Mechanisms of Toxicity
Introduction and Concepts
Mechanistic toxicology is the study of how chemical or physical agents interact with living organisms to cause toxicity. Knowledge of the mechanism of toxicity of a substance enhances the ability to prevent toxicity and design more desirable chemicals; it constitutes the basis for therapy upon overexposure, and frequently enables a further understanding of fundamental biological processes. For purposes of this Encyclopaedia the emphasis will be placed on animals to predict human toxicity. Different areas of toxicology include mechanistic, descriptive, regulatory, forensic and environmental toxicology (Klaassen, Amdur and Doull 1991). All of these benefit from understanding the fundamental mechanisms of toxicity.
Why Understand Mechanisms of Toxicity?
Understanding the mechanism by which a substance causes toxicity enhances different areas of toxicology in different ways. Mechanistic understanding helps the governmental regulator to establish legally binding safe limits for human exposure. It helps toxicologists in recommending courses of action regarding clean-up or remediation of contaminated sites and, along with physical and chemical properties of the substance or mixture, can be used to select the degree of protective equipment required. Mechanistic knowledge is also useful in forming the basis for therapy and the design of new drugs for treatment of human disease. For the forensic toxicologist the mechanism of toxicity often provides insight as to how a chemical or physical agent can cause death or incapacitation.
If the mechanism of toxicity is understood, descriptive toxicology becomes useful in predicting the toxic effects of related chemicals. It is important to understand, however, that a lack of mechanistic information does not deter health professionals from protecting human health. Prudent decisions based on animal studies and human experience are used to establish safe exposure levels. Traditionally, a margin of safety was established by using the “no adverse effect level” or a “lowest adverse effect level” from animal studies (using repeated-exposure designs) and dividing that level by a factor of 100 for occupational exposure or 1,000 for other human environmental exposure. The success of this process is evident from the few incidents of adverse health effects attributed to chemical exposure in workers where appropriate exposure limits had been set and adhered to in the past. In addition, the human lifespan continues to increase, as does the quality of life. Overall the use of toxicity data has led to effective regulatory and voluntary control. Detailed knowledge of toxic mechanisms will enhance the predictability of newer risk models currently being developed and will result in continuous improvement.
Understanding environmental mechanisms is complex and presumes a knowledge of ecosystem disruption and homeostasis (balance). While not discussed in this article, an enhanced understanding of toxic mechanisms and their ultimate consequences in an ecosystem would help scientists to make prudent decisions regarding the handling of municipal and industrial waste material. Waste management is a growing area of research and will continue to be very important in the future.
Techniques for Studying Mechanisms of Toxicity
The majority of mechanistic studies start with a descriptive toxicological study in animals or clinical observations in humans. Ideally, animal studies include careful behavioural and clinical observations, careful biochemical examination of elements of the blood and urine for signs of adverse function of major biological systems in the body, and a post-mortem evaluation of all organ systems by microscopic examination to check for injury (see OECD test guidelines; EC directives on chemical evaluation; US EPA test rules; Japan chemicals regulations). This is analogous to a thorough human physical examination that would take place in a hospital over a two- to three-day time period except for the post-mortem examination.
Understanding mechanisms of toxicity is the art and science of observation, creativity in the selection of techniques to test various hypotheses, and innovative integration of signs and symptoms into a causal relationship. Mechanistic studies start with exposure, follow the time-related distribution and fate in the body (pharmacokinetics), and measure the resulting toxic effect at some level of the system and at some dose level. Different substances can act at different levels of the biological system in causing toxicity.
Exposure
The route of exposure in mechanistic studies is usually the same as for human exposure. Route is important because there can be effects that occur locally at the site of exposure in addition to systemic effects after the chemical has been absorbed into the blood and distributed throughout the body. A simple yet cogent example of a local effect would be irritation and eventual corrosion of the skin following application of strong acid or alkaline solutions designed for cleaning hard surfaces. Similarly, irritation and cellular death can occur in cells lining the nose and/or lungs following exposure to irritant vapours or gases such as oxides of nitrogen or ozone. (Both are constituents of air pollution, or smog). Following absorption of a chemical into blood through the skin, lungs or gastrointestinal tract, the concentration in any organ or tissue is controlled by many factors which determine the pharmacokinetics of the chemical in the body. The body has the ability to activate as well as detoxify various chemicals as noted below.
Role of Pharmacokinetics in Toxicity
Pharmacokinetics describes the time relationships for chemical absorption, distribution, metabolism (biochemical alterations in the body) and elimination or excretion from the body. Relative to mechanisms of toxicity, these pharmacokinetic variables can be very important and in some instances determine whether toxicity will or will not occur. For instance, if a material is not absorbed in a sufficient amount, systemic toxicity (inside the body) will not occur. Conversely, a highly reactive chemical that is detoxified quickly (seconds or minutes) by digestive or liver enzymes may not have the time to cause toxicity. Some polycyclic halogenated substances and mixtures as well as certain metals like lead would not cause significant toxicity if excretion were rapid; but accumulation to sufficiently high levels determines their toxicity since excretion is not rapid (sometimes measured in years). Fortunately, most chemicals do not have such long retention in the body. Accumulation of an innocuous material still would not induce toxicity. The rate of elimination from the body and detoxication is frequently referred to as the half-life of the chemical, which is the time for 50% of the chemical to be excreted or altered to a non-toxic form.
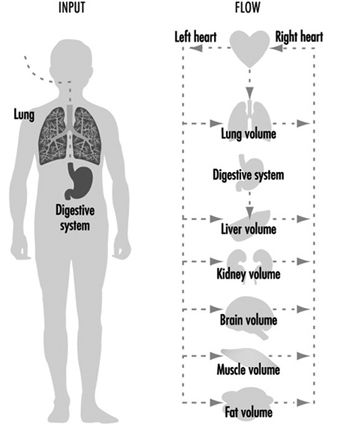
However, if a chemical accumulates in a particular cell or organ, that may signal a reason to further examine its potential toxicity in that organ. More recently, mathematical models have been developed to extrapolate pharmacokinetic variables from animals to humans. These pharmacokinetic models are extremely useful in generating hypotheses and testing whether the experimental animal may be a good representation for humans. Numerous chapters and texts have been written on this subject (Gehring et al. 1976; Reitz et al. 1987; Nolan et al. 1995). A simplified example of a physiological model is depicted in figure 1.
Figure 1. A simplified pharmacokinetic model
Different Levels and Systems Can Be Adversely Affected
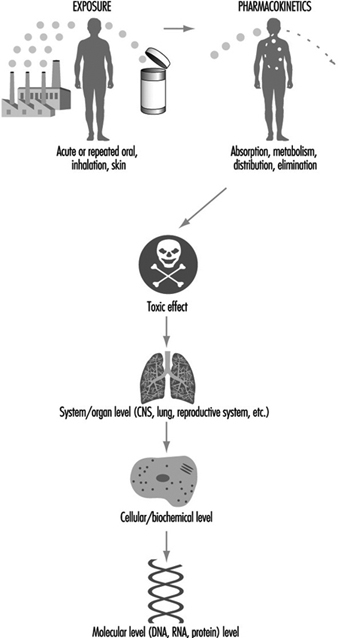
Toxicity can be described at different biological levels. Injury can be evaluated in the whole person (or animal), the organ system, the cell or the molecule. Organ systems include the immune, respiratory, cardiovascular, renal, endocrine, digestive, muscolo-skeletal, blood, reproductive and central nervous systems. Some key organs include the liver, kidney, lung, brain, skin, eyes, heart, testes or ovaries, and other major organs. At the cellular/biochemical level, adverse effects include interference with normal protein function, endocrine receptor function, metabolic energy inhibition, or xenobiotic (foreign substance) enzyme inhibition or induction. Adverse effects at the molecular level include alteration of the normal function of DNA-RNA transcription, of specific cytoplasmic and nuclear receptor binding, and of genes or gene products. Ultimately, dysfunction in a major organ system is likely caused by a molecular alteration in a particular target cell within that organ. However, it is not always possible to trace a mechanism back to a molecular origin of causation, nor is it necessary. Intervention and therapy can be designed without a complete understanding of the molecular target. However, knowledge about the specific mechanism of toxicity increases the predictive value and accuracy of extrapolation to other chemicals. Figure 2 is a diagrammatic representation of the various levels where interference of normal physiological processes can be detected. The arrows indicate that the consequences to an individual can be determined from top down (exposure, pharmaco- kinetics to system/organ toxicity) or from bottom up (molecular change, cellular/biochemical effect to system/organ toxicity).
