Fire and combustion have been defined in various ways. For our purposes, the most important statements in connection with combustion, as a phenomenon, are as follows:
- Combustion represents a self-sustaining run of reactions consisting of physical and chemical transformations.
- The materials involved enter into reaction with the oxidizing agent in their surroundings, which in most cases is with the oxygen in the air.
- Ignition requires favourable starting conditions, which are generally a sufficient heating up of the system that covers the initial energy demand of the chain reaction of burning.
- The resultant of the reactions are often exothermic, which means that during burning, heat is released and this phenomenon is often accompanied by visibly observable flaming.
Ignition may be considered the first step of the self-sustaining process of combustion. It may occur as piloted ignition (or forced ignition) if the phenomenon is caused by any outer ignition source, or it may occur as auto ignition (or self ignition) if the phenomenon is the result of reactions taking place in the combustible material itself and coupled with heat release.
The inclination to ignition is characterized by an empirical parameter, the ignition temperature (i.e., the lowest temperature, to be determined by test, to which the material has to be heated to for ignition). Depending upon whether or not this parameter is determined—with special test methods—by the use of any ignition source, we distinguish between the piloted ignition temperature and the auto ignition temperature.
In the case of piloted ignition, the energy required for the activation of the materials involved in the burning reaction is supplied by ignition sources. However, there is no direct relationship between the heat quantity needed for ignition and the ignition temperature, because although the chemical composition of the components in the combustible system is an essential parameter of ignition temperature, it is considerably influenced by the sizes and shapes of materials, the pressure of the environment, conditions of air flow, parameters of ignition source, the geometrical features of the testing device, etc. This is the reason for which the data published in literature for autoignition temperature and piloted ignition temperature can be significantly different.
The ignition mechanism of materials in different states may be simply illustrated. This involves examining materials as either solids, liquids or gases.
Most solid materials take up energy from any outer ignition source either by conduction, convection or radiation (mostly by their combination), or are heated up as a result of the heat-producing processes taking place internally that start decomposition on their surfaces.
For ignition to occur with liquids, these must have the formation of a vapour space above their surface that is capable of burning. The vapours released and the gaseous decomposition products mix with the air above the surface of liquid or solid material.
The turbulent flows that arise in the mixture and/or the diffusion help the oxygen to reach the molecules, atoms and free radicals on and above the surface, which are already suitable for reaction. The particles induced enter into interaction, resulting in the release of heat. The process steadily accelerates, and as the chain reaction starts, the material comes to ignition and burns.
The combustion in the layer under the surface of solid combustible materials is called smouldering, and the burning reaction taking place on the interface of solid materials and gas is called glowing. Burning with flames (or flaming) is the process in the course of which the exothermic reaction of burning runs in the gas phase. This is typical for the combustion of both liquid and solid materials.
Combustible gases burn naturally in the gas phase. It is an important empirical statement that the mixtures of gases and air are capable of ignition in a certain range of concentration only. This is valid also for the vapours of liquids. The lower and upper flammable limits of gases and vapours depend on the temperature and pressure of the mixture, the ignition source and the concentration of the inert gases in the mixture.
Ignition Sources
The phenomena supplying heat energy may be grouped into four fundamental categories as to their origin (Sax 1979):
1. heat energy generated during chemical reactions (heat of oxidation, heat of combustion, heat of solution, spontaneous heating, heat of decomposition, etc.)
2. electrical heat energy (resistance heating, induction heating, heat from arcing, electric sparks, electrostatical discharges, heat generated by lightning stroke, etc.)
3. mechanical heat energy (frictional heat, friction sparks)
4. heat generated by nuclear decomposition.
The following discussion addresses the most frequently encountered sources of ignition.
Open flames
Open flames may be the simplest and most frequently used ignition source. A large number of tools in general use and various types of technological equipment operate with open flames, or enable the formation of open flames. Burners, matches, furnaces, heating equipment, flames of welding torches, broken gas and oil pipes, etc. may practically be considered potential ignition sources. Because with an open flame the primary ignition source itself represents an existing self-sustaining combustion, the ignition mechanism means in essence the spreading of burning to another system. Provided that the ignition source with open flame possesses sufficient energy for initiating ignition, burning will start.
