79. Pharmaceutical Industry
Chapter Editor: Keith D. Tait
Table of Contents
Tables and Figures
Pharmaceutical Industry
Keith D. Tait
Case Study: Effects of Synthetic Oestrogens on Pharmaceutical Workers: A United States Example
Dennis D. Zaebst
Tables
Click a link below to view table in article context.
1. Major categories of pharmaceutical agents
2. Solvents used in the pharmaceutical industry
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Pharmaceutical Industry
Definitions
These terms are used frequently in the pharmaceutical industry:
Biologics are bacterial and viral vaccines, antigens, antitoxins and analogous products, serums, plasmas and other blood derivatives for therapeutically protecting or treating humans and animals.
Bulks are active drug substances used to manufacture dosage- form products, process medicated animal feeds or compound prescription medications.
Diagnostic agents assist the diagnosis of diseases and disorders in humans and animals. Diagnostic agents may be inorganic chemicals for examining the gastrointestinal tract, organic chemicals for visualizing the circulatory system and liver and radioactive compounds for measuring the function of organ system.
Drugs are substances with active pharmacological properties in humans and animals. Drugs are compounded with other materials, such as pharmaceutical necessities, to produce a medicinal product.
Ethical pharmaceuticals are biological and chemicals agents for preventing, diagnosing or treating disease and disorders in humans or animals. These products are dispensed by prescription or approval of a medical, pharmacy or veterinary professional.
Excipients are inert ingredients which are combined with drug substances to create a dosage form product. Excipients may affect the rate of absorption, dissolution, metabolism and distribution in humans or animals.
Over-the-counter pharmaceuticals are drug products sold in a retail store or pharmacy which do not require a prescription or the approval of a medical, pharmacy or veterinary professional.
Pharmacy is the art and science of preparing and dispensing drugs for preventing, diagnosing or treating diseases or disorders in humans and animals.
Pharmacokinetics is the study of metabolic processes relating to the absorption, distribution, biotransformation, and elimination of a drug in humans or animals.
Pharmacodynamics is the study of drug action relating to its chemical structure, site of action, and the biochemical and physiological consequences in humans and animals.
The pharmaceutical industry is an important component of health care systems throughout the world; it is comprised of many public and private organizations that discover, develop, manufacture and market medicines for human and animal health (Gennaro 1990). The pharmaceutical industry is based primarily upon the scientific research and development (R&D) of medicines that prevent or treat diseases and disorders. Drug substances exhibit a wide range of pharmacological activity and toxicological properties (Hardman, Gilman and Limbird 1996; Reynolds 1989). Modern scientific and technological advances are accelerating the discovery and development of innovative pharmaceuticals with improved therapeutic activity and reduced side effects. Molecular biologists, medicinal chemists and pharmacists are improving the benefits of drugs through increased potency and specificity. These advances create new concerns for protecting the health and safety of workers within the pharmaceutical industry (Agius 1989; Naumann et al. 1996; Sargent and Kirk 1988; Teichman, Fallon and Brandt-Rauf 1988).
Many dynamic scientific, social and economic factors affect the pharmaceutical industry. Some pharmaceutical companies operate in both national and multinational markets. Therefore, their activities are subject to legislation, regulation and policies relating to drug development and approval, manufacturing and quality control, marketing and sales (Spilker 1994). Academic, government and industry scientists, practising physicians and pharmacists, as well as the public, influence the pharmaceutical industry. Health care providers (e.g., physicians, dentists, nurses, pharmacists and veterinarians) in hospitals, clinics, pharmacies and private practice may prescribe drugs or recommend how they should be dispensed. Government regulations and health care policies on pharmaceuticals are influenced by the public, advocacy groups and private interests. These complex factors interact to influence the discovery and development, manufacturing, marketing and sales of drugs.
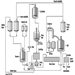
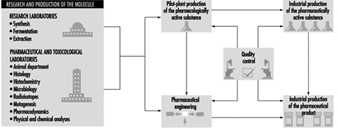
The pharmaceutical industry is largely driven by scientific discovery and development, in conjunction with toxicological and clinical experience (see figure 1). Major differences exist between large organizations which engage in a broad range of drug discovery and development, manufacturing and quality control, marketing and sales and smaller organizations which focus on a specific aspect. Most multinational pharmaceutical companies are involved in all these activities; however, they may specialize in one aspect based upon local market factors. Academic, public and private organizations perform scientific research to discover and develop new drugs. The biotechnology industry is becoming a major contributor to innovative pharmaceutical research (Swarbick and Boylan 1996). Often, collaborative agreements between research organizations and large pharmaceutical companies are formed to explore the potential of new drug substances.
Figure 1. Drug development in the pharmaceutical industry
Many countries have specific legal protections for proprietary drugs and manufacturing processes, known as intellectual property rights. In instances when legal protections are limited or do not exist, some companies specialize in manufacturing and marketing generic drugs (Medical Economics Co. 1995). The pharmaceutical industry requires large amounts of capital investment due to the high expenses associated with R&D, regulatory approval, manufacturing, quality assurance and control, marketing and sales (Spilker 1994). Many countries have extensive government regulations affecting the development and approval of drugs for commercial sale. These countries have strict requirements for good manufacturing practices to ensure the integrity of drug manufacturing operations and the quality, safety and efficacy of pharmaceutical products (Gennaro 1990).
International and domestic trade, as well as tax and finance policies and practices, affect how the pharmaceutical industry operates within a country (Swarbick and Boylan 1996). Significant differences exist between developed and developing countries, regarding their needs for pharmaceutical substances. In developing countries, where malnutrition and infectious diseases are prevalent, nutritional supplements, vitamins and anti-infective drugs are most needed. In developed countries, where the diseases associated with ageing and specific ailments are primary health concerns, cardiovascular, central nervous system, gastrointestinal, anti-infective, diabetes and chemotherapy drugs are in the greatest demand.
Human and animal health drugs share similar R&D activities and manufacturing processes; however, they have unique therapeutic benefits and mechanisms for their approval, distribution, marketing and sales (Swarbick and Boylan 1996). Veterinarians administer drugs to control infectious diseases and parasitic organisms in agricultural and companion animals. Vaccines and anti-infective and antiparasitic drugs are commonly used for this purpose. Nutritional supplements, antibiotics and hormones are widely employed by modern agriculture to promote the growth and health of farm animals. The R&D of pharmaceuticals for human and animal health are often allied, due to concurrent needs to control infectious agents and disease.
Hazardous Industrial Chemicals and Drug-related Substances
Many different biological and chemical agents are discovered, developed and used in the pharmaceutical industry (Hardman, Gilman and Limbird 1996; Reynolds 1989). Some manufacturing processes in the pharmaceutical, biochemical and synthetic organic chemical industries are similar; however, the greater diversity, smaller scale and specific applications in the pharmaceutical industry are unique. Since the primary purpose is to produce medicinal substances with pharmacological activity, many agents in pharmaceutical R&D and manufacturing are hazardous to workers. Proper control measures must be implemented to protect workers from industrial chemicals and drug substances during many R&D, manufacturing and quality control operations (ILO 1983; Naumann et al. 1996; Teichman, Fallon and Brandt-Rauf 1988).
The pharmaceutical industry uses biological agents (e.g., bacteria and viruses) in many special applications, such as vaccine production, fermentation processes, derivation of blood-based products and biotechnology. Biological agents are not addressed by this profile due to their unique pharmaceutical applications, but other references are readily available (Swarbick and Boylan 1996). Chemical agents may be categorized as industrial chemicals and drug-related substances (Gennaro 1990). These may be raw materials, intermediates or finished products. Special situations arise when industrial chemicals or drug substances are employed in laboratory R&D, quality assurance and control assays, engineering and maintenance, or when they are created as by-products or wastes.
Industrial chemicals
Industrial chemicals are used in researching and developing active drug substances and manufacturing bulk substances and finished pharmaceutical products. Organic and inorganic chemicals are raw materials, serving as reactants, reagents, catalysts and solvents. The use of industrial chemicals is determined by the specific manufacturing process and operations. Many of these materials may be hazardous to workers. Since worker exposures to industrial chemicals may be hazardous, occupational exposure limits, such as threshold limit values (TLVs) have been established by government, technical and professional organizations (ACGIH 1995).
Drug-related substances
Pharmacologically active substances may be categorized as natural products and synthetic drugs. Natural products are derived from plant and animal sources, while synthetic drugs are produced by microbiological and chemical technologies. Antibiotics, steroid and peptide hormones, vitamins, enzymes, prostaglandins and pheromones are important natural products. Scientific research is focusing increasingly on synthetic drugs due to recent scientific advances in molecular biology, biochemistry, pharmacology and computer technology. Table 1 lists the principal pharmaceutical agents.