Figure 2. Reresentation of mechanisms of toxicity
Examples of Mechanisms of Toxicity
Mechanisms of toxicity can be straightforward or very complex. Frequently, there is a difference among the type of toxicity, the mechanism of toxicity, and the level of effect, related to whether the adverse effects are due to a single, acute high dose (like an accidental poisoning), or a lower-dose repeated exposure (from occupational or environmental exposure). Classically, for testing purposes, an acute, single high dose is given by direct intubation into the stomach of a rodent or exposure to an atmosphere of a gas or vapour for two to four hours, whichever best resembles the human exposure. The animals are observed over a two-week period following exposure and then the major external and internal organs are examined for injury. Repeated-dose testing ranges from months to years. For rodent species, two years is considered a chronic (lifetime) study sufficient to evaluate toxicity and carcinogenicity, whereas for non-human primates, two years would be considered a subchronic (less than lifetime) study to evaluate repeated dose toxicity. Following exposure a complete examination of all tissues, organs and fluids is conducted to determine any adverse effects.
Acute Toxicity Mechanisms
The following examples are specific to high-dose, acute effects which can lead to death or severe incapacitation. However, in some cases, intervention will result in transient and fully reversible effects. The dose or severity of exposure will determine the result.
Simple asphyxiants. The mechanism of toxicity for inert gases and some other non-reactive substances is lack of oxygen (anoxia). These chemicals, which cause deprivation of oxygen to the central nervous system (CNS), are termed simple asphyxiants. If a person enters a closed space that contains nitrogen without sufficient oxygen, immediate oxygen depletion occurs in the brain and leads to unconsciousness and eventual death if the person is not rapidly removed. In extreme cases (near zero oxygen) unconsciousness can occur in a few seconds. Rescue depends on rapid removal to an oxygenated environment. Survival with irreversible brain damage can occur from delayed rescue, due to the death of neurons, which cannot regenerate.
Chemical asphyxiants. Carbon monoxide (CO) competes with oxygen for binding to haemoglobin (in red blood cells) and therefore deprives tissues of oxygen for energy metabolism; cellular death can result. Intervention includes removal from the source of CO and treatment with oxygen. The direct use of oxygen is based on the toxic action of CO. Another potent chemical asphyxiant is cyanide. The cyanide ion interferes with cellular metabolism and utilization of oxygen for energy. Treatment with sodium nitrite causes a change in haemoglobin in red blood cells to methaemoglobin. Methaemoglobin has a greater binding affinity to the cyanide ion than does the cellular target of cyanide. Consequently, the methaemoglobin binds the cyanide and keeps the cyanide away from the target cells. This forms the basis for antidotal therapy.
Central nervous system (CNS) depressants. Acute toxicity is characterized by sedation or unconsciousness for a number of materials like solvents which are not reactive or which are transformed to reactive intermediates. It is hypothesized that sedation/anaesthesia is due to an interaction of the solvent with the membranes of cells in the CNS, which impairs their ability to transmit electrical and chemical signals. While sedation may seem a mild form of toxicity and was the basis for development of the early anaesthetics, “the dose still makes the poison”. If sufficient dose is administered by ingestion or inhalation the animal can die due to respiratory arrest. If anaesthetic death does not occur, this type of toxicity is usually readily reversible when the subject is removed from the environment or the chemical is redistributed or eliminated from the body.
Skin effects. Adverse effects to the skin can range from irritation to corrosion, depending on the substance encountered. Strong acids and alkaline solutions are incompatible with living tissue and are corrosive, causing chemical burns and possible scarring. Scarring is due to death of the dermal, deep skin cells responsible for regeneration. Lower concentrations may just cause irritation of the first layer of skin.
Another specific toxic mechanism of skin is that of chemical sensitization. As an example, sensitization occurs when 2,4-dinitrochlorobenzene binds with natural proteins in the skin and the immune system recognizes the altered protein-bound complex as a foreign material. In responding to this foreign material, the immune system activates special cells to eliminate the foreign substance by release of mediators (cytokines) which cause a rash or dermatitis (see “Immunotoxicology”). This is the same reaction of the immune system when exposure to poison ivy occurs. Immune sensitization is very specific to the particular chemical and takes at least two exposures before a response is elicited. The first exposure sensitizes (sets up the cells to recognize the chemical), and subsequent exposures trigger the immune system response. Removal from contact and symptomatic therapy with steroid-containing anti-inflammatory creams are usually effective in treating sensitized individuals. In serious or refractory cases a systemic acting immunosuppresant like prednisone is used in conjunction with topical treatment.
Lung sensitization. An immune sensitization response is elicited by toluene diisocyanate (TDI), but the target site is the lungs. TDI over-exposure in susceptible individuals causes lung oedema (fluid build-up), bronchial constriction and impaired breathing. This is a serious condition and requires removing the individual from potential subsequent exposures. Treatment is primarily symptomatic. Skin and lung sensitization follow a dose response. Exceeding the level set for occupational exposure can cause adverse effects.
Eye effects. Injury to the eye ranges from reddening of the outer layer (swimming-pool redness) to cataract formation of the cornea to damage to the iris (coloured part of the eye). Eye irritation tests are conducted when it is believed serious injury will not occur. Many of the mechanisms causing skin corrosion can also cause injury to the eyes. Materials corrosive to the skin, like strong acids (pH less than 2) and alkali (pH greater than 11.5), are not tested in the eyes of animals because most will cause corrosion and blindness due to a mechanism similar to that which causes skin corrosion. In addition, surface active agents like detergents and surfactants can cause eye injury ranging from irritation to corrosion. A group of materials that requires caution is the positively charged (cationic) surfactants, which can cause burns, permanent opacity of the cornea and vascularization (formation of blood vessels). Another chemical, dinitrophenol, has a specific effect of cataract formation. This appears to be related to concentration of this chemical in the eye, which is an example of pharmacokinetic distributional specificity.
While the listing above is far from exhaustive, it is designed to give the reader an appreciation for various acute toxicity mechanisms.
Subchronic and Chronic Toxicity Mechanisms
When given as a single high dose, some chemicals do not have the same mechanism of toxicity as when given repeatedly as a lower but still toxic dose. When a single high dose is given, there is always the possibility of exceeding the person’s ability to detoxify or excrete the chemical, and this can lead to a different toxic response than when lower repetitive doses are given. Alcohol is a good example. High doses of alcohol lead to primary central nervous system effects, while lower repetitive doses result in liver injury.
Anticholinesterase inhibition. Most organophosphate pesticides, for example, have little mammalian toxicity until they are metabolically activated, primarily in the liver. The primary mechanism of action of organophosphates is the inhibition of acetylcholinesterase (AChE) in the brain and peripheral nervous system. AChE is the normal enzyme that terminates the stimulation of the neurotransmitter acetylcholine. Slight inhibition of AChE over an extended period has not been associated with adverse effects. At high levels of exposure, inability to terminate this neuronal stimulation results in overstimulation of the cholinergic nervous system. Cholinergic overstimulation ultimately results in a host of symptoms, including respiratory arrest, followed by death if not treated. The primary treatment is the administration of atropine, which blocks the effects of acetylcholine, and the administration of pralidoxime chloride, which reactivates the inhibited AChE. Therefore, both the cause and the treatment of organophosphate toxicity are addressed by understanding the biochemical basis of toxicity.
Metabolic activation. Many chemicals, including carbon tetrachloride, chloroform, acetylaminofluorene, nitrosamines, and paraquat are metabolically activated to free radicals or other reactive intermediates which inhibit and interfere with normal cellular function. At high levels of exposure this results in cell death (see “Cellular injury and cellular death”). While the specific interactions and cellular targets remain unknown, the organ systems which have the capability to activate these chemicals, like the liver, kidney and lung, are all potential targets for injury. Specifically, particular cells within an organ have a greater or lesser capacity to activate or detoxify these intermediates, and this capacity determines the intracellular susceptibility within an organ. Metabolism is one reason why an understanding of pharmacokinetics, which describes these types of transformations and the distribution and elimination of these intermediates, is important in recognizing the mechanism of action of these chemicals.