Spontaneous ignition
The chemical reactions generating heat spontaneously imply the risk of ignition and burning as “internal ignition sources”. The materials inclined to spontaneous heating and spontaneous ignition may, however, become secondary ignition sources and give rise to ignition of the combustible materials in the surroundings.
Although some gases (e.g., hydrogen phosphide, boron hydride, silicon hydride) and liquids (e.g., metal carbonyls, organometallic compositions) are inclined to spontaneous ignition, most spontaneous ignitions occur as surface reactions of solid materials. Spontaneous ignition, like all ignitions, depends on the chemical structure of the material, but its occurrence is determined by the grade of dispersity. The large specific surface enables the local accumulation of reaction heat and contributes to the increase of temperature of material above spontaneous ignition temperature.
Spontaneous ignition of liquids is also promoted if they come into contact with air on solid materials of large specific surface area. Fats and especially unsaturated oils containing double bonds, when absorbed by fibrous materials and their products, and when impregnated into textiles of plant or animal origin, are inclined to spontaneous ignition under normal atmospheric conditions. Spontaneous ignition of glass-wool and mineral-wool products produced from non-combustible fibres or inorganic materials covering large specific surfaces and contaminated by oil have caused very severe fire accidents.
Spontaneous ignition has been observed mainly with dusts of solid materials. For metals with good heat conductivity, local heat accumulation needed for ignition necessitates very fine crushing of metal. As the particle size decreases, the likelihood of spontaneous ignition increases, and with some metal dusts (for example, iron) pyrophorosity ensues. When storing and handling coal dust, soot of fine distribution, dusts of lacquers and synthetic resins, as well as during the technological operations carried out with them, special attention should be given to the preventive measures against fire to reduce the hazard of spontaneous ignition.
Materials inclined to spontaneous decomposition show special ability to ignite spontaneously. Hydrazine, when set on any material with a large surface area, bursts into flames immediately. The peroxides, which are widely used by the plastics industry, easily decompose spontaneously, and as a consequence of decomposition, they become dangerous ignition sources, occasionally initiating explosive burning.
The violent exothermic reaction that occurs when certain chemicals come into contact with each other may be considered a special case of spontaneous ignition. Examples of such cases are contact of concentrated sulphuric acid with all the organic combustible materials, chlorates with sulphur or ammonium salts or acids, the organic halogen compounds with alkali metals, etc. The feature of these materials to be “unable to bear each other” (incompatible materials) requires special attention particularly when storing and co-storing them and elaborating the regulations of fire-fighting.
It is worth mentioning that such hazardously high spontaneous heating may, in some cases, be due to the wrong technological conditions (insufficient ventilation, low cooling capacity, discrepancies of maintenance and cleaning, overheating of reaction, etc.), or promoted by them.
Certain agricultural products, such as fibrous feedstuffs, oily seeds, germinating cereals, final products of the processing industry (dried beetroot slices, fertilizers, etc.), show an inclination for spontaneous ignition. The spontaneous heating of these materials has a special feature: the dangerous temperature conditions of the systems are exacerbated by some exothermic biological processes that cannot be controlled easily.
Electric ignition sources
Power machines, instruments and heating devices operated by electric energy, as well as the equipment for power transformation and lighting, typically do not present any fire hazard to their surroundings, provided that they have been installed in compliance with the relevant regulations of safety and requirements of standards and that the associated technological instructions have been observed during their operation. Regular maintenance and periodic supervision considerably diminish the probability of fires and explosions. The most frequent causes of fires in electric devices and wiring are overloading, short circuits, electric sparks and high contact resistances.
Overloading exists when the wiring and electrical appliances are exposed to higher current than that for which they are designed. The overcurrent passing through the wiring, devices and equipment might lead to such an overheating that the overheated components of the electrical system become damaged or broken, grow old or carbonize, resulting in cord and cable coatings melting down, metal parts glowing and the combustible structural units coming to ignition and, depending on the conditions, also spreading fire to the environment. The most frequent cause of overloading is that the number of consumers connected is higher than permitted or their capacity exceeds the value stipulated.