Table 1. Major categories of pharmaceutical agents
|
Central nervous |
Renal and |
Gastrointestinal |
Anti-infectives |
Immune system |
Chemotherapy |
Blood and |
Endocrine system |
|
Analgesics Psychotherapeutics |
Antidiabetics |
Gastrointestinal agents |
Systemic |
Analgesics Immunodilators and immuno- |
Antineoplastics |
Blood modifiers stimulating |
Diagnostics Prostaglandins |
Active drug substances and inert materials are combined during pharmaceutical manufacturing to produce dosage forms of medicinal products (e.g., tablets, capsules, liquids, powders, creams and ointments) (Gennaro 1990). Drugs may be categorized by their manufacturing process and therapeutic benefits (EPA 1995). Drugs are medicinally administered by strictly prescribed means (e.g., oral, injection, skin) and dosages, whereas workers may be exposed to drug substances by inadvertently breathing airborne dust or vapours or accidentally swallowing contaminated foods or beverages. Occupational exposure limits (OELs) are developed by toxicologists and occupational hygienists to provide guidance on limiting worker exposures to drug substances (Naumann et al. 1996; Sargent and Kirk 1988).
Pharmaceutical necessities (e.g., binders, fillers, flavouring and bulking agents, preservatives and antioxidants) are mixed with active drug substances, providing the desired physical and pharmacological properties in the dosage form products (Gennaro 1990). Many pharmaceutical necessities have no or limited therapeutic value and are relatively non-hazardous to workers during drug development and manufacturing operations. These materials are anti-oxidants and preservatives, colouring, flavouring and diluting agents, emulsifiers and suspending agents, ointment bases, pharmaceutical solvents and excipients.
Pharmaceutical Operations, Related Hazards and Workplace Control Measures
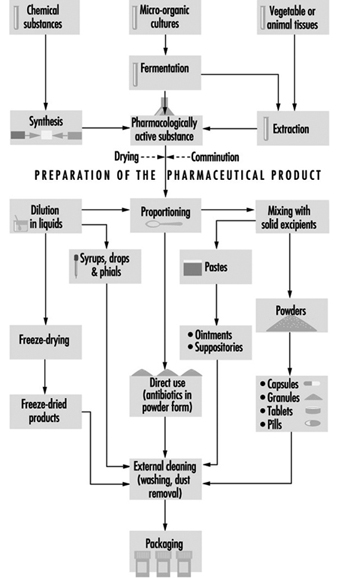
Pharmaceutical manufacturing operations may be categorized as basic production of bulk drug substances and pharmaceutical manufacturing of dosage form products. Figure 2 illustrates the manufacturing process.
Figure 2. Manufacturing process in pharmaceutical industry
Basic production of bulk drug substances may employ three major types of processes: fermentation, organic chemical synthesis, and biological and natural extraction (Theodore and McGuinn 1992). These manufacturing operations may be discrete batch, continuous or a combination of these processes. Antibiotics, steroids and vitamins are produced by fermentation, whereas many new drug substances are produced by organic synthesis. Historically, most drug substances were derived from natural sources such as plants, animals, fungi and other organisms. Natural medicines are pharmacologically diverse and difficult to produce commercially due to their complex chemistry and limited potency.
Fermentation
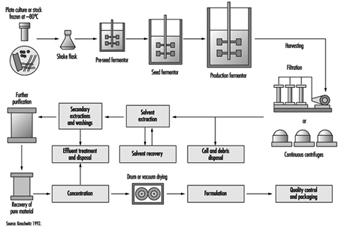
Fermentation is a biochemical process employing selected micro-organisms and microbiological technologies to produce a chemical product. Batch fermentation processes involve three basic steps: inoculum and seed preparation, fermentation, and product recovery or isolation (Theodore and McGuinn 1992). A schematic diagram of a fermentation process is given in figure 3. Inoculum preparation begins with a spore sample from a microbial strain. The strain is selectively cultured, purified and grown using a battery of microbiological techniques to produce the desired product. The spores of the microbial strain are activated with water and nutrients in warm conditions. Cells from the culture are grown through a series of agar plates, test tubes and flasks under controlled environmental conditions to create a dense suspension.
Figure 3. Diagram of a fermentation process
The cells are transferred to a seed tank for further growth. The seed tank is a small fermentation vessel designed to optimize the growth of the inoculum. The cells from the seed tank are charged to a steam sterilized production fermentor. Sterilized nutrients and purified water are added to the vessel to begin the fermentation. During aerobic fermentation, the contents of the fermentor are heated, agitated and aerated by a perforated pipe or sparger, maintaining an optimum air flow rate and temperature. After the biochemical reactions are complete, the fermentation broth is filtered to remove the micro-organisms, or mycelia. The drug product, which may be present in the filtrate or within the mycelia, is recovered by various steps, such as solvent extraction, precipitation, ion exchange and absorption.
Solvents used for extracting the product (table 2) generally can be recovered; however, small portions remain in the process wastewater, depending upon their solubility and the design of the process equipment. Precipitation is a method to separate the drug product from the aqueous broth. The drug product is filtered from the broth and extracted from the solid residues. Copper and zinc are common precipitating agents in this process. Ion exchange or adsorption removes the product from the broth by chemical reaction with solid materials, such as resins or activated carbon. The drug product is recovered from the solid phase by a solvent which may be recovered by evaporation.
Table 2. Solvents used in the pharmaceutical industry
|
Solvents |
Processes |
||
|
Acetone |
C |
F |
B |
|
Acetonitrile |
C |
F |
B |
|
Ammonia (aqueous) |
C |
F |
B |
|
n-Amyl acetate |
C |
F |
B |
|
Amyl alcohol |
C |
F |
B |
|
Aniline |
C |
||
|
Benzene |
C |
||
|
2-Butanone (MEK) |
C |
||
|
n-Butyl acetate |
C |
F |
|
|
n-Butyl alcohol |
C |
F |
B |
|
Chlorobenzene |
C |
||
|
Chloroform |
C |
F |
B |
|
Chloromethene |
C |
||
|
Cyclohexane |
C |
||
|
o-Dichlorobenzene (1,2-Dichlorobenzene) |
C |
||
|
1,2-Dichloroethane |
C |
B |
|
|
Diethylamine |
C |
B |
|
|
Diethyl ether |
C |
B |
|
|
N,N-Dimethyl acetamide |
C |
||
|
Dimethylamine |
C |
||
|
N,N-dimethylaniline |
C |
||
|
N,N-dimethylformamide |
C |
F |
B |
|
Dimethyl sulphoxide |
C |
B |
|
|
1,4-Dioxane |
C |
B |
|
|
Ethanol |
C |
F |
B |
|
Ethyl acetate |
C |
F |
B |
|
Ethylene glycol |
C |
B |
|
|
Formaldehyde |
C |
F |
B |
|
Formamide |
C |
||
|
Furfural |
C |
||
|
n-Heptane |
C |
F |
B |
|
n-Hexane |
C |
F |
B |
|
Isobutyraldehyde |
C |
||
|
Isopropanol |
C |
F |
B |
|
Isopropyl acetate |
C |
F |
B |
|
Isopropyl ether |
C |
B |
|
|
Methanol |
C |
F |
B |
|
Methylamine |
C |
||
|
Methyl cellosolve |
C |
F |
|
|
Methylene chloride |
C |
F |
B |
|
Methyl formate |
C |
||
|
Methyl isobutyl ketone (MIBK) |
C |
F |
B |
|
2-Methylpyridine |
C |
||
|
Petroleum naphtha |
C |
F |
B |
|
Phenol |
C |
F |
B |
|
Polyethylene glycol 600 |
C |
||
|
n-Propanol |
C |
B |
|
|
Pyridine |
C |
B |
|
|
Tetrahydrofuran |
C |
||
|
Toluene |
C |
F |
B |
|
Trichlorofluoromethane |
C |
||
|
Triethylamine |
C |
F |
|
|
Xylenes |
C |
||
C = chemical synthesis, F = fermentation, B = biological or natural extraction.
Source: EPA 1995.
Worker health and safety
Worker safety hazards may be posed by moving machine parts and equipment; high pressure steam, hot water, heated surfaces and hot workplace environments; corrosive and irritating chemicals; heavy manual handling of materials and equipment; and high noise levels. Worker exposures to solvent vapours may occur when recovering or isolating products. Worker exposures to solvents may result from uncontained filtration equipment and fugitive emissions for leaking pumps, valves and manifold stations during extraction and purification steps. Since the isolation and growth of micro-organisms are essential for fermentation, biological hazards are reduced by employing non-pathogenic microbes, maintaining closed process equipment and treating spent broth before its discharge.
Generally, process safety concerns are less important during fermentation than during organic synthesis operations, since fermentation is primarily based upon aqueous chemistry and requires process containment during seed preparation and fermentation. Fire and explosion hazards may arise during solvent extractions; however, the flammability of solvents is reduced by dilution with water in filtration and recovery steps. Safety hazards (i.e., thermal burns and scalding) are posed by the large volumes of pressurized steam and hot water associated with fermentation operations.
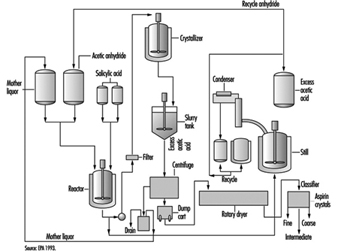
Chemical synthesis
Chemical synthesis processes use organic and inorganic chemicals in batch operations to produce drug substances with unique physical and pharmacological properties. Typically, a series of chemical reactions are performed in multi-purpose reactors and the products are isolated by extraction, crystallization and filtration (Kroschwitz 1992). The finished products are usually dried, milled and blended. Organic synthesis plants, process equipment and utilities are comparable in the pharmaceutical and fine chemical industries. A schematic diagram of an organic synthesis process is given in figure 4.