Cancer mechanisms. Cancer is a multiplicity of diseases, and while the understanding of certain types of cancer is increasing rapidly due to the many molecular biological techniques that have been developed since 1980, there is still much to learn. However, it is clear that cancer development is a multi-stage process, and critical genes are key to different types of cancer. Alterations in DNA (somatic mutations) in a number of these critical genes can cause increased susceptibility or cancerous lesions (see “Genetic toxic- ology”). Exposure to natural chemicals (in cooked foods like beef and fish) or synthetic chemicals (like benzidine, used as a dye) or physical agents (ultraviolet light from the sun, radon from soil, gamma radiation from medical procedures or industrial activity) are all contributors to somatic gene mutations. However, there are natural and synthetic substances (such as anti-oxidants) and DNA repair processes which are protective and maintain homeostasis. It is clear that genetics is an important factor in cancer, since genetic disease syndromes such as xeroderma pigmentosum, where there is a lack of normal DNA repair, dramatically increase susceptibility to skin cancer from exposure to ultraviolet light from the sun.
Reproductive mechanisms. Similar to cancer, many mechanisms of reproductive and/or developmental toxicity are known, but much is to be learned. It is known that certain viruses (such as rubella), bacterial infections and drugs (such as thalidomide and vitamin A) will adversely affect development. Recently, work by Khera (1991), reviewed by Carney (1994), show good evidence that the abnormal developmental effects in animal tests with ethylene glycol are attributable to maternal metabolic acidic metabolites. This occurs when ethylene glycol is metabolized to acid metabolites including glycolic and oxalic acid. The subsequent effects on the placenta and foetus appear to be due to this metabolic toxication process.
Conclusion
The intent of this article is to give a perspective on several known mechanisms of toxicity and the need for future study. It is important to understand that mechanistic knowledge is not absolutely necessary to protect human or environmental health. This knowledge will enhance the professional’s ability to better predict and manage toxicity. The actual techniques used in elucidating any particular mechanism depend upon the collective knowledge of the scientists and the thinking of those who make decisions regarding human health.
Cellular Injury and Cellular Death
Virtually all of medicine is devoted to either preventing cell death, in diseases such as myocardial infarction, stroke, trauma and shock, or causing it, as in the case of infectious diseases and cancer. It is, therefore, essential to understand the nature and mechanisms involved. Cell death has been classified as “accidental”, that is, caused by toxic agents, ischaemia and so on, or “programmed”, as occurs during embryological development, including formation of digits, and resorption of the tadpole tail.
Cell injury and cell death are, therefore, important both in physiology and in pathophysiology. Physiological cell death is extremely important during embryogenesis and embryonic development. The study of cell death during development has led to important and new information on the molecular genetics involved, especially through the study of development in invertebrate animals. In these animals, the precise location and the significance of cells that are destined to undergo cell death have been carefully studied and, with the use of classic mutagenesis techniques, several involved genes have now been identified. In adult organs, the balance between cell death and cell proliferation controls organ size. In some organs, such as the skin and the intestine, there is a continual turnover of cells. In the skin, for example, cells differentiate as they reach the surface, and finally undergo terminal differentiation and cell death as keratinization proceeds with the formation of crosslinked envelopes.
Many classes of toxic chemicals are capable of inducing acute cell injury followed by death. These include anoxia and ischaemia and their chemical analogues such as potassium cyanide; chemical carcinogens, which form electrophiles that covalently bind to proteins in nucleic acids; oxidant chemicals, resulting in free radical formation and oxidant injury; activation of complement; and a variety of calcium ionophores. Cell death is also an important component of chemical carcinogenesis; many complete chemical carcinogens, at carcinogenic doses, produce acute necrosis and inflammation followed by regeneration and preneoplasia.
Definitions
Cell injury
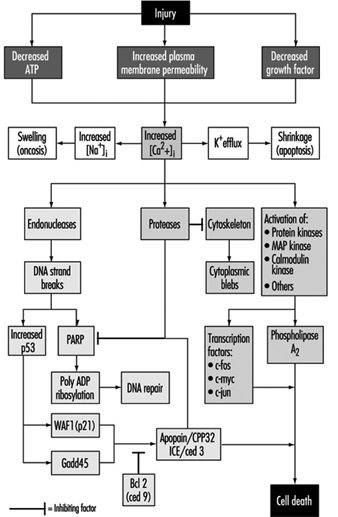
Cell injury is defined as an event or stimulus, such as a toxic chemical, that perturbs the normal homeostasis of the cell, thus causing a number of events to occur (figure 1). The principal targets of lethal injury illustrated are inhibition of ATP synthesis, disruption of plasma membrane integrity or withdrawal of essential growth factors.
Lethal injuries result in the death of a cell after a variable period of time, depending on temperature, cell type and the stimulus; or they can be sublethal or chronic—that is, the injury results in an altered homeostatic state which, though abnormal, does not result in cell death (Trump and Arstila 1971; Trump and Berezesky 1992; Trump and Berezesky 1995; Trump, Berezesky and Osornio-Vargas 1981). In the case of a lethal injury, there is a phase prior to the time of cell death
during this time, the cell will recover; however, after a particular point in time (the “point of no return” or point of cell death), the removal of the injury does not result in recovery but instead the cell undergoes degradation and hydrolysis, ultimately reaching physical-chemical equilibrium with the environment. This is the phase known as necrosis. During the prelethal phase, several principal types of change occur, depending on the cell and the type of injury. These are known as apoptosis and oncosis.
Apoptosis
Apoptosis is derived from the Greek words apo, meaning away from, and ptosis, meaning to fall. The term falling away from is derived from the fact that, during this type of prelethal change, the cells shrink and undergo marked blebbing at the periphery. The blebs then detach and float away. Apoptosis occurs in a variety of cell types following various types of toxic injury (Wyllie, Kerr and Currie 1980). It is especially prominent in lymphocytes, where it is the predominant mechanism for turnover of lymphocyte clones. The resulting fragments result in the basophilic bodies seen within macrophages in lymph nodes. In other organs, apoptosis typically occurs in single cells which are rapidly cleared away before and following death by phagocytosis of the fragments by adjacent parenchymal cells or by macrophages. Apoptosis occurring in single cells with subsequent phagocytosis typically does not result in inflammation. Prior to death, apoptotic cells show a very dense cytosol with normal or condensed mitochondria. The endoplasmic reticulum (ER) is normal or only slightly dilated. The nuclear chromatin is markedly clumped along the nuclear envelope and around the nucleolus. The nuclear contour is also irregular and nuclear fragmentation occurs. The chromatin conden- sation is associated with DNA fragmentation which, in many instances, occurs between nucleosomes, giving a characteristic ladder appearance on electrophoresis.
In apoptosis, increased [Ca2+]i may stimulate K+ efflux resulting in cell shrinkage, which probably requires ATP. Injuries that totally inhibit ATP synthesis, therefore, are more likely to result in apoptosis. A sustained increase of [Ca2+]i has a number of deleterious effects including activation of proteases, endonucleases, and phospholipases. Endonuclease activation results in single and double DNA strand breaks which, in turn, stimulate increased levels of p53 and in poly-ADP ribosylation, and of nuclear proteins which are essential in DNA repair. Activation of proteases modifies a number of substrates including actin and related proteins leading to bleb formation. Another important substrate is poly(ADP-ribose) polymerase (PARP), which inhibits DNA repair. Increased [Ca2+]i is also associated with activation of a number of protein kinases, such as MAP kinase, calmodulin kinase and others. Such kinases are involved in activation of transcription factors which initiate transcription of immediate-early genes, for example, c-fos, c-jun and c-myc, and in activation of phospholipase A2 which results in permeabilization of the plasma membrane and of intracellular membranes such as the inner membrane of mitochondria.
Oncosis
Oncosis, derived from the Greek word onkos, to swell, is so named because in this type of prelethal change the cell begins to swell almost immediately following the injury (Majno and Joris 1995). The reason for the swelling is an increase in cations in the water within the cell. The principal cation responsible is sodium, which is normally regulated to maintain cell volume. However, in the absence of ATP or if Na-ATPase of the plasmalemma is inhibited, volume control is lost because of intracellular protein, and sodium in the water continuing to increase. Among the early events in oncosis are, therefore, increased [Na+]i which leads to cellular swelling and increased [Ca2+]i resulting either from influx from the extracellular space or release from intracellular stores. This results in swelling of the cytosol, swelling of the endoplasmic reticulum and Golgi apparatus, and the formation of watery blebs around the cell surface. The mitochondria initially undergo condensation, but later they too show high-amplitude swelling because of damage to the inner mitochondrial membrane. In this type of prelethal change, the chromatin undergoes condensation and ultimately degradation; however, the characteristic ladder pattern of apoptosis is not seen.
