosimetry, bioassay and skin decontamination
- coordinated and interpreted analysis of 4,000 urine and faecal samples in a four-month period
- whole-body counted 600 individuals
- coordinated radio-contamination monitoring of 112,000 individuals (249 were contaminated)
- performed aerial survey of entire city and suburbs utilizing hastily assembled NaI detectors
- performed auto-mounted NaI detector surveys of over 2,000 km of roads
- set up action levels for decontamination of people, buildings, autos, soil and so on
- coordinated 550 workers employed in decontamination efforts
- coordinated demolition of seven houses and decontamination of 85 houses
- coordinated hauling of 275 truckloads of contaminated waste
- coordinated decontamination of 50 vehicles
- coordinated packaging of 3,500 cubic metres of contaminated waste
- utilized 55 survey meters, 23 contamination monitors and 450 self-reading dosimeters.
Results
Acute radiation syndrome patients
Four patients died as a result of absorbed doses ranging from 4 to 6 Gy. Two patients exhibited severe bone marrow depression, but lived in spite of absorbed doses of 6.2 and 7.1 Gy (cytogenetic estimate). Four patients survived with estimated absorbed doses from 2.5 to 4 Gy.
Radiation-induced skin injury
Nineteen of twenty hospitalized patients had radiation-induced skin injuries, which started with swelling and blistering. These lesions later ruptured and secreted fluid. Ten of the nineteen skin injuries developed deep lesions about four to five weeks after irradiation. These deep lesions were indicative of significant gamma exposure of deeper tissues.
All skin lesions were contaminated with 137Cs, with absorbed dose rates up to 15 mGy/h.
The six-year-old girl who ingested 1 TBq of 137Cs (and who died one month later) had generalized skin contamination that averaged 3 mGy/h.
One patient required an amputation about a month after exposure. Blood-pool imaging was useful in determining the demarcation between injured and normal arterioles.
Internal contamination result
Statistical tests showed no significant differences between body burdens determined by whole body counting as opposed to those determined by urinary excretion data.
Models that related bioassay data with intakes and body burden were validated. These models were also applicable for different age groups.
Prussian Blue was useful in promoting the elimination of 137CsCl from the body (if dosage was greater than 3 Gy/d).
Seventeen patients received diuretics for the elimination of 137CsCl body burdens. These diuretics were ineffective in de-corporating 137Cs and their use was stopped.
Skin decontamination
Skin decontamination using soap and water, acetic acid, and titanium dioxide (TiO2) was performed on all patients. This decontamination was only partly successful. It was surmised that sweating resulted in recontaminating the skin from the 137Cs body burden.
Contaminated skin lesions are very difficult to decontaminate. Sloughing of necrotic skin significantly reduced contamination levels.
Follow-up study on cytogenetic analysis dose assessment
Frequency of aberrations in lymphocytes at different times after the accident followed three main patterns:
In two cases the frequencies of incidence of aberrations remained constant up to one month after the accident and declined to about 30% of the initial frequency three months later.
In two cases a gradual decrease of about 20% every three months was found.
In two of the cases of highest internal contamination there were increases in the frequency of incidence of aberrations (by about 50% and 100%) over a three-month period.
Follow-up studies on 137Cs body burdens
- Patients’ actual committed doses followed by bioassay.
- Effects of Prussian Blue administration followed.
- In vivo measurements for 20 people made on blood samples, wounds and organs to look for non-homogenous distribution of 137Cs and its retention in body tissues.
- A woman and her newborn baby studied to look for retention and transfer by nursing.
Action levels for intervention
House evacuation was recommended for absorbed dose rates greater than 10 μGy/h at 1 m height inside the house.
Remedial decontamination of property, clothing, soil and food was based on a person not exceeding 5 mGy in a year. Applying this criterion for different pathways resulted in decontaminating the inside of a house if the absorbed dose could exceed 1 mGy in a year and decontaminating soil if the absorbed dose rate could exceed 4 mGy in a year (3 mGy from external radiation and 1 mGy from internal radiation).
The Chernobyl Nuclear Power Reactor Unit 4 Accident of 1986
General description of the accident
The world’s worst nuclear power reactor accident occurred on 26 April 1986 during a very low-powered electrical engineering test. In order to perform this test, a number of safety systems were switched off or blocked.
This unit was a model RBMK-1000, the type of reactor that produced about 65% of all nuclear power generated in the USSR. It was a graphite-moderated, boiling-water reactor that generated 1,000 MW of electricity (MWe). The RBMK-1000 does not have a pressure-tested containment building and is not commonly built in most countries.
The reactor went prompt critical and produced a series of steam explosions. The explosions blew off the entire top of the reactor, destroyed the thin structure covering the reactor, and started a series of fires on the thick asphalt roofs of units 3 and 4. Radioactive releases lasted for ten days, and 31 people died. The USSR delegation to the International Atomic Energy Agency studied the accident. They stated that the Chernobyl Unit 4 RBMK experiments that caused the accident had not received required approval and that the written rules on reactor safety measures were inadequate. The delegation further stated, “The staff involved were not adequately prepared for the tests and were not aware of the possible dangers.” This series of tests created the conditions for the emergency situation and led to a reactor accident which most believed could never occur.