Figure 4. Diagram of an organic synthesis process
Pharmaceutical chemistry is becoming increasingly complex with multi-step processing, where the product from one step becomes a starting material for the next step, until the finished drug product is synthesized. Bulk chemicals which are intermediates of the finished product may be transferred between organic synthesis plants for various technical, financial and legal considerations. Most intermediates and products are produced in a series of batch reactions on a campaign basis. Manufacturing processes operate for discrete periods of time, before materials, equipment and utilities are changed to prepare for a new process. Many organic synthesis plants in the pharmaceutical industry are designed to maximize their operating flexibility, due to the diversity and complexity of modern medicinal chemistry. This is achieved by constructing facilities and installing process equipment that can be modified for new manufacturing processes, in addition to their utility requirements.
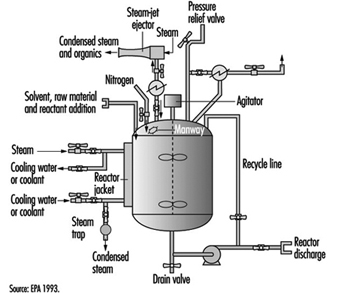
Multi-purpose reactors are the primary processing equipment in chemical synthesis operations (see figure 5). They are reinforced pressure vessels with stainless, glass or metal alloy linings. The nature of chemical reactions and physical properties of materials (e.g., reactive, corrosive, flammable) determine the design, features and construction of reactors. Multi-purpose reactors have external shells and internal coils which are filled with cooling water, steam or chemicals with special heat-transfer properties. The reactor shell is heated or cooled, based upon the requirements of the chemical reactions. Multi-purpose reactors have agitators, baffles and many inlets and outlets connecting them to other process vessels, equipment and bulk chemical supplies. Temperature-, pressure- and weight-sensing instruments are installed to measure and control the chemical process in the reactor. Reactors may be operated at high pressures or low vacuums, depending upon their engineering design and features and the requirements of the process chemistry.
Figure 5. Diagram of a chemical reactor in organic synthesis
Heat exchangers are connected to reactors to heat or cool the reaction and condense solvent vapours when they are heated above their boiling point, creating a reflux or recycling of the condensed vapours. Air pollution control devices (e.g., scrubbers and impingers) can be connected to the exhaust vents on process vessels, reducing gas, vapour and dust emissions (EPA 1993). Volatile solvents and toxic chemicals may be released to the workplace or atmosphere, unless they are controlled during the reaction by heat exchangers or air control devices. Some solvents (see table 2) and reactants are difficult to condense, absorb or adsorb in air control devices (e.g., methylene chloride and chloroform) due to their chemical and physical properties.
Bulk chemical products are recovered or isolated by separation, purification and filtration operations. Typically, these products are contained in mother liquors, as dissolved or suspended solids in a solvent mixture. The mother liquors may be transferred between process vessels or equipment in temporary or permanent pipes or hoses, by pumps, pressurized inert gases, vacuum or gravity. Transferring materials is a concern due to the rates of reaction, critical temperatures or pressures, features of processing equipment and potential for leaks and spills. Special precautions to minimize static electricity are required when processes use or generate flammable gases and liquids. Charging flammable liquids through submerged dip tubes and grounding and bonding conductive materials and maintaining inert atmospheres inside process equipment reduce the risk of a fire or explosion (Crowl and Louvar 1990).
Worker health and safety
Many worker health and safety hazards are posed by synthesis operations. They include safety hazards from moving machine parts, pressurized equipment and pipes; heavy manual handling of materials and equipment; steam, hot liquids, heated surfaces and hot workplace environments; confined spaces and hazardous energy sources (e.g., electricity); and high noise levels.
Acute and chronic health risks may result from worker exposures to hazardous chemicals during synthesis operations. Chemicals with acute health effects can damage the eyes and skin, be corrosive or irritating to body tissues, cause sensitization or allergic reactions or be asphyxiants, causing suffocation or oxygen deficiency. Chemicals with chronic health effects may cause cancer, or damage the liver, kidneys or lungs or affect the nervous, endocrine, reproductive or other organ systems. Health and safety hazards may be controlled by implementing appropriate control measures (e.g., process modifications, engineering controls, administrative practices, personal and respiratory protective equipment).
Organic synthesis reactions may create major process safety risks from highly hazardous materials, fire, explosion or uncontrolled chemical reactions which impact the community surrounding the plant. Process safety can be very complex in organic synthesis. It is addressed in several ways: by examining the dynamics of chemical reactions, properties of highly hazardous materials, design, operation and maintenance of equipment and utilities, training of operating and engineering staff, and emergency preparedness and response of the facility and local community. Technical guidance is available on process hazard analysis and management activities to reduce the risks of chemical synthesis operations (Crowl and Louvar 1990; Kroschwitz 1992).
Biological and natural extraction
Large volumes of natural materials, such as plant and animal matter, may be processed to extract substances which are pharmacologically active (Gennaro 1990; Swarbick and Boylan 1996). In each step of the process, the volumes of materials are reduced by a series of batch processes, until the final drug product is obtained. Typically, processes are performed in campaigns lasting a few weeks, until the desired quantity of finished product is obtained. Solvents are used to remove insoluble fats and oils, thereby extracting the finished drug substance. The pH (acidity) of the extraction solution and waste products can be adjusted by neutralizing them with strong acids and bases. Metal compounds frequently serve as precipitating agents, and phenol compounds as disinfectants.
Worker health and safety
Some workers may develop allergic and/or skin irritation from handling certain plants. Animal matter may be contaminated with infectious organisms unless appropriate precautions are taken. Workers may be exposed to solvents and corrosive chemicals during biological and natural extraction operations. Fire and explosion risks are posed by storing, handling, processing and recovering flammable liquids. Moving mechanical parts; hot steam, water, surfaces and workplaces; and high noise levels are risks to worker safety.
Process safety issues are often reduced by the large volumes of plant or animal materials, and smaller scale of solvent extraction activities. Fire and explosion hazards, and worker exposures to solvents or corrosive or irritating chemicals may occur during extraction and recovery operations, depending upon the specific chemistry and containment of process equipment.
Pharmaceutical manufacturing of dosage forms
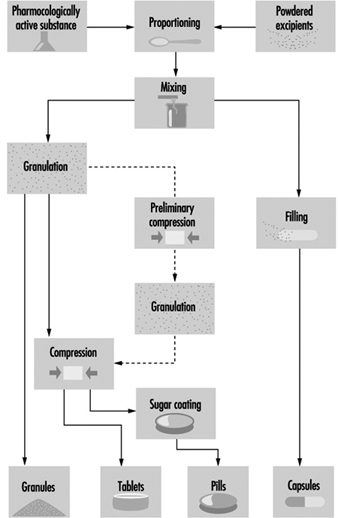
Drug substances are converted into dosage-form products before they are dispensed or administered to humans or animals. Active drug substances are mixed with pharmaceutical necessities, such as binders, fillers, flavouring and bulking agents, preservatives and antioxidants. These ingredients may be dried, milled, blended, compressed and granulated to achieve the desired properties before they are manufactured as a final formulation. Tablets and capsules are very common oral dosage forms; another common form is sterile liquids for injection or ophthalmic application. Figure 6 illustrates typical unit operations for manufacturing of pharmaceutical dosage-form products.
Figure 6. Pharmaceutical manufacturing of dosage-form products
Pharmaceutical blends may be compressed by wet granulation, direct compression or slugging to obtain the desired physical properties, before their formulation as a finished drug product. In wet granulation, the active ingredients and excipients are wetted with aqueous or solvent solutions to produce course granules with enlarged particle sizes. The granules are dried, mixed with lubricants (e.g., magnesium stearate), disintegrants or binders, then compressed into tablets. During direct compression, a metal die holds a measured amount of the drug blend while a punch compresses the tablet. Drugs that are not sufficiently stable for wet granulation or cannot be directly compressed are slugged. Slugging or dry granulation blends and compresses relatively large tablets which are ground and screened to a desired mesh size, then recompressed into the final tablet. Blended and granulated materials may also be produced in capsule form. Hard gelatin capsules are dried, trimmed, filled and joined on capsule-filling machines.
Liquids may be produced as sterile solutions for injection into the body or administration to the eyes; liquids, suspensions and syrups for oral ingestion; and tinctures for application on the skin (Gennaro 1990). Highly controlled environmental conditions, contained process equipment and purified raw materials are required for manufacturing sterile liquids to prevent microbiological and particulate contamination (Cole 1990; Swarbick and Boylan 1996). Facility utilities (e.g., ventilation, steam and water), process equipment and workplace surfaces must be cleaned and maintained to prevent and minimize contamination. Water at high temperatures and pressures is used to destroy and filter bacteria and other contaminants from the sterile water supply when making solutions for injection. Parenteral liquids are injected by intradermal, intramuscular or intravenous administration into the body. These liquids are sterilized by dry or moist heat under high pressure with bacteria-retaining filters. Although liquid solutions for oral or topical use do not require sterilization, solutions to be administered to the eyes (ophthalmic) must be sterilized. Oral liquids are prepared by mixing the active drug substances with a solvent or preservative to inhibit mold and bacterial growth. Liquid suspensions and emulsions are produced by colloid mills and homogenizers, respectively. Creams and ointments are prepared by blending or compounding active ingredients with petrolatum, heavy greases or emollients before packaging in metal or plastic tubes.