Necrosis
Necrosis refers to the series of changes that occur following cell death when the cell is converted to debris which is typically removed by the inflammatory response. Two types can be distinguished: oncotic necrosis and apoptotic necrosis. Oncotic necrosis typically occurs in large zones, for example, in a myocardial infarct or regionally in an organ after chemical toxicity, such as the renal proximal tubule following administration of HgCl2. Broad zones of an organ are involved and the necrotic cells rapidly incite an inflammatory reaction, first acute and then chronic. In the event that the organism survives, in many organs necrosis is followed by clearing away of the dead cells and regeneration, for example, in the liver or kidney following chemical toxicity. In contrast, apoptotic necrosis typically occurs on a single cell basis and the necrotic debris is formed within the phagocytes of macrophages or adjacent parenchymal cells. The earliest characteristics of necrotic cells include interruptions in plasma membrane continuity and the appearance of flocculent densities, representing denatured proteins within the mitochondrial matrix. In some forms of injury that do not initially interfere with mitochondrial calcium accumulation, calcium phosphate deposits can be seen within the mitochondria. Other membrane systems are similarly fragmenting, such as the ER, the lysosomes and the Golgi apparatus. Ultimately, the nuclear chromatin undergoes lysis, resulting from attack by lysosomal hydrolases. Following cell death, lysosomal hydrolases play an important part in clearing away debris with cathepsins, nucleolases and lipases since these have an acid pH optimum and can survive the low pH of necrotic cells while other cellular enzymes are denatured and inactivated.
Mechanisms
Initial stimulus
In the case of lethal injuries, the most common initial interactions resulting in injury leading to cell death are interference with energy metabolism, such as anoxia, ischaemia or inhibitors of respiration, and glycolysis such as potassium cyanide, carbon monoxide, iodo-acetate, and so on. As mentioned above, high doses of compounds that inhibit energy metabolism typically result in oncosis. The other common type of initial injury resulting in acute cell death is modification of the function of the plasma membrane (Trump and Arstila 1971; Trump, Berezesky and Osornio-Vargas 1981). This can either be direct damage and permeabilization, as in the case of trauma or activation of the C5b-C9 complex of complement, mechanical damage to the cell membrane or inhibition of the sodium-potassium (Na+-K+) pump with glycosides such as ouabain. Calcium ionophores such as ionomycin or A23187, which rapidly carry [Ca2+] down the gradient into the cell, also cause acute lethal injury. In some cases, the pattern in the prelethal change is apoptosis; in others, it is oncosis.
Signalling pathways
With many types of injury, mitochondrial respiration and oxidative phosphorylation are rapidly affected. In some cells, this stimulates anaerobic glycolysis, which is capable of maintaining ATP, but with many injuries this is inhibited. The lack of ATP results in failure to energize a number of important homeostatic processes, in particular, control of intracellular ion homeostasis (Trump and Berezesky 1992; Trump, Berezesky and Osornio-Vargas 1981). This results in rapid increases of [Ca2+]i, and increased [Na+] and [Cl-] results in cell swelling. Increases in [Ca2+]i result in the activation of a number of other signalling mechanisms discussed below, including a series of kinases, which can result in increased immediate early gene transcription. Increased [Ca2+]i also modifies cytoskeletal function, in part resulting in bleb formation and in the activation of endonucleases, proteases and phospholipases. These seem to trigger many of the important effects discussed above, such as membrane damage through protease and lipase activation, direct degradation of DNA from endonuclease activation, and activation of kinases such as MAP kinase and calmodulin kinase, which act as transcription factors.
Through extensive work on development in the invertebrate C. elegans and Drosophila, as well as human and animal cells, a series of pro-death genes have been identified. Some of these invertebrate genes have been found to have mammalian counterparts. For example, the ced-3 gene, which is essential for programmed cell death in C. elegans, has protease activity and a strong homology with the mammalian interleukin converting enzyme (ICE). A closely related gene called apopain or prICE has recently been identified with even closer homology (Nicholson et al. 1995). In Drosophila, the reaper gene seems to be involved in a signal that leads to programmed cell death. Other pro-death genes include the Fas membrane protein and the important tumour-suppressor gene, p53, which is widely conserved. p53 is induced at the protein level following DNA damage and when phosphorylated acts as a transcription factor for other genes such as gadd45 and waf-1, which are involved in cell death signalling. Other immediate early genes such as c-fos, c-jun, and c-myc also seem to be involved in some systems.
At the same time, there are anti-death genes which appear to counteract the pro-death genes. The first of these to be identified was ced-9 from C. elegans, which is homologous to bcl-2 in humans. These genes act in an as yet unknown way to prevent cell killing by either genetic or chemical toxins. Some recent evidence indicates that bcl-2 may act as an antioxidant. Currently, there is much effort underway to develop an understanding of the genes involved and to develop ways to activate or inhibit these genes, depending on the situation.
Genetic Toxicology
Genetic toxicology, by definition, is the study of how chemical or physical agents affect the intricate process of heredity. Genotoxic chemicals are defined as compounds that are capable of modifying the hereditary material of living cells. The probability that a particular chemical will cause genetic damage inevitably depends on several variables, including the organism’s level of exposure to the chemical, the distribution and retention of the chemical once it enters the body, the efficiency of metabolic activation and/or detoxification systems in target tissues, and the reactivity of the chemical or its metabolites with critical macromolecules within cells. The probability that genetic damage will cause disease ultimately depends on the nature of the damage, the cell’s ability to repair or amplify genetic damage, the opportunity for expressing whatever alteration has been induced, and the ability of the body to recognize and suppress the multiplication of aberrant cells.
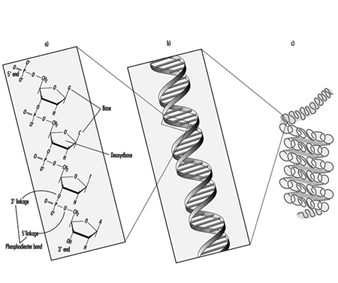
In higher organisms, hereditary information is organized in chromosomes. Chromosomes consist of tightly condensed strands of protein-associated DNA. Within a single chromosome, each DNA molecule exists as a pair of long, unbranched chains of nucleotide subunits linked together by phosphodiester bonds that join the 5 carbon of one deoxyribose moiety to the 3 carbon of the next (figure 1). In addition, one of four different nucleotide bases (adenine, cytosine, guanine or thymine) is attached to each deoxyribose subunit like beads on a string. Three-dimensionally, each pair of DNA strands forms a double helix with all of the bases oriented toward the inside of the spiral. Within the helix, each base is associated with its complementary base on the opposite DNA strand; hydrogen bonding dictates strong, noncovalent pairing of adenine with thymine and guanine with cytosine (figure 1). Since the sequence of nucleotide bases is complementary throughout the entire length of the duplex DNA molecule, both strands carry essentially the same genetic information. In fact, during DNA replication each strand serves as a template for the production of a new partner strand.
Figure 1. The (a) primary, (b) secondary and (c) tertiary organization of human hereditary information
 Using RNA and an array of different proteins, the cell ultimately deciphers the information encoded by the linear sequence of bases within specific regions of DNA (genes) and produces proteins that are essential for basic cell survival as well as normal growth and differentiation. In essence, the nucleotides function like a biological alphabet which is used to code for amino acids, the building blocks of proteins.
Using RNA and an array of different proteins, the cell ultimately deciphers the information encoded by the linear sequence of bases within specific regions of DNA (genes) and produces proteins that are essential for basic cell survival as well as normal growth and differentiation. In essence, the nucleotides function like a biological alphabet which is used to code for amino acids, the building blocks of proteins.
When incorrect nucleotides are inserted or nucleotides are lost, or when unnecessary nucleotides are added during DNA synthesis, the mistake is called a mutation. It has been estimated that less than one mutation occurs for every 109 nucleotides incorporated during the normal replication of cells. Although mutations are not necessarily harmful, alterations causing inactivation or overexpression of important genes can result in a variety of disorders, including cancer, hereditary disease, developmental abnormalities, infertility and embryonic or perinatal death. Very rarely, a mutation can lead to enhanced survival; such occurrences are the basis of natural selection.