Release of Chernobyl Unit 4 accident fission products
Total activity released
Roughly 1,900 PBq of fission products and fuel (which together were labelled corium by the Three Mile Island Accident Recovery Team) were released over the ten days that it took to put out all the fires and seal off Unit 4 with a neutron absorbing shielding material. Unit 4 is now a permanently sealed steel and concrete sarcophagus that properly contains the residual corium in and around the remains of the destroyed reactor core.
Twenty-five per cent of the 1,900 PBq was released on the first day of the accident. The rest was released during the next nine days.
The most radiologically significant releases were 270 PBq of 131I, 8.1 PBq of 90Sr and 37 PBq of 137Cs. This can be compared with the Three Mile Island accident, which released 7.4 TBq of 131I and no measurable 90Sr or 137Cs.
Environmental dispersion of radioactive materials
The first releases went in a generally northern direction, but subsequent releases went toward the westerly and southwesterly directions. The first plume arrived in Sweden and Finland on 27 April. Nuclear power plant radiological environmental monitoring programmes immediately discovered the release and alerted the world about the accident. Part of this first plume drifted into Poland and East Germany. Subsequent plumes swept into eastern and central Europe on 29 and 30 April. After this, the United Kingdom saw Chernobyl releases on 2 May, followed by Japan and China on 4 May, India on 5 May and Canada and the US on 5 and 6 May. The southern hemisphere did not report detecting this plume.
The deposition of the plume was governed mostly by precipitation. The fallout pattern of the major radionuclides (131I, 137Cs, 134Cs, and 90Sr) was highly variable, even within the USSR. The major risk came from external irradiation from surface deposition, as well as from ingestion of contaminated food.
Radiological consequences of the Chernobyl Unit 4 accident
General acute health consequences
Two persons died immediately, one during the building collapse and one 5.5 hours later from thermal burns. An additional 28 of the reactor’s staff and fire-fighting crew died from radiation injuries. Radiation doses to the offsite population were below levels that can cause immediate radiation effects.
The Chernobyl accident almost doubled the worldwide total of deaths due to radiation accidents through 1986 (from 32 to 61). (It is interesting to note that the three dead from the SL-1 reactor accident in the US are listed as due to a steam explosion and that the first two to die at Chernobyl are also not listed as radiation accident deaths.)
Factors which influenced onsite health consequences of the accident
Personnel dosimetry for the onsite persons at highest risk was not available. The absence of nausea or vomiting for the first six hours after exposure reliably indicated those patients who had received less than potentially fatal absorbed doses. This also was a good indication of patients who did not require immediate medical attention because of radiation exposure. This information together with blood data (decrease in lymphocyte count) was more useful than personnel dosimetry data.
Fire-fighters’ heavy protective garments (a porous canvas) allowed high specific activity fission products to contact bare skin. These beta doses caused severe skin burns and were a significant factor in many of the deaths. Fifty-six workers received severe skin burns. The burns were extremely difficult to treat and were a serious complicating element. They made it impossible to decontaminate the patients prior to transport to hospitals.
There were no clinically significant internal radioactive material body burdens at this time. Only two people had high (but not clinically significant) body burdens.
Of the about 1,000 people screened, 115 were hospitalized due to acute radiation syndrome. Eight medical attendants working onsite incurred the acute radiation syndrome.
As expected, there was no evidence of neutron exposure. (The test looks for sodium-24 (24Na) in blood.)
Factors which influenced offsite health consequences of the accident
Public protective actions can be divided into four distinct periods.
- The first 24 h: The downwind public remained indoors with doors and windows shut. Distribution of potassium iodide (KI) began in order to block thyroid uptake of 131I.
- One to seven days: Pripyat was evacuated after safe evacuation routes were established. Decontamination stations were established. The Kiev region was evacuated. The total number of people evacuated was more than 88,000.
- One to six weeks: The total number of evacuated people rose to 115,000. All these were medically examined and resettled. Potassium iodide was administered to 5.4 million Russians, including 1.7 million children. Thyroid doses were reduced by about 80 to 90%. Tens of thousands of cattle were removed from contaminated areas. Local milk and foodstuffs were banned over a large area (as dictated by derived intervention levels).
- After 6 weeks: The 30 km radius circle of evacuation was divided into three sub-zones: (a) a zone of 4 to 5 km where no public re-entry is expected in the foreseeable future, (b) a 5 to 10 km zone where limited public re-entry will be allowed after a specific time and (c) a 10 to 30 km zone where the public will eventually be allowed to return.
A great effort has been expended in decontaminating offsite areas.
The total radiological dose to the USSR population was reported by the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) to be 226,000 person-Sv (72,000 person-Sv committed during the first year). The worldwide estimated collective dose equivalent is on the order of 600,000 person-Sv. Time and further study will refine this estimate (UNSCEAR 1988).
International Organizations
International Atomic Energy Agency
P.O. Box 100
A-1400 Vienna
AUSTRIA
International Commission on Radiation Units and Measurements
7910 Woodmont Avenue
Bethesda, Maryland 20814
U.S.A.
International Commission on Radiological Protection
P.O. Box No. 35
Didcot, Oxfordshire
OX11 0RJ
U.K.
International Radiation Protection Association
Eindhoven University of Technology
P.O. Box 662
5600 AR Eindhoven
NETHERLANDS
United Nations Committee on the Effects of Atomic Radiation
BERNAM ASSOCIATES
4611-F Assembly Drive
Lanham, Maryland 20706-4391
U.S.A.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."