Worker health and safety
Worker health and safety risks during pharmaceutical manufacturing are created by moving machine parts (e.g., exposed gears, belts and shafts) and hazardous energy sources (e.g., electrical, pneumatic, thermal, etc.); manual handling of material and equipment; high-pressure steam, hot water and heated surfaces; flammable and corrosive liquids; and high noise levels. Worker exposures to airborne dusts may occur during dispensing, drying, milling and blending operations. Exposure to pharmaceutical products is a particular concern when mixtures containing high proportions of active drug substances are handled or processed. Wet granulation, compounding and coating operations may create high worker exposures to solvent vapours.
Process safety issues primarily relate to the risks of fire or explosion during pharmaceutical manufacturing of dosage forms. Many of these operations (e.g., granulation, blending, compounding and drying) use flammable liquids, which may create flammable or explosive atmospheres. Since some pharmaceutical dusts are highly explosive, their physical properties should be examined before they are processed. Fluid bed drying, milling and slugging are a particular concern when they involve potentially explosive materials. Engineering measures and safe work practices reduce the risks of explosive dusts and flammable liquids (e.g., vapour- and dust-tight electrical equipment and utilities, grounding and bonding of equipment, sealed containers with pressure relief and inert atmospheres).
Control measures
Fire and explosion prevention and protection; process containment of hazardous substances, machine hazards and high noise levels; dilution and local exhaust ventilation (LEV); use of respirators (e.g., dust and organic vapour masks and, in some cases, powered air-purifying respirators or air-supplied masks and suits) and personal protective equipment (PPE); and worker training on workplace hazards and safe work practices are workplace control measures applicable during all of the various pharmaceutical manufacturing operations described below. Specific issues involve substituting less hazardous materials whenever possible during drug development and manufacturing. Also, minimizing material transfers, unsealed or open processing and sampling activities decreases the potential for worker exposures.
The engineering design and features of facilities, utilities and process equipment can prevent environmental pollution and reduce worker exposures to hazardous substances. Modern pharmaceutical manufacturing facilities and process equipment are reducing environmental, health and safety risks by preventing pollution and improving the containment of hazards. Worker health and safety and quality control objectives are achieved by improving the isolation, containment and cleanliness of pharmaceutical facilities and process equipment. Preventing worker exposures to hazardous substances and pharmaceutical products is highly compatible with the concurrent need to prevent workers from accidentally contaminating raw materials and finished products. Safe work procedures and good manufacturing practices are complementary activities.
Facility design and process-engineering issues
The engineering design and features of pharmaceutical facilities and process equipment influences worker health and safety. The construction materials, process equipment and housekeeping practices greatly affect the cleanliness of the workplace. Dilution and LEV systems control fugitive vapours and dust emissions during manufacturing operations. Fire and explosion prevention and protection measures (e.g., vapour- and dust-tight electrical equipment and utilities, extinguishing systems, fire and smoke detectors and emergency alarms) are needed when flammable liquids and vapours are present. Storage and handling systems (e.g., storage vessels, portable containers, pumps and piping) are installed to move liquids within pharmaceutical manufacturing facilities. Hazardous solids can be handled and processed in enclosed equipment and vessels, individual bulk containers (IBCs) and sealed drums and bags. The isolation or containment of facilities, process equipment and hazardous materials promotes worker health and safety. Mechanical hazards are controlled by installing barrier guards on moving machine parts.
The process equipment and utilities may be controlled by manual or automatic means. In manual plants, chemical operators read instruments and control process equipment and utilities near the process equipment. In automated plants, the process equipment, utilities and control devices are controlled by distributed systems, allowing them to be operated from a remote location such as a control room. Manual operations are often employed when materials are charged or transferred, products are discharged and packaged and when maintenance is performed or nonroutine conditions arise. Written instructions should be prepared, to describe standard operating procedures as well as worker health and safety hazards and control measures.
Verification of workplace controls
Workplace control measures are evaluated periodically to protect workers from health and safety hazards and minimize environmental pollution. Many manufacturing processes and pieces of equipment are validated in the pharmaceutical industry to ensure the quality of products (Cole 1990; Gennaro 1990; Swarbick and Boylan 1996). Similar validation practices may be implemented for workplace control measures to ensure that they are effective and reliable. Periodically, process instructions and safe work practices are revised. Preventive maintenance activities identify when process and engineering equipment may fail, thereby precluding problems. Training and supervision informs and educates workers about environmental, health and safety hazards, reinforcing safe work practices and the use of respirators and personal protective equipment. Inspection programmes examine whether safe workplace conditions and work practices are maintained. This includes inspecting respirators and to ensure they are properly selected, worn and maintained by workers. Audit programmes review the management systems for identifying, evaluating and controlling environmental, health and safety hazards.
Pharmaceutical unit operations
Weighing and dispensing
Weighing and dispensing of solids and liquids is a very common activity throughout the pharmaceutical industry (Gennaro 1990). Usually workers dispense materials by hand-scooping solids and pouring or pumping liquids. Weighing and dispensing are often performed in a warehouse during bulk chemical production or in a pharmacy during pharmaceutical dosage-form manufacturing. Due to the likelihood of spills, leaks and fugitive emissions during weighing and dispensing, proper workplace control measures are necessary to protect workers. Weighing and dispensing should be performed in a partitioned workplace area with good dilution ventilation. The work surfaces in areas where materials are weighed and dispensed should be smooth and sealed, permitting their proper cleaning. LEV with backdraft or sidedraft hoods prevents the release of air contaminants when weighing and dispensing dusty solids or volatile liquids (Cole 1990). Weighing and dispensing highly toxic materials may require additional control measures such as laminar ventilation hoods or isolation devices (e.g., glove boxes or glove bags) (Naumann et al. 1996).
Charging and discharging solids and liquids
Solids and liquids are frequently charged and discharged from containers and process equipment in pharmaceutical manufacturing operations (Gennaro 1990). Charging and discharging of materials are often performed manually by workers; however, other methods are employed (e.g., gravity, mechanical or pneumatic transfer systems). Contained process equipment, transfer systems and engineering controls prevent worker exposures during charging and discharging of highly hazardous materials. Gravity charging from enclosed containers and vacuum, pressure and pumping systems eliminate fugitive emissions during charging and discharging operations. LEV with flanged inlets captures fugitive dusts and vapours which are released at open transfer points.
Liquid separations
Liquids are separated based upon their physical properties (e.g., density, solubility and miscibility) (Kroschwitz 1992). Liquid separations are commonly performed during bulk chemical production and pharmaceutical manufacturing operations. Hazardous liquids should be transferred, processed and separated in closed vessels and piping systems to reduce worker exposures to liquid spills and airborne vapours. Eyewashes and safety showers should be located near operations where hazardous liquids are transferred, processed or separated. Spill control measures and fire and explosion prevention and protection are needed when using flammable liquids.
Transferring liquids
Liquids are often transferred between storage vessels, containers and process equipment during pharmaceutical manufacturing operations. Ideally, facility and manufacturing processes are designed to minimize the need for transferring hazardous materials, thereby decreasing the chance of spills and worker exposures. Liquids may be transferred between process vessels and equipment through manifold stations, areas where many pipe flanges are located close together (Kroschwitz 1992). This allows temporary connections to be made between piping systems. Spills, leaks and vapour emissions may occur at manifold stations; therefore proper gaskets and tight seals on hoses and pipes are needed to prevent environmental pollution and workplace releases. Drainage systems with sealed tanks or sumps capture spilled liquids so they can be reclaimed and recovered. Sealed vessels and containers and piping systems are highly desirable when transferring large volumes of liquids. Special precautions should be taken when using inert gases to pressurize transfer lines or process equipment, since this may increase the release of volatile organic compounds (VOCs) and hazardous air pollutants. Recirculation or condensation of exhaust gases and vapours reduces air pollution.
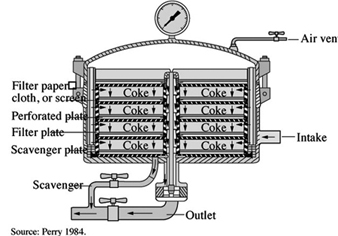
Filtration
Solids and liquids are separated during filtration operations. Filters have different designs and features with varying containment and control of liquids and vapours (Kroschwitz 1992; Perry 1984). When open filters are used for hazardous materials, workers may be exposed to liquids, wet solids, vapours and aerosols during loading and unloading operations. Closed process equipment can be used to filter highly hazardous materials, reducing vapour emissions and preventing worker exposures (see figure 7). Filtration should be performed in areas with spill control and good dilution and LEV. Volatile solvent vapours can be exhausted through vents on sealed process equipment and controlled by air emissions devices (e.g., condensers, scrubbers and adsorbers).