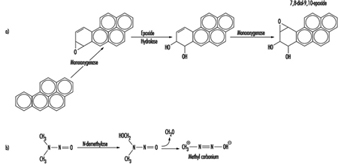
Although some chemicals react directly with DNA, most require metabolic activation. In the latter case, electrophilic intermediates such as epoxides or carbonium ions are ultimately responsible for inducing lesions at a variety of nucleophilic sites within the genetic material (figure 2). In other instances, genotoxicity is mediated by by-products of compound interaction with intracellular lipids, proteins, or oxygen.
Figure 2. Bioactivation of: a) benzo(a)pyrene; and b) N-nitrosodimethylamine
Because of their relative abundance in cells, proteins are the most frequent target of toxicant interaction. However, modification of DNA is of greater concern due to the central role of this molecule in regulating growth and differentiation through multiple generations of cells.
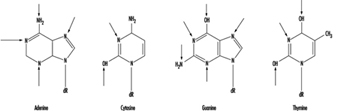
At the molecular level, electrophilic compounds tend to attack oxygen and nitrogen in DNA. The sites that are most prone to modification are illustrated in figure 3. Although oxygens within phosphate groups in the DNA backbone are also targets for chemical modification, damage to bases is thought to be biologically more relevant since these groups are considered to be the primary informational elements in the DNA molecule.
Figure 3. Primary sites of chemically-induced DNA damage
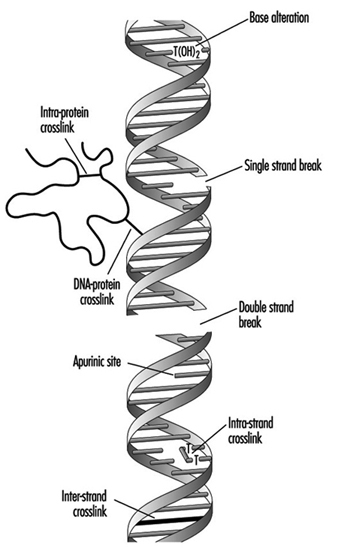
Compounds that contain one electrophilic moiety typically exert genotoxicity by producing mono-adducts in DNA. Similarly, compounds that contain two or more reactive moieties can react with two different nucleophilic centres and thereby produce intra- or inter-molecular crosslinks in genetic material (figure 4). Interstrand DNA-DNA and DNA-protein crosslinks can be particularly cytotoxic since they can form complete blocks to DNA replication. For obvious reasons, the death of a cell eliminates the possibility that it will be mutated or neoplastically transformed. Genotoxic agents can also act by inducing breaks in the phosphodiester backbone, or between bases and sugars (producing abasic sites) in DNA. Such breaks may be a direct result of chemical reactivity at the damage site, or may occur during the repair of one of the aforementioned types of DNA lesion.
Figure 4. Various types of damage to the protein-DNA complex
Over the past thirty to forty years, a variety of techniques have been developed to monitor the type of genetic damage induced by various chemicals. Such assays are described in detail elsewhere in this chapter and Encyclopaedia.
Misreplication of “microlesions” such as mono-adducts, abasic sites or single-strand breaks may ultimately result in nucleotide base-pair substitutions, or the insertion or deletion of short polynucleotide fragments in chromosomal DNA. In contrast, “macrolesions,” such as bulky adducts, crosslinks, or double-strand breaks may trigger the gain, loss or rearrangement of relatively large pieces of chromosomes. In any case, the consequences can be devastating to the organism since any one of these events can lead to cell death, loss of function or malignant transformation of cells. Exactly how DNA damage causes cancer is largely unknown. It is currently believed the process may involve inappropriate activation of proto-oncogenes such as myc and ras, and/or inactivation of recently identified tumour suppressor genes such as p53. Abnormal expression of either type of gene abrogates normal cellular mechanisms for controlling cell proliferation and/or differentiation.
The preponderance of experimental evidence indicates that the development of cancer following exposure to electrophilic compounds is a relatively rare event. This can be explained, in part, by the cell’s intrinsic ability to recognize and repair damaged DNA or the failure of cells with damaged DNA to survive. During repair, the damaged base, nucleotide or short stretch of nucleotides surrounding the damage site is removed and (using the opposite strand as a template) a new piece of DNA is synthesized and spliced into place. To be effective, DNA repair must occur with great accuracy prior to cell division, before opportunities for the propagation of mutation.
Clinical studies have shown that people with inherited defects in the ability to repair damaged DNA frequently develop cancer and/or developmental abnormalities at an early age (table 1). Such examples provide strong evidence linking accumulation of DNA damage to human disease. Similarly, agents that promote cell proliferation (such as tetradecanoylphorbol acetate) often enhance carcinogenesis. For these compounds, the increased likelihood of neoplastic transformation may be a direct consequence of a decrease in the time available for the cell to carry out adequate DNA repair.
Table 1. Hereditary, cancer-prone disorders that appear to involve defects in DNA repair
| Syndrome | Symptoms | Cellular phenotype |
| Ataxia telangiectasia | Neurological deterioration Immunodeficiency High incidence of lymphoma |
Hypersensitivity to ionizing radiation and certain alkylating agents. Dysregulated replication of damaged DNA (may indicate shortened time for DNA repair) |
| Bloom’s syndrome | Developmental abnormalities Lesions on exposed skin High incidence of tumours of the immune system and gastrointestinal tract |
High frequency of chromosomal aberrations Defective ligation of breaks associated with DNA repair |
| Fanconi’s anaemia | Growth retardation High incidence of leukaemia |
Hypersensitivity to crosslinking agents High frequency of chromosomal aberrations Defective repair of crosslinks in DNA |
| Hereditary nonpolyposis colon cancer | High incidence of colon cancer | Defect in DNA mismatch repair (when insertion of wrong nucleotide occurs during replication) |
| Xeroderma pigmentosum | High incidence of epithelioma on exposed areas of skin Neurological impairment (in many cases) |
Hypersensitivity to UV light and many chemical carcinogens Defects in excision repair and/or replication of damaged DNA |
The earliest theories on how chemicals interact with DNA can be traced back to studies conducted during the development of mustard gas for use in warfare. Further understanding grew out of efforts to identify anticancer agents that would selectively arrest the replication of rapidly dividing tumour cells. Increased public concern over hazards in our environment has prompted additional research into the mechanisms and consequences of chemical interaction with the genetic material. Examples of various types of chemicals which exert genotoxicity are presented in table 2.
Table 2. Examples of chemicals that exhibit genotoxicity in human cells
| Class of chemical | Example | Source of exposure | Probable genotoxic lesion |
| Aflatoxins | Aflatoxin B1 | Contaminated food | Bulky DNA adducts |
| Aromatic amines | 2-Acetylaminofluorene | Environmental | Bulky DNA adducts |
| Aziridine quinones | Mitomycin C | Cancer chemotherapy | Mono-adducts, interstrand crosslinks and single-strand breaks in DNA. |
| Chlorinated hydrocarbons | Vinyl chloride | Environmental | Mono-adducts in DNA |
| Metals and metal compounds | Cisplatin | Cancer chemotherapy | Both intra- and inter-strand crosslinks in DNA |
| Nickel compounds | Environmental | Mono-adducts and single-strand breaks in DNA | |
| Nitrogen mustards | Cyclophosphamide | Cancer chemotherapy | Mono-adducts and interstrand crosslinks in DNA |
| Nitrosamines | N-Nitrosodimethylamine | Contaminated food | Mono-adducts in DNA |
| Polycyclic aromatic hydrocarbons | Benzo(a)pyrene | Environmental | Bulky DNA adducts |
Immunotoxicology
The functions of the immune system are to protect the body from invading infectious agents and to provide immune surveillance against arising tumour cells. It has a first line of defence that is non-specific and that can initiate effector reactions itself, and an acquired specific branch, in which lymphocytes and antibodies carry the specificity of recognition and subsequent reactivity towards the antigen.
Immunotoxicology has been defined as “the discipline concerned with the study of the events that can lead to undesired effects as a result of interaction of xenobiotics with the immune system. These undesired events may result as a consequence of (1) a direct and/or indirect effect of the xenobiotic (and/or its biotransformation product) on the immune system, or (2) an immunologically based host response to the compound and/or its metabolite(s), or host antigens modified by the compound or its metabolites” (Berlin et al. 1987).