The working safety of electrical systems is most frequently endangered by short circuits. They are always the consequences of any damage and occur when the parts of the electrical wiring or the equipment at the same potential level or various potential levels, insulated from each other and the earth, come into contact with each other or with the earth. This contact may arise directly as metal-metal contact or indirectly, through electric arc. In cases of short circuits, when some units of the electric system come in contact with each other, the resistance will be considerably lower, and as a consequence, the intensity of current will be extremely high, perhaps several orders of magnitude lower. The heat energy released during overcurrents with large short circuits might result in a fire in the device affected by the short circuit, with the materials and equipment in the surrounding area coming to ignition and with the fire spreading to the building.
Electric sparks are heat energy sources of a small nature, but as shown by experience, act frequently as ignition sources. Under normal working conditions, most electrical appliances do not release sparks, but the operation of certain devices is normally accompanied by sparks.
Sparking introduces a hazard foremost at places where, in the zone of their generation, explosive concentrations of gas, vapour or dust might arise. Consequently, equipment normally releasing sparks during operation is permitted to be set up only at places where the sparks cannot give rise to fire. On its own, the energy content of sparks is insufficient for the ignition of the materials in the environment or to initiate an explosion.
If an electrical system has no perfect metallic contact between the structural units through which the current flows, high contact resistance will occur at this spot. This phenomenon is in most cases due to the faulty construction of joints or to unworkmanlike installations. The disengagement of joints during operation and natural wear may also be cause for high contact resistance. A large portion of the current flowing through places with increased resistance will transform to heat energy. If this energy cannot be dissipated sufficiently (and the reason cannot be eliminated), the extremely large increase of temperature might lead to a fire condition that endangers the surrounding.
If the devices work on the basis of the induction concept (engines, dynamos, transformers, relays, etc.) and are not properly calculated, eddy currents may arise during operation. Due to the eddy currents, the structural units (coils and their iron cores) might warm up, which might lead to the ignition of insulating materials and the burning of the equipment. Eddy currents might arise—with these harmful consequences—also in the metal structural units around high-voltage equipment.
Electrostatic sparks
Electrostatic charging is a process in the course of which any material, originally with electric neutrality (and independent of any electric circuit) becomes charged positively or negatively. This may occur in one of three ways:
1. charging with separation, such that charges of subtractive polarity accumulate on two bodies simultaneously
2. charging with passing, such that the charges passing away leave charges of opposed polarity signs behind
3. charging by taking up, such that the body receives charges from outside.
These three ways of charging may arise from various physical processes, including separation after contact, splitting, cutting, pulverizing, moving, rubbing, flowing of powders and fluids in pipe, hitting, change of pressure, change of state, photoionization, heat ionization, electrostatical distribution or high-voltage discharge.
Electrostatic charging may occur both on conducting bodies and insulating bodies as a result of any of the processes mentioned above, but in most cases the mechanical processes are responsible for the accumulation of the unwanted charges.
From the large number of the harmful effects and risks due to electrostatic charging and the spark discharge resulting from it, two risks can be mentioned in particular: endangering of electronic equipment (for example, computer for process control) and the hazard of fire and explosion.
Electronic equipment is endangered first of all if the discharge energy from the charging is sufficiently high to cause destruction of the input of any semi-conductive part. The development of electronic units in the last decade has been followed by the rapid increase of this risk.
The development of fire or explosion risk necessitates the coincidence in space and time of two conditions: the presence of any combustible medium and the discharge with ability for ignition. This hazard occurs mainly in the chemical industry. It may be estimated on the basis of the so-called spark sensitivity of hazardous materials (minimum ignition energy) and depends on the extent of charging.
It is an essential task to reduce these risks, namely, the large variety of consequences that extend from technological troubles to catastrophes with fatal accidents. There are two means of protecting against the consequences of electrostatic charging:
1. preventing the initiation of the charging process (it is evident, but usually very difficult to realize)
2. restricting the accumulation of charges to prevent the occurrence of dangerous discharges (or any other risk).
Lightning is an atmospherical electric phenomenon in nature and may be considered an ignition source. The static charging produced in the clouds is equalized towards the earth (lightning stroke) and is accompanied