Figure 7. A sparkler filter
Compounding
Solids and liquids are mixed in compounding operations to produce solutions, suspensions, syrups, ointments and pastes. Contained process equipment and transfer systems are recommended when compounding highly hazardous materials (Kroschwitz 1992; Perry 1984). Buffering agents, detergents and germicides that are neutralizing, cleaning and biocidal agents may be hazardous to workers. Eyewashes and safety showers reduce injuries, if workers accidentally contact corrosive or irritating substances. Due to the wet surfaces in compounding areas, workers need to be protected from electrical hazards of equipment and utilities. Thermal hazards are posed by steam and hot water during compounding and cleaning activities. Worker injuries from burns and falls are prevented by installing insulation on hot surfaces and maintaining dry non-slip floors.
Figure 8. A high steam granulator
Figure MISSING
Granulation
Dry and wet solids are granulated to change their physical properties. Granulators have different designs and features with varying containment and control of mechanical hazards and airborne dusts and vapours (Perry 1984; Swarbick and Boylan 1996). Enclosed granulators can be vented to air-control devices, reducing emissions of solvent vapours or dusts to the workplace and atmosphere (see figure 8). Material-handling concerns arise when loading and unloading granulators. Mechanical equipment (e.g., elevated platforms, lift tables and pallet jacks) assists workers to perform heavy manual tasks. Eyewashes and safety showers are needed, if workers accidentally contact solvents or irritating dusts.
Figure 9. A rotary vacuum dryer
Figure MISSING
Drying
Water- or solvent-wet solids are dried during many pharmaceutical manufacturing operations. Dryers have different designs and features with varying containment and control of vapours and dusts (see figure 9). Flammable solvent vapours and explosive airborne dusts may create flammable or explosive atmospheres; explosion relief venting is particularly important on contained dryers. Dilution and LEV reduces the risk of fire or explosion, in addition to controlling worker exposures to solvent vapours when handling wet cakes, or to airborne dusts when unloading dried products. Heavy material handling may be involved when loading or unloading dryer trays, bins or containers (see figure 10). Mechanical equipment (e.g., drum jacks, lifts and work platforms) assists these manual tasks. Eyewashes and safety showers should be located nearby, in case workers accidentally contact solvents and dusts.
Figure 10. A vacuum shelf dryer
Figure MISSING
Milling
Dry solids are milled to change their particle characteristics and produce free-flowing powders. Mills have different designs and features with varying containment and control of mechanical hazards and airborne dusts (Kroschwitz 1992; Perry 1984). Prior to milling materials, their physical properties and hazards should be reviewed or tested. Explosion prevention and protection measures involve installing dust-tight electrical equipment and utilities, grounding and bonding equipment and accessories to eliminate electrostatic sparking, installing safety relief valves on enclosed mills, and constructing blast relief panels in walls. These measures may be necessary due to the explosivity of some drug substances and excipients, high dust levels and energies associated with milling operations.
Blending
Dry solids are blended to produce homogeneous mixtures. Blenders have different designs and features with varying containment and control of mechanical hazards and airborne dusts (Kroschwitz 1992; Perry 1984). Worker exposures to drug substances, excipients and blends may occur when loading and unloading blending equipment. LEV with flanged inlets reduces fugitive dust emissions during blending. Heavy material handling may be required when charging and discharging solids from blenders. Mechanical equipment (e.g., work platforms, hoists and drum and pallet jacks) reduces the physical demands of heavy material handling.
Compression
Dry solids are compressed or slugged to compact them, changing their particle properties. Compression equipment has different designs and features with varying containment and control of mechanical hazards and airborne dusts (Gennaro 1990; Swarbick and Boylan 1996). Compression equipment may pose serious mechanical hazards if inadequately guarded. High noise levels may also be produced by compression and slugging operations. Enclosing impact sources, isolating vibrating equipment, rotating workers and using hearing-protective devices (e.g., ear muffs and plugs) reduce the impact of noise exposures.
Figure 11. Tablet press with load hopper and spiral dust pickups for product recovery
Figure MISSING
Solid dosage-form manufacturing
Tablets and capsules are the most common oral dosage forms. Compressed or moulded tablets contain mixtures of drug substances and excipients. These tablets may be uncoated or coated with solvent mixtures or aqueous solutions. Capsules are soft or hard gelatin shells. Tablet presses (see figure 11), tablet-coating equipment and capsule-filling machines have different designs and features with varying containment and control of mechanical hazards and airborne dusts (Cole 1990). Workers may be exposed to solvent vapours when spray-coating tablets. Modern tablet-coating equipment is highly contained; however, LEV can be installed in older open coating pans to control fugitive solvent vapours. Tablet-coating equipment can be vented to air emission devices to control VOCs from the process (see figure 12). Whenever possible, recovered solvents should be reused by the process or aqueous mixtures substituted for solvent mixtures for tablet coating. Modern tablet presses and capsule-filling machines are enclosed by interlocked panels, reducing the hazards of fast-moving parts, high noise levels and dust emissions during their operation. Hearing-protective devices can reduce worker noise exposures during tablet and capsule operations.
Figure 12. A tablet coating machine
Figure MISSING
Sterile manufacturing
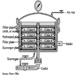
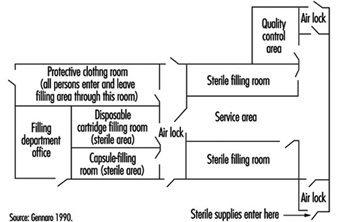
Sterile products are manufactured in pharmaceutical manufacturing plants with modular design (see figure 13), clean workplace and equipment surfaces, and high efficiency particulate air (HEPA) filtered ventilation systems (Cole 1990; Gennaro 1990). The principles and practices of controlling contamination in sterile liquid manufacturing are similar to those in the microelectronics industry. Workers wear protective clothing to prevent them from contaminating products during sterile manufacturing operations. Sterile pharmaceutical technologies to control contamination involve freeze-drying products, using liquid germicides and sterilizing gases, installing laminar flow ventilation, isolating modules with differential air pressures and containing manufacturing and filling equipment.
Figure 13. Diagram of a sterile liquid manufacturing facility
Chemical hazards are posed by toxic germicides (e.g., formaldehyde and glutaraldehyde) and sterilizing gases (i.e., ethylene oxide). Whenever possible, less hazardous agents should be selected (e.g., alcohols, ammonium compounds). Sterilization of raw materials and equipment may be performed by high-pressure steam or toxic gases (i.e., diluted ethylene oxide gas mixtures) (Swarbick and Boylan 1996). Sterilization vessels can be located in separate areas with remote instrument and control systems, non-recirculated air and LEV to extract toxic gas emissions. Workers should be trained on standard operating instructions, safe work practices and appropriate emergency response. Gas sterilization chambers should be fully evacuated under vacuum and purged with air to minimize fugitive workplace emissions before sterilized goods are removed. Gas emissions from sterilization chambers can be vented to air control devices (e.g., carbon adsorption or catalytic converters) to reduce atmospheric emissions. Occupational hygiene monitoring measures worker exposures to chemical germicides and sterilizing gases, helping to assess the adequacy of control measures. Safety hazards involve high-pressure steam and hot water, moving machine parts in washing, filling, capping and packaging equipment, high noise levels and repetitive manual tasks.
Cleaning and maintenance activities
Non-routine tasks may occur when cleaning, repairing and maintaining equipment, utilities and workplaces. Although unique hazards may arise during non-routine tasks, recurring health and safety concerns are encountered. Workplace and equipment surfaces may be contaminated by hazardous materials and drug substances, requiring them to be cleaned before unprotected workers conduct servicing or maintenance work. Cleaning is performed by washing or wiping liquids and sweeping or vacuuming dusts. Dry sweeping and blowing solids with compressed air are not recommended, since they create high worker exposures to airborne dusts. Wet mopping and vacuuming reduce worker exposures to dusts during cleaning activities. Vacuum cleaners with HEPA filters may be needed when cleaning hazardous substances and high-potency drugs. Explosion-proof equipment and conductive materials may be required in vacuum systems for explosive dusts. Eyewashes and safety showers and PPE reduce the effect of workers’ accidental contact with corrosive and irritating detergents and cleaning liquids.
Hazardous mechanical, electrical, pneumatic or thermal energy may need to be released or controlled before equipment and utilities are serviced, repaired or maintained. Contract workers may perform special production or engineering tasks in pharmaceutical plants without adequate training on safety precautions. Careful supervision of contract workers is important, so they do not violate safety rules or perform work that creates a fire, explosion or other serious health and safety hazards. Special contractor safety programmes are required when working with highly hazardous materials (e.g., toxic, reactive, flammable or explosive) and processes (e.g., exothermic or high pressure) in bulk pharmaceutical and dosage-form manufacturing facilities.
Packaging
Pharmaceutical packaging operations are performed with a series of integrated machines and repetitive manual tasks (Gennaro 1990; Swarbick and Boylan 1996). Finished dosage-form products may be packaged in many different types of containers (e.g., plastic or glass bottles, foil blister packs, pouches or sachets, tubes and sterile vials). The mechanical equipment fills, caps, labels, cartons and packs the finished products in shipping containers. Worker proximity to packaging equipment necessitates barrier guarding on moving machine parts, accessible control switches and emergency stop cables and employee training on machine hazards and safe work practices. Enclosure and isolation of equipment reduces sound and vibration levels in packaging areas. Use of hearing-protective devices (e.g., ear muffs and plugs) reduces worker exposures to noise. Good industrial design promotes the productivity, comfort and safety of employees, by addressing ergonomic hazards from poor body postures, material handling and highly repetitive tasks.