When the immune system acts as a passive target of chemical insults, the result can be decreased resistance to infection and certain forms of neoplasia, or immune disregulation/stimulation that can exacerbate allergy or auto-immunity. In the case that the immune system responds to the antigenic specificity of the xenobiotic or host antigen modified by the compound, toxicity can become manifest as allergies or autoimmune diseases.
Animal models to investigate chemical-induced immune suppression have been developed, and a number of these methods are validated (Burleson, Munson, and Dean 1995; IPCS 1996). For testing purposes, a tiered approach is followed to make an adequate selection from the overwhelming number of assays available. Generally, the objective of the first tier is to identify potential immunotoxicants. If potential immunotoxicity is identified, a second tier of testing is performed to confirm and characterize further the changes observed. Third-tier investigations include special studies on the mechanism of action of the compound. Several xenobiotics have been identified as immunotoxicants causing immunosuppression in such studies with laboratory animals.
The database on immune function disturbances in humans by environmental chemicals is limited (Descotes 1986; NRC Subcommittee on Immunotoxicology 1992). The use of markers of immunotoxicity has received little attention in clinical and epidemiological studies to investigate the effect of these chemicals on human health. Such studies have not been performed frequently, and their interpretation often does not permit unequivocal conclusions to be drawn, due for instance to the uncontrolled nature of exposure. Therefore, at present, immunotoxicity assessment in rodents, with subsequent extrapolation to man, forms the basis of decisions regarding hazard and risk.
Hypersensitivity reactions, notably allergic asthma and contact dermatitis, are important occupational health problems in industrialized countries (Vos, Younes and Smith 1995). The phenomenon of contact sensitization was investigated first in the guinea pig (Andersen and Maibach 1985). Until recently this has been the species of choice for predictive testing. Many guinea pig test methods are available, the most frequently employed being the guinea pig maximization test and the occluded patch test of Buehler. Guinea pig tests and newer approaches developed in mice, such as ear swelling tests and the local lymph node assay, provide the toxicologist with the tools to assess skin sensitization hazard. The situation with respect to sensitization of the respiratory tract is very different. There are, as yet, no well-validated or widely accepted methods available for the identification of chemical respiratory allergens although progress in the development of animal models for the investigation of chemical respiratory allergy has been achieved in the guinea pig and mouse.
Human data show that chemical agents, in particular drugs, can cause autoimmune diseases (Kammüller, Bloksma and Seinen 1989). There are a number of experimental animal models of human autoimmune diseases. Such comprise both spontaneous pathology (for example systemic lupus erythematosus in New Zealand Black mice) and autoimmune phenomena induced by experimental immunization with a cross-reactive autoantigen (for example the H37Ra adjuvant induced arthritis in Lewis strain rats). These models are applied in the preclinical evaluation of immunosuppressive drugs. Very few studies have addressed the potential of these models for assessment of whether a xenobiotic exacerbates induced or congenital autoimmunity. Animal models that are suitable to investigate the ability of chemicals to induce autoimmune diseases are virtually lacking. One model that is used to a limited extent is the popliteal lymph node assay in mice. Like the situation in humans, genetic factors play a crucial role in the development of autoimmune disease (AD) in laboratory animals, which will limit the predictive value of such tests.
The Immune System
The major function of the immune system is defence against bacteria, viruses, parasites, fungi and neoplastic cells. This is achieved by the actions of various cell types and their soluble mediators in a finely tuned concert. The host defence can be roughly divided into non-specific or innate resistance and specific or acquired immunity mediated by lymphocytes (Roitt, Brostoff and Male 1989).
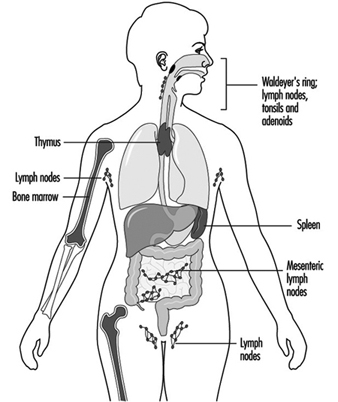
Components of the immune system are present throughout the body (Jones et al. 1990). The lymphocyte compartment is found within lymphoid organs (figure 1). The bone marrow and thymus are classified as primary or central lymphoid organs; the secondary or peripheral lymphoid organs include lymph nodes, spleen and lymphoid tissue along secretory surfaces such as the gastrointestinal and respiratory tracts, the so-called mucosa-associated lymphoid tissue (MALT). About half of the body’s lymphocytes are located at any one time in MALT. In addition the skin is an important organ for the induction of immune responses to antigens present on the skin. Important in this process are epidermal Langerhans cells that have an antigen-presenting function.
Figure 1. Primary and secondary lymphoid organs and tissues
Phagocytic cells of the monocyte/macrophage lineage, called the mononuclear phagocyte system (MPS), occur in lymphoid organs and also at extranodal sites; the extranodal phagocytes include Kupffer cells in the liver, alveolar macrophages in the lung, mesangial macrophages in the kidney and glial cells in the brain. Polymorphonuclear leukocytes (PMNs) are present mainly in blood and bone marrow, but accumulate at sites of inflammation.
Non-specific defence
A first line of defence to micro-organisms is executed by a physical and chemical barrier, such as at the skin, the respiratory tract and the alimentary tract. This barrier is helped by non-specific protective mechanisms including phagocytic cells, such as macrophages and polymorphonuclear leukocytes, which are able to kill pathogens, and natural killer cells, which can lyse tumour cells and virus-infected cells. The complement system and certain microbial inhibitors (e.g., lysozyme) also take part in the non-specific response.
Specific immunity
After initial contact of the host with the pathogen, specific immune responses are induced. The hallmark of this second line of defence is specific recognition of determinants, so-called antigens or epitopes, of the pathogens by receptors on the cell surface of B- and T-lymphocytes. Following interaction with the specific antigen, the receptor-bearing cell is stimulated to undergo proliferation and differentiation, producing a clone of progeny cells that are specific for the eliciting antigen. The specific immune responses help the non-specific defence presented to the pathogens by stimulating the efficacy of the non-specific responses. A fundamental characteristic of specific immunity is that memory develops. Secondary contact with the same antigen provokes a faster and more vigorous but well-regulated response.
The genome does not have the capacity to carry the codes of an array of antigen receptors sufficient to recognize the number of antigens that can be encountered. The repertoire of specificity develops by a process of gene rearrangements. This is a random process, during which various specificities are brought about. This includes specificities for self components, which are undesirable. A selection process that takes place in the thymus (T cells), or bone marrow (B cells) operates to delete these undesirable specificities.
Normal immune effector function and homeostatic regulation of the immune response is dependent upon a variety of soluble products, known collectively as cytokines, which are synthesized and secreted by lymphocytes and by other cell types. Cytokines have pleiotropic effects on immune and inflammatory responses. Cooperation between different cell populations is required for the immune response—the regulation of antibody responses, the accumulation of immune cells and molecules at inflammatory sites, the initiation of acute phase responses, the control of macrophage cytotoxic function and many other processes central to host resistance. These are influenced by, and in many cases are dependent upon, cytokines acting individually or in concert.
Two arms of specific immunity are recognized—humoral immunity and cell-mediated or cellular immunity:
Humoral immunity. In the humoral arm B-lymphocytes are stimulated following recognition of antigen by cell-surface receptors. Antigen receptors on B-lymphocytes are immunoglobulins (Ig). Mature B cells (plasma cells) start the production of antigen-specific immunoglobulins that act as antibodies in serum or along mucosal surfaces. There are five major classes of immunoglobulins: (1) IgM, pentameric Ig with optimal agglutinating capacity, which is first produced after antigenic stimulation; (2) IgG, the main Ig in circulation, which can pass the placenta; (3) IgA, secretory Ig for the protection of mucosal surfaces; (4) IgE, Ig fixing to mast cells or basophilic granulocytes involved in immediate hypersensitivity reactions and (5) IgD, whose major function is as a receptor on B-lymphocytes.
Cell-mediated immunity. The cellular arm of the specific immune system is mediated by T-lymphocytes. These cells also have antigen receptors on their membranes. They recognize antigen if presented by antigen presenting cells in the context of histocompatibility antigens. Hence, these cells have a restriction in addition to the antigen specificity. T cells function as helper cells for various (including humoral) immune responses, mediate recruitment of inflammatory cells, and can, as cytotoxic T cells, kill target cells after antigen-specific recognition.