Laboratory operations
Laboratory operations in the pharmaceutical industry are diverse. They may pose biological, chemical and physical hazards, depending upon the specific agents, operations, equipment and work practices employed. Major distinctions exist between labs which conduct scientific research and product and process development and those which evaluate quality assurance and control activities (Swarbick and Boylan 1996). Lab workers may conduct scientific research to discover drug substances, develop manufacturing processes for bulk chemical and dosage-form products or analyze raw materials, intermediates and finished products. Lab activities should be evaluated individually, although good lab practices apply to many situations (National Research Council 1981). Clearly defined responsibilities, training and information, safe work practices and control measures and emergency response plans are important means for effectively managing environmental, health and safety hazards.
The health and safety hazards of flammable and toxic materials are reduced by minimizing their inventories in labs and storing them in separate cabinets. Lab assays and operations which may release air contaminants can be performed in ventilated exhaust fume hoods to protect workers. Biological safety hoods provide downward and inward laminar flow, preventing the release of micro-organisms (Gennaro 1990; Swarbick and Boylan 1996). Worker training and information describes the hazards of lab work, safe work practices and proper emergency response to fires and spills. Food and beverages should not be consumed in lab areas. Lab safety is enhanced by requiring supervisors to approve and manage highly hazardous operations. Good lab practices separate, treat and dispose of biological and chemical wastes. Physical hazards (e.g., radiation and electromagnetic energy sources) are often certified and operated, according to specific regulations.
General Health and Safety Hazards
Ergonomics and material handling
The materials shipped, stored, handled, processed and packaged in the pharmaceutical industry range from large quantities of raw materials to small packages containing pharmaceutical products. Raw materials for bulk chemical production are shipped in bulk containers (e.g., tank trucks, rail cars), metal and fibre drums, reinforced paper and plastic bags. Pharmaceutical production uses smaller quantities of raw materials due to the reduced scale of the operations. Material-handling devices (e.g., fork-lift trucks, pallet lifts, vacuum hoists and drum jacks) assist material handling during warehousing and production operations. Heavy manual work may create ergonomic risks when moving materials and equipment if mechanical devices are not available. Good industrial engineering and facility management practices reduce injuries from material handling by improving the design and features of equipment and the workplace and decreasing the size and weight of containers (Cole 1990). Engineering control measures (e.g., ergonomic design of tools, materials and equipment) and administrative practices (e.g., rotating workers, providing worker training) reduce the risks of cumulative trauma injuries during highly repetitive production and packaging operations.
Machine guarding and control of hazardous energy
Unguarded moving machine parts in pharmaceutical manufacturing and packaging equipment create mechanical hazards. Exposed “crush and nip points” in open equipment may seriously injure workers. Mechanical hazards are exacerbated by the large numbers and different designs of equipment, crowded workplace conditions and frequent interactions between workers and equipment. Interlocked guards, control switches, emergency stop devices and operator training are important means of reducing mechanical hazards. Loose hair, long-sleeved clothing, jewellery or other objects may become trapped in equipment. Routine inspection and repair activities identify and control mechanical hazards during production and packaging operations. Hazardous electrical, pneumatic and thermal energy must be released or controlled before working on active equipment and utilities. Workers are protected from sources of hazardous energy by implementing lockout/tagout procedures.
Noise exposures
High sound levels may be generated by manufacturing equipment and utilities (e.g., compressed air, vacuum sources and ventilation systems). Due to the enclosed design of pharmaceutical workplace modules, workers are often located close to machines during manufacturing and packaging operations. Workers observe and interact with production and packaging equipment, thereby increasing their exposure to noise. Engineering methods reduce sound levels by modifying, enclosing and dampening noise sources. Employee rotation and use of hearing-protective devices (e.g., ear muffs and plugs) reduce workers’ exposure to high noise levels. Comprehensive hearing conservation programmes identify noise sources, reduce workplace sound levels, and train workers on the hazards of noise exposure and proper use of hearing-protective devices. Noise monitoring and medical surveillance (i.e., audiometry) assess worker exposures to noise and their resulting loss of hearing. This helps to identify noise problems and evaluate the adequacy of corrective measures.
Solvent vapour and potent compound exposures
Special concerns may arise when workers are exposed to toxic solvent vapours and potent drugs as airborne dusts. Worker exposures to solvent vapours and potent compounds may occur during various manufacturing operations, which need to be identified, evaluated and controlled to ensure that workers are protected. Engineering controls are the preferred means of controlling these exposures, due to their inherent effectiveness and reliability (Cole 1990; Naumann et al. 1996). Enclosed process equipment and material handling systems prevent worker exposures, while LEV and PPE supplement these measures. Increased facility and process containment is needed for controlling highly toxic solvents (e.g., benzene, chlorinated hydrocarbons, ketones) and potent compounds. Positive-pressure respirators (e.g., powered-air purifying and supplied-air) and PPE are needed when highly toxic solvents and potent compounds are handled and processed. Special concerns are posed by operations where high levels of solvent vapours (e.g., compounding, granulating and tablet coating) and dusts (e.g., drying, milling and blending) are generated. Locker and shower rooms, decontamination practices and good sanitary practices (e.g., washing and showering) are necessary to prevent or minimize the effects of worker exposures inside and outside the workplace.
Process safety management
Process safety programmes are implemented in the pharmaceutical industry due to the complex chemistry, hazardous materials and operations in bulk chemical manufacturing (Crowl and Louvar 1990). Highly hazardous materials and processes may be employed in multi-step organic synthesis reactions to produce the desired drug substance. The thermodynamics and kinetics of these chemical reactions must be evaluated, since they may involve highly toxic and reactive materials, lachrymators and flammable or explosive compounds.
Process safety management involves conducting physical hazard testing of materials and reactions, performing hazard analysis studies to review the process chemistry and engineering practices, examining preventive maintenance and mechanical integrity of the process equipment and utilities, implementing worker training and developing operating instructions and emergency response procedures. Special engineering features for process safety include selecting proper pressure-rated vessels, installing isolation and suppression systems, and providing pressure relief venting with catch tanks. Process safety management practices are similar in the pharmaceutical and chemical industries when manufacturing bulk pharmaceuticals as speciality organic chemicals (Crowl and Louvar 1990; Kroschwitz 1992).
Environmental Issues
The different pharmaceutical manufacturing processes each have their own environmental issues, as discussed below.
Fermentation
Fermentation generates large volumes of solid waste which contains mycelia and spent filter cakes (EPA 1995; Theodore and McGuinn 1992). Filter cakes contain mycelia, filter media and small amounts of nutrients, intermediates and residual products. These solid wastes are typically non-hazardous, yet they may contain solvents and small amounts of residual chemicals depending upon the specific chemistry of the fermentation process. Environmental problems may develop if fermentation batches become infected with a viral phage which attacks the micro-organisms in the fermentation process. Although phage infections are rare, they create a significant environmental problem by generating large amounts of waste broth.
Spent fermentation broth contains sugars, starches, proteins, nitrogen, phosphates and other nutrients with high biochemical oxygen demand (BOD), chemical oxygen demand (COD) and total suspended solids (TSS) with pH values ranging from 4 to 8. Fermentation broths can be treated by microbiological wastewater systems, after the effluent is equalized to promote the stable operation of the treatment system. Steam and small amounts of industrial chemicals (e.g., phenols, detergents and disinfectants) maintain the sterility of the equipment and products during fermentation. Large volumes of moist air are exhausted from fermentors, containing carbon dioxide and odours which may be treated before they are emitted to the atmosphere.
Organic synthesis
Wastes from chemical synthesis are complex due to the variety of hazardous materials, reactions and unit operations (Kroschwitz 1992; Theodore and McGuinn 1992). Organic synthesis processes may generate acids, bases, aqueous or solvent liquors, cyanides and metal wastes in liquid or slurry form. Solid wastes may include filter cakes containing inorganic salts, organic by-products and metal complexes. Waste solvents in organic synthesis are usually recovered by distillation and extraction. This allows the solvents to be reused by other processes and reduces the volume of liquid hazardous wastes to be disposed of. Residues from distillation (still bottoms) need to be treated before they are disposed. Typical treatment systems include steam stripping to remove solvents, followed by microbiological treatment of other organic substances. Volatile organic and hazardous substance emissions during organic synthesis operations should be controlled by air pollution control devices (e.g., condensers, scrubbers, venturi impingers).
Waste water from synthesis operations may contain aqueous liquors, wash water, discharges from pumps, scrubbers and cooling systems, and fugitive leaks and spills (EPA 1995). This waste water may contain many organic and inorganic substances with different chemical compositions, toxicities and biodegradabilities. Trace amounts of raw materials, solvents and by-products may be present in aqueous mother liquors from crystallizations and wash layers from extractions and equipment cleaning. These waste waters are high in BOD, COD and TSS, with varying acidity or alkalinity and pH values ranging from 1 to 11.
Biological and natural extraction
Spent raw materials and solvents, wash water and spills are the primary sources of solid and liquid wastes (Theodore and McGuinn 1992). Organic and inorganic chemicals may be present as residues in these waste streams. Usually, waste waters have low BOD, COD and TSS, with relatively neutral pH values ranging from 6 to 8.