Mechanisms of Immunotoxicity
Immunosuppression
Effective host resistance is dependent upon the functional integrity of the immune system, which in turn requires that the component cells and molecules which orchestrate immune responses are available in sufficient numbers and in an operational form. Congenital immunodeficiencies in humans are often characterized by defects in certain stem cell lines, resulting in impaired or absent production of immune cells. By analogy with congenital and acquired human immunodeficiency diseases, chemical-induced immunosuppression may result simply from a reduced number of functional cells (IPCS 1996). The absence, or reduced numbers, of lymphocytes may have more or less profound effects on immune status. Some immunodeficiency states and severe immunosuppression, as can occur in transplantation or cytostatic therapy, have been associated in particular with increased incidences of opportunistic infections and of certain neoplastic diseases. The infections can be bacterial, viral, fungal or protozoan, and the predominant type of infection depends on the associated immunodeficiency. Exposure to immunosuppressive environmental chemicals may be expected to result in more subtle forms of immunosuppression, which may be difficult to detect. These may lead, for example, to an increased incidence of infections such as influenza or the common cold.
In view of the complexity of the immune system, with the wide variety of cells, mediators and functions that form a complicated and interactive network, immunotoxic compounds have numerous opportunities to exert an effect. Although the nature of the initial lesions induced by many immunotoxic chemicals have not yet been elucidated, there is increasing information available, mostly derived from studies in laboratory animals, regarding the immunobiological changes which result in depression of immune function (Dean et al. 1994). Toxic effects might occur at the following critical functions (and some examples are given of immunotoxic compounds affecting these functions):
- development and expansion of different stem cell populations (benzene exerts immunotoxic effects at the stem cell level, causing lymphocytopenia)
- proliferation of various lymphoid and myeloid cells as well as supportive tissues in which these cells mature and function (immunotoxic organotin compounds suppress the proliferative activity of lymphocytes in the thymic cortex through direct cytotoxicity; the thymotoxic action of 2,3,7,8-tetrachloro-dibenzo-p-dioxin (TCDD) and related compounds is likely due to an impaired function of thymic epithelial cells, rather than to direct toxicity for thymocytes)
- antigen uptake, processing and presentation by macrophages and other antigen-presenting cells (one of the targets of 7,12-dimethylbenz(a)anthracene (DMBA) and of lead is antigen presentation by macrophages; a target of ultraviolet radiation is the antigen-presenting Langerhans cell)
- regulatory function of T-helper and T-suppressor cells (T-helper cell function is impaired by organotins, aldicarb, polychlorinated biphenyls (PCBs), TCDD and DMBA; T-suppressor cell function is reduced by low-dose cyclophosphamide treatment)
- production of various cytokines or interleukins (benzo(a)pyrene (BP) suppresses interleukin-1 production; ultraviolet radiation alters production of cytokines by keratinocytes)
- synthesis of various classes of immunoglobulins IgM and IgG is suppressed following PCB and tributyltin oxide (TBT) treatment, and increased after hexachlorobenzene (HCB) exposure).
- complement regulation and activation (affected by TCDD)
- cytotoxic T cell function (3-methylcholanthrene (3-MC), DMBA, and TCDD suppress cytotoxic T cell activity)
- natural killer (NK) cell function (pulmonary NK activity is suppressed by ozone; splenic NK activity is impaired by nickel)
- macrophage and polymorphonuclear leukocyte chemotaxis and cytotoxic functions (ozone and nitrogen dioxide impair the phagocytic activity of alveolar macrophages).
Allergy
Allergy may be defined as the adverse health effects which result from the induction and elicitation of specific immune responses. When hypersensitivity reactions occur without involvement of the immune system the term pseudo-allergy is used. In the context of immunotoxicology, allergy results from a specific immune response to chemicals and drugs that are of interest. The ability of a chemical to sensitize individuals is generally related to its ability to bind covalently to body proteins. Allergic reactions may take a variety of forms and these differ with respect to both the underlying immunological mechanisms and the speed of the reaction. Four major types of allergic reactions have been recognized: Type I hypersensitivity reactions, which are effectuated by IgE antibody and where symptoms are manifest within minutes of exposure of the sensitized individual. Type II hypersensitivity reactions result from the damage or destruction of host cells by antibody. In this case symptoms become apparent within hours. Type III hypersensitivity, or Arthus, reactions are also antibody mediated, but against soluble antigen, and result from the local or systemic action of immune complexes. Type IV, or delayed-type hypersensitivity, reactions are effected by T-lymphocytes and normally symptoms develop 24to 48hours following exposure of the sensitized individual.
The two types of chemical allergy of greatest relevance to occupational health are contact sensitivity or skin allergy and allergy of the respiratory tract.
Contact hypersensitivity. A large number of chemicals are able to cause skin sensitization. Following topical exposure of a susceptible individual to a chemical allergen, a T-lymphocyte response is induced in the draining lymph nodes. In the skin the allergen interacts directly or indirectly with epidermal Langerhans cells, which transport the chemical to the lymph nodes and present it in an immunogenic form to responsive T-lymphocytes. Allergen- activated T-lymphocytes proliferate, resulting in clonal expansion. The individual is now sensitized and will respond to a second dermal exposure to the same chemical with a more aggressive immune response, resulting in allergic contact dermatitis. The cutaneous inflammatory reaction which characterizes allergic contact dermatitis is secondary to the recognition of the allergen in the skin by specific T-lymphocytes. These lymphocytes become activated, release cytokines and cause the local accumulation of other mononuclear leukocytes. Symptoms develop some 24 to 48 hours following exposure of the sensitized individual, and allergic contact dermatitis therefore represents a form of delayed-type hypersensitivity. Common causes of allergic contact dermatitis include organic chemicals (such as 2,4-dinitrochlorobenzene), metals (such as nickel and chromium) and plant products (such as urushiol from poison ivy).
Respiratory hypersensitivity. Respiratory hypersensitivity is usually considered to be a Type I hypersensitivity reaction. However, late phase reactions and the more chronic symptoms associated with asthma may involve cell-mediated (Type IV) immune processes. The acute symptoms associated with respiratory allergy are effected by IgE antibody, the production of which is provoked following exposure of the susceptible individual to the inducing chemical allergen. The IgE antibody distributes systemically and binds, via membrane receptors, to mast cells which are found in vascularized tissues, including the respiratory tract. Following inhalation of the same chemical a respiratory hypersensitivity reaction will be elicited. Allergen associates with protein and binds to, and cross-links, IgE antibody bound to mast cells. This in turn causes the degranulation of mast cells and the release of inflammatory mediators such as histamine and leukotrienes. Such mediators cause bronchoconstriction and vasodilation, resulting in the symptoms of respiratory allergy; asthma and/or rhinitis. Chemicals known to cause respiratory hypersensitivity in man include acid anhydrides (such as trimellitic anhydride), some diisocyanates (such as toluene diisocyanate), platinum salts and some reactive dyes. Also, chronic exposure to beryllium is known to cause hypersensitivity lung disease.
Autoimmunity
Autoimmunity can be defined as the stimulation of specific immune responses directed against endogenous “self” antigens. Induced autoimmunity can result either from alterations in the balance of regulatory T-lymphocytes or from the association of a xenobiotic with normal tissue components such as to render them immunogenic (“altered self”). Drugs and chemicals known to incidentally induce or exacerbate effects like those of autoimmune disease (AD) in susceptible individuals are low molecular weight compounds (molecular weight 100 to 500) that are generally considered to be not immunogenic themselves. The mechanism of AD by chemical exposure is mostly unknown. Disease can be produced directly by means of circulating antibody, indirectly through the formation of immune complexes, or as a consequence of cell-mediated immunity, but likely occurs through a combination of mechanisms. The pathogenesis is best known in immune haemolytic disorders induced by drugs:
- The drug can attach to the red-cell membrane and interact with a drug-specific antibody.
- The drug can alter the red-cell membrane so that the immune system regards the cell as foreign.
- The drug and its specific antibody form immune complexes that adhere to the red-cell membrane to produce injury.
- Red-cell sensitization occurs due to the production of red-cell autoantibody.
A variety of chemicals and drugs, in particular the latter, have been found to induce autoimmune-like responses (Kamüller, Bloksma and Seinen 1989). Occupational exposure to chemicals may incidentally lead to AD-like syndromes. Exposure to monomeric vinyl chloride, trichloroethylene, perchloroethylene, epoxy resins and silica dust may induce scleroderma-like syndromes. A syndrome similar to systemic lupus erythematosus (SLE) has been described after exposure to hydrazine. Exposure to toluene diisocyanate has been associated with the induction of thrombocytopenic purpura. Heavy metals such as mercury have been implicated in some cases of immune complex glomerulonephritis.
Human Risk Assessment
The assessment of human immune status is performed mainly using peripheral blood for analysis of humoral substances like immunoglobulins and complement, and of blood leukocytes for subset composition and functionality of subpopulations. These methods are usually the same as those used to investigate humoral and cell-mediated immunity as well as nonspecific resistance of patients with suspected congenital immunodeficiency disease. For epidemiological studies (e.g., of occupationally exposed populations) parameters should be selected on the basis of their predictive value in human populations, validated animal models, and the underlying biology of the markers (see table 1). The strategy in screening for immunotoxic effects after (accidental) exposure to environmental pollutants or other toxicants is much dependent on circumstances, such as type of immunodeficiency to be expected, time between exposure and immune status assessment, degree of exposure and number of exposed individuals. The process of assessing the immunotoxic risk of a particular xenobiotic in humans is extremely difficult and often impossible, due largely to the presence of various confounding factors of endogenous or exogenous origin that influence the response of individuals to toxic damage. This is particularly true for studies which investigate the role of chemical exposure in autoimmune diseases, where genetic factors play a crucial role.
Table 1. Classification of tests for immune markers
| Test category | Characteristics | Specific tests |
| Basic-general Should be included with general panels |
Indicators of general health and organ system status | Blood urea nitrogen, blood glucose, etc. |
| Basic-immune Should be included with general panels |
General indicators of immune status Relatively low cost Assay methods are standardized among laboratories Results outside reference ranges are clinically interpretable |
Complete blood counts Serum IgG, IgA, IgM levels Surface marker phenotypes for major lymphocyte subsets |
| Focused/reflex Should be included when indicated by clinical findings, suspected exposures, or prior test results |
Indicators of specific immune functions/events Cost varies Assay methods are standardized among laboratories Results outside reference ranges are clinically interpretable |
Histocompatibility genotype Antibodies to infectious agents Total serum IgE Allergen-specific IgE Autoantibodies Skin tests for hypersensitivity Granulocyte oxidative burst Histopathology (tissue biopsy) |
| Research Should be included only with control populations and careful study design |
Indicators of general or specific immune functions/events Cost varies; often expensive Assay methods are usually not standardized among laboratories Results outside reference ranges are often not clinically interpretable |
In vitro stimulation assays Cell activation surface markers Cytokine serum concentrations Clonality assays (antibody, cellular, genetic) Cytotoxicity tests |
As adequate human data are seldom available, the assessment of risk for chemical-induced immunosuppression in humans is in the majority of cases based upon animal studies. The identification of potential immunotoxic xenobiotics is undertaken primarily in controlled studies in rodents. In vivo exposure studies present, in this regard, the optimal approach to estimate the immunotoxic potential of a compound. This is due to the multifactoral and complex nature of the immune system and of immune responses. In vitro studies are of increasing value in the elucidation of mechanisms of immunotoxicity. In addition, by investigating the effects of the compound using cells of animal and human origin, data can be generated for species comparison, which can be used in the “parallelogram” approach to improve the risk assessment process. If data are available for three cornerstones of the parallelogram (in vivo animal, and in vitro animal and human) it may be easier to predict the outcome at the remaining cornerstone, that is, the risk in humans.
When assessment of risk for chemical-induced immunosuppression has to rely solely upon data from animal studies, an approach can be followed in the extrapolation to man by the application of uncertainty factors to the no observed adverse effect level (NOAEL). This level can be based on parameters determined in relevant models, such as host resistance assays and in vivo assessment of hypersensitivity reactions and antibody production. Ideally, the relevance of this approach to risk assessment requires confirmation by studies in humans. Such studies should combine the identification and measurement of the toxicant, epidemiological data and immune status assessments.
To predict contact hypersensitivity, guinea pig models are available and have been used in risk assessment since the 1970s. Although sensitive and reproducible, these tests have limitations as they depend on subjective evaluation; this can be overcome by newer and more quantitative methods developed in the mouse. Regarding chemical-induced hypersensitivity induced by inhalation or ingestion of allergens, tests should be developed and evaluated in terms of their predictive value in man. When it comes to setting safe occupational exposure levels of potential allergens, consideration has to be given to the biphasic nature of allergy: the sensitization phase and the elicitation phase. The concentration required to elicit an allergic reaction in a previously sensitized individual is considerably lower than the concentration necessary to induce sensitization in the immunologically naïve but susceptible individual.
As animal models to predict chemical-induced autoimmunity are virtually lacking, emphasis should be given to the development of such models. For the development of such models, our knowledge of chemical-induced autoimmunity in humans should be advanced, including the study of genetic and immune system markers to identify susceptible individuals. Humans that are exposed to drugs that induce autoimmunity offer such an opportunity.
Target Organ Toxicology
The study and characterization of chemicals and other agents for toxic properties is often undertaken on the basis of specific organs and organ systems. In this chapter, two targets have been selected for in-depth discussion: the immune system and the gene. These examples were chosen to represent a complex target organ system and a molecular target within cells. For more comprehensive discussion of the toxicology of target organs, the reader is referred to standard toxicology texts such as Casarett and Doull, and Hayes. The International Programme on Chemical Safety (IPCS) has also published several criteria documents on target organ toxicology, by organ system.
Target organ toxicology studies are usually undertaken on the basis of information indicating the potential for specific toxic effects of a substance, either from epidemiological data or from general acute or chronic toxicity studies, or on the basis of special concerns to protect certain organ functions, such as reproduction or foetal development. In some cases, specific target organ toxicity tests are expressly mandated by statutory authorities, such as neurotoxicity testing under the US pesticides law (see “The United States approach to risk assessment of reproductive toxicants and neurotoxic agents,” and mutagenicity testing under the Japanese Chemical Substance Control Law (see “Principles of hazard identification: The Japanese approach”).
As discussed in “Target organ and critical effects,” the identification of a critical organ is based upon the detection of the organ or organ system which first responds adversely or to the lowest doses or exposures. This information is then used to design specific toxicology investigations or more defined toxicity tests that are designed to elicit more sensitive indications of intoxication in the target organ. Target organ toxicology studies may also be used to determine mechanisms of action, of use in risk assessment (see “The United States approach to risk assessment of reproductive toxicants and neurotoxic agents”).
Methods of Target Organ Toxicity Studies
Target organs may be studied by exposure of intact organisms and detailed analysis of function and histopathology in the target organ, or by in vitro exposure of cells, tissue slices, or whole organs maintained for short or long term periods in culture (see “Mechanisms of toxicology: Introduction and concepts”). In some cases, tissues from human subjects may also be available for target organ toxicity studies, and these may provide opportunities to validate assumptions of cross-species extrapolation. However, it must be kept in mind that such studies do not provide information on relative toxicokinetics.
In general, target organ toxicity studies share the following common characteristics: detailed histopathological examination of the target organ, including post mortem examination, tissue weight, and examination of fixed tissues; biochemical studies of critical pathways in the target organ, such as important enzyme systems; functional studies of the ability of the organ and cellular constituents to perform expected metabolic and other functions; and analysis of biomarkers of exposure and early effects in target organ cells.
Detailed knowledge of target organ physiology, biochemistry and molecular biology may be incorporated in target organ studies. For instance, because the synthesis and secretion of small-molecular-weight proteins is an important aspect of renal function, nephrotoxicity studies often include special attention to these parameters (IPCS 1991). Because cell-to-cell communication is a fundamental process of nervous system function, target organ studies in neurotoxicity may include detailed neurochemical and biophysical measurements of neurotransmitter synthesis, uptake, storage, release and receptor binding, as well as electrophysiological measurement of changes in membrane potential associated with these events.
A high degree of emphasis is being placed upon the development of in vitro methods for target organ toxicity, to replace or reduce the use of whole animals. Substantial advances in these methods have been achieved for reproductive toxicants (Heindel and Chapin 1993).
In summary, target organ toxicity studies are generally undertaken as a higher order test for determining toxicity. The selection of specific target organs for further evaluation depends upon the results of screening level tests, such as the acute or subchronic tests used by OECD and the European Union; some target organs and organ systems may be a priori candidates for special investigation because of concerns to prevent certain types of adverse health effects.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."