Pharmaceutical manufacturing of dosage forms
Pharmaceutical manufacturing of dosage-form products generates solid and liquid wastes during cleaning and sterilization, and from leaks and spills and rejected products (Theodore and McGuinn 1992). Drying, milling and blending operations generate atmospheric and fugitive dust emissions. These emissions can be controlled and recycled to the manufacturing of dosage form products; however, quality control practices may prevent this if other residues are present. When solvents are used during wet granulation, compounding and tablet coating, VOCs and hazardous air pollutants may be released to the atmosphere or in the workplace as process or fugitive emissions. Waste waters may contain inorganic salts, sugars, syrups and traces of drug substances. These waste waters usually have low BOD, COD and TSS, with neutral pH values. Some antiparasitic or anti-infective drugs for humans and animals may be toxic to aquatic organisms, requiring special treatment of liquid wastes.
Environmental pollution prevention
Waste minimization and pollution prevention
Good engineering and administrative practices minimize the environmental impact of bulk chemical production and pharmaceutical manufacturing operations. Pollution prevention employs modifying processes and equipment, recycling and recovering materials and maintaining good housekeeping and operating practices (Theodore and McGuinn 1992). These activities enhance the management of environmental issues, as well as worker health and safety.
Process modifications
Processes may be modified to reformulate products by using materials that are less hazardous or persistent or changing manufacturing operations to reduce air emissions, liquid effluents and solid wastes. Reducing the amount and toxicity of wastes is wise, since it improves the efficiency of manufacturing processes and reduces the costs and impacts of waste disposal. Government drug approval regulations may limit the ability of pharmaceutical manufacturers to change hazardous materials, manufacturing processes, equipment and facilities (Spilker 1994). Drug manufacturers must anticipate the environmental, health and safety impacts of selecting hazardous materials and designing manufacturing process at an early stage. It becomes increasingly difficult to make changes during the later stages of drug development and regulatory approval, without considerable loss of time and expense.
It is very desirable to develop manufacturing processes with less hazardous solvents. Ethyl acetate, alcohols and acetone are preferable to highly toxic solvents such as benzene, chloroform and trichloroethylene. Whenever possible, some materials should be avoided due to their physical properties, ecotoxicity or persistence in the environment (e.g., heavy metals, methylene chloride) (Crowl and Louvar 1990). Substituting aqueous washes for solvents during filtrations in bulk chemical production reduces liquid wastes and vapour emissions. Also, substituting aqueous for solvent-based solutions during tablet coating reduces environmental, health and safety concerns. Pollution prevention is promoted by improving and automating process equipment, as well as performing routine calibration, servicing and preventive maintenance. Optimizing organic synthesis reactions increases product yields, often decreasing the generation of wastes. Incorrect or inefficient temperature, pressure and material control systems cause inefficient chemical reactions, creating additional gaseous, liquid and solid wastes.
The following are examples of process modifications in bulk pharmaceutical production (Theodore and McGuinn 1992):
- Minimize the quantities of hazardous materials used and select materials whose wastes can be controlled, recovered and recycled, whenever possible.
- Develop and install systems for recycling raw materials (e.g., solvents), intermediates, wastes and utility materials (e.g., cooling water, heat transfer liquids, lubricants, steam condensate).
- Examine reactants, solvents and catalysts to optimize the efficiency of chemical reactions.
- Modify the design and features of processing equipment to minimize pollution and wastes.
- Improve processes to maximize product yields and desired properties, eliminating additional processing (e.g., re-crystallization, drying and milling).
- Consider using multi-purpose equipment (e.g., reactors, filters and dryers) to reduce pollution and wastes during transfers, cleaning and additional process steps.
- Use appropriate instruments, automated control systems and computer programs to maximize the efficiency of processes and reduce pollution and wastes.
Resource recovery and recycling
Resource recovery uses waste products and reclaims materials during processing by separating waste impurities from desired materials. Solid wastes from fermentation (e.g., mycelia) may be added to animal feeds as a nutritional supplement or as soil conditioners and fertilizers. Inorganic salts may be recovered from chemical liquors produced during organic synthesis operations. Spent solvents are often recycled by separation and distillation. Air emission control devices (e.g., condensers, compression and refrigeration equipment) greatly reduce emissions of volatile organic compounds to the atmosphere (EPA 1993). These devices capture solvent vapours by condensation, enabling the reuse of solvents as raw materials or for cleaning vessels and equipment. Scrubbers neutralize or absorb acid, caustic and soluble gases and vapours, discharging their effluents to waste treatment systems.
Recycled solvents may be reused as media for performing reactions and extractions, and cleaning operations. Different types of solvents should not be mixed, since this reduces their ability to be recycled. Some solvents should be segregated during processing (e.g., chlorinated and non-chlorinated, aliphatic and aromatic, aqueous and flammable solvents). Dissolved and suspended solids are extracted or separated from the solvents, before the solvents are recovered. Laboratory analysis identifies the composition and properties of waste solvents and recycled raw materials. Many new waste prevention and control technologies are being developed for solid, liquid and gaseous wastes.
General housekeeping and operating practices
Written operating procedures, material-handling instructions and waste management practices reduce the generation and improve the treatment of wastes (Theodore and McGuinn 1992). Good operating and housekeeping practices identify specific responsibilities for generating, handling and treating wastes. Training and supervision of operating staff increases their ability to improve and maintain efficient manufacturing and waste management operations. Workers should be trained on the hazards of waste management practices and the proper means of responding to emergency spills, leaks and fugitive emissions. Worker training should address material handling, cleaning or neutralizing wastes and wearing respirators and PPE. Spill and leak detection devices prevent pollution by routinely monitoring production equipment and utilities, identifying and controlling fugitive emissions and leaks. These activities may be successfully integrated with preventive maintenance practices to clean, calibrate, replace and repair equipment that creates pollution.
Written instructions describing normal operating procedures, as well as start-up, shut-down and emergency procedures, prevent pollution and reduce risks to worker health and safety. Careful management of material inventories decreases the excessive purchasing of raw materials and generation of wastes. Computer systems can assist the effective management of plant operations, maintenance practices and material inventories. Automatic weighing, monitoring and alarm systems can be installed to improve the management of materials and equipment (e.g., storage tanks, process equipment and waste treatment systems). Modern instrument and control systems often increase the productivity of operations, reducing pollution and health and safety hazards. Comprehensive pollution prevention programmes examine all wastes generated at a facility and examine the options for eliminating, reducing or treating them. Environmental audits examine the strengths and weaknesses of pollution prevention and waste management programmes, seeking to optimize their performance.
Case Study: Effects of Synthetic Oestrogens on Pharmaceutical Workers: A United States Example
Background
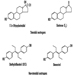
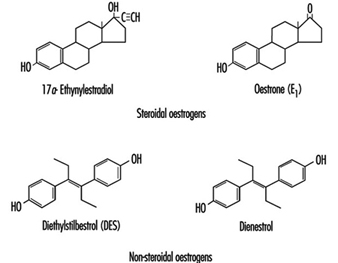
Oestrogens used in the pharmaceutical industry can generally be classified as natural or synthetic and as steroidal or non-steroidal. All steroidal oestrogens, both natural (e.g., oestrone) and synthetic (e.g., ethynyloestradiol and moestranol) have a typical multi-ring structure, as depicted in figure 6 Diethylstilboestrol (DES) and dienoestrol are examples of the non-steroidal oestrogens. The principal uses of oestrogenic compounds are in oral contraceptive tablets and tablets intended for oestrogen replacement therapy. The pure compounds (naturally derived or synthesized) are no longer manufactured in the United States, but are imported.
Figure 1. Examples of steroidal and non-steroidal oestrogen structure
Manufacturing processes
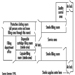
The following description is a generalized, and composite, description of the manufacturing process used in many US pharmaceutical companies. Specific product processes may not follow the flow exactly as described below; some steps may be absent in some processes, and, in other cases, additional steps may be present that are not described here.
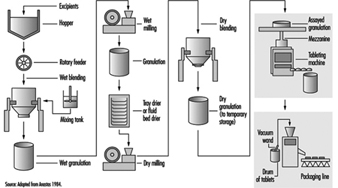
As with most dry-product drugs, pharmaceutical products made from oestrogenic compounds are manufactured in a step-wise batch operation (figure 2). The manufacturing steps begin with the assembly and pre-weighing of both active ingredients and excipients (inactive ingredients) in an isolated room under local exhaust ventilation. When needed, the ingredients are moved to a blending room equipped with mechanical blenders. Excipients are usually loaded dry from a hopper above the blender. The active ingredients are almost always dissolved first in an alcohol, and are added manually or are fed through tubing through the side of the blender. The initial blending of the ingredients is done in a wet state. At the end of the wet blending process, the granulation is typically moved to a wet mill, where particles in the mix are reduced to a specific size. The milled granulation is then dried using a fluid bed drier or is tray-dried in ovens designed for the purpose. The dried granulation may or may not undergo the addition of a lubricant before dry-blending and/or dry-milling, depending on the specific product and process. The final granulation, ready to be made into tablets, is then stored in sealed containers. The raw materials and granulation, and sometimes the intermediate products, are typically sampled and assayed by quality-control personnel prior to being moved to the next process step.
Figure 2. Typical oral contraceptive tablet manufacturing process flow
When needed, the granulation is moved to a compression room, where it is made into tablets by means of a tablet press. The granulation is typically fed from the storage container (typically a plastic-lined fibre drum or a lined stainless steel container) into the tablet press hopper by gravity or pneumatically by means of a vacuum wand. Formed tablets exit from the machine through tubing at the side, and drop into plastic-lined drums. When filled, the drums are sampled and inspected. After assay by quality-control personnel, the drums are sealed, stored and staged for packaging operations. Some tablets also undergo a coating process, in which layers of edible wax and sometimes sugars are used to seal the tablet.
The tablets are packaged by sealing them in blister packs or bottled, depending on the nature of the product. In this process, the containers of tablets are moved to the packaging area. The tablets may be manually scooped into the packaging machine hopper or fed by means of a vacuum wand. The tablets are then either immediately sealed between layers of aluminium foil and plastic film (blister-packaging) or they are bottled. The blister packs or bottles are then conveyed along a line on which they are inspected and placed in pouches or boxed with appropriate inserts.
Health effects on male and female pharmaceutical workers
Reports of occupational exposures and the effects on males have been relatively few, compared with the considerable literature that exists regarding acute and chronic effects of oestrogens in women as a result of non-occupational exposures. The non-occupational literature is primarily a result of widespread contraceptive and other medical uses of oestrogenic pharmaceuticals (but also environmental pollutants with oestrogenic properties, such as the organochlorines) and focuses particularly on the relationships between that exposure and a variety of human cancers, such as that of the endometrium, cervix and breast in women (Hoover 1980; Houghton and Ritter 1995). In the occupational literature, the hyperoestrogenic syndrome in both male and female workers has been associated with exposures to DES and its derivatives, natural or conjugated oestrogens, hexoestrol and its derivatives and steroidal synthetics such as ethynyloestradiol and moestranol. Shortly after the initiation of commercial production of oestrogens, reports began to surface of their effects, such as gynaecomastia (abnormal enlargement of the breasts in a male) and decreased libido among male workers, and menstrual disorders (increased flow or inter-menstrual spotting) among female workers (Scarff and Smith 1942; Fitzsimons 1944; Klavis 1953; Pagani 1953; Watrous 1947; Watrous and Olsen 1959; Pacynski et al. 1971; Burton and Shumnes 1973; Meyer, Peteet and Harrington 1978; Katzenellenbogen 1956; Dunn 1940; Stoppleman and van Valkenburg 1955; Goldzieher and Goldzieher 1949; Fisk 1950). There have also been a few reports of toxicity syndrome associated with some progoestogenic compounds, including acetoxyprogoesterone (Suciu et al. 1973), and vinyloestrenolone in combination with ethynyloestradiol (Gambini, Farine and Arbosti 1976).
A total of 181 cases of hyperoestrogenism in both males and females (occurring over the period 1940–1978) were recorded and reported by company physicians in 10 pharmaceutical companies (13 plant sites) in the United States (Zaebst, Tanaka and Haring 1980). The 13 plant sites included 9 sites manufacturing primarily oral contraceptives containing various synthetic oestrogens and progoestogens, one firm manufacturing oestrogen replacement pharmaceuticals from natural conjugated oestrogens and one firm manufacturing pharmaceuticals from DES (which had in earlier years also synthesized DES).
Investigators from the US National Institute for Occupational Safety and Health (NIOSH) conducted a pilot industrial hygiene and medical study in 1984 of male and female workers in two plants (Tanaka and Zaebst 1984). Measurable exposures were documented to both moestranol and natural conjugated oestrogens, both inside and outside the respiratory protective equipment used. However, no statistically significant changes in oestrogen-stimulated neurophysins (ESN), corticosteroid-binding globulins (CBG), testosterone, thyroid function, blood-clotting factors, liver function, glucose, blood lipids or gonadotropic hormones were noted in these workers. On physical examination, no adverse physical changes were noted in either male or female workers. However, in the plant using moestranol and norethindrone to manufacture oral contraceptive tablets, serum ethynyloestradiol levels appeared to show possible oestrogen exposure and absorption despite the use of respirators. Inside-respirator air samples obtained at this plant suggested less effective workplace protection factors than expected.
Hyperoestrogenic symptoms in males reported in these studies have included nipple sensitivity (manifested as tingling or tenderness of the nipple) or a feeling of pressure in the breast area and, in some cases, breast hyperplasia and gynaecomastia. Additional subjective symptoms reported by some of the male workers also included decreased libido and/or sexual potency. Findings in females included irregular menstruation, nausea, headaches, breast pain, leucorrhoea (thick, whitish discharge from the vagina or cervical canal) and ankle oedema. There have been no long-term follow-up studies in persons occupationally exposed to oestrogens or progoestogens.
Hazards and control of exposure
One of the most serious hazards in the manufacture of oestrogenic pharmaceuticals is inhalation (and to some extent oral ingestion) of the pure active oestrogenic compound during weighing, assembly and quality-assurance testing. However, substantial inhalation of the dry, blended dust (which contains a low percentage of active ingredient) may also occur to workers during granulation, compression and packaging operations. Skin absorption may also occur, particularly during the wet phases of granulation, since alcohol solutions are used. Quality-control and laboratory personnel are also at risk of exposure while sampling, assaying or otherwise handling pure oestrogenic substances, granulation or tablets. Maintenance personnel can be exposed while cleaning, repairing or inspecting mixers, hoppers, mills, vacuum lines and ventilation systems, or changing filters. NIOSH investigators have conducted an in-depth evaluation of engineering controls which have been used during the manufacturing of oral contraceptive tablets (Anastas 1984). This report provides a detailed review of controls and an evaluation of their effectiveness for granulation, milling, material transfers, powder and tablet feed equipment, and general and local exhaust ventilation systems.
The four main elements of hazard control employed in plants using oestrogenic pharmaceuticals are:
- Engineering controls. These include isolation of processing equipment rooms, control of air flow within a facility from least contaminated areas to most contaminated, local exhaust ventilation at any open transfer points, enclosure of machines, sealed process streams and enclosed powder feed systems. Frequently, implementation of engineering controls, such as general or local exhaust ventilation, is complicated by the fact that good manufacturing regulations (such as those required by the US Food and Drug Administration), which are designed to ensure a safe and effective product, conflict with the best health and safety practices. For example, pressure differentials achieved by general ventilation systems, designed to protect workers outside the hazardous process, conflict with the regulatory requirement to prevent contamination of the product by dust or contaminants external to the process. Because it eliminates direct contact between people and the hazardous contaminants, process or equipment containment is often the best option.
- Good work practices. These include separate clean and contaminated locker rooms separated by showers, changes of clothing, washing or showering before exiting contaminated areas and, where it is feasible and appropriate, systematic rotations of all workers between exposed and non-exposed areas. Appropriate training and education regarding the hazards of oestrogens, and good work practices, are an integral part of an effective worker protection programme. The best engineering controls and personal protective equipment can be defeated if the operators are not knowledgeable about the hazards and controls, and if they are not properly trained to take advantage of the controls and to use the personal protective equipment provided.
- Aggressive environmental and medical monitoring of exposed workers. In addition to normally administered physicals, routine screening should, at a minimum, include review for symptoms (breast tenderness, libido change and so on), examinations of the breast and axillary nodes and measurement of areolae. The screening frequency will vary, depending on the severity of the exposure hazard. Of course, medical screening and monitoring (e.g., physical exams, health questionnaires or testing of body fluids) should be implemented with the utmost sensitivity to workers’ overall welfare, their health and their privacy, since their cooperation and assistance in such a programme are critical to its success. Monitoring of worker exposures to the active oestrogenic or progoestogenic substances should be done regularly and should include not only breathing-zone sampling for air contaminants, but also evaluations of skin contamination and the effectiveness of personal protective equipment.
- Use of appropriate personal protective equipment: Personal protective equipment typically includes disposable or launderable coveralls; separate steroid-area shoes, socks, underclothing and rubber gloves; and effective respirators tailored to the degree of hazard. In the most hazardous areas, air-supplied respiratory protective equipment and impervious (to dusts and/or organic solvents) suits may be required.
Because of the potency of the oestrogenic substances, particularly the synthetic ones such as moestranol and ethynyloestradiol, all of these measures are needed to control exposures adequately. The use of personal protective equipment alone may not provide complete protection. Primary reliance should be placed on controlling exposures at the source, by process containment and by isolation.
Monitoring methods
Both high-performance liquid chromatography and radio-immunoassay procedures have been used to determine oestrogens or progoestogens in environmental samples. Serum samples have been analysed for the exogenous active compound, its metabolite (e.g., ethynyloestradiol is the main metabolite of moestranol), oestrogen-stimulated neurophysins or any of a number of other hormones (e.g., gonadotropic hormones and CBGs) considered appropriate for the specific process and hazard. Airborne monitoring usually includes breathing-zone personal monitoring, but area sampling can be useful in detecting departures from expected values over time. Personal monitoring has the advantages of detecting breakdowns or problems with processing equipment, personal protective equipment or ventilation systems and can provide an earlier warning of exposure. Biological monitoring, on the other hand, can detect exposures which may be missed by environmental monitoring (e.g., skin absorption or ingestion). In general, good practice combines both environmental and biological sampling to protect workers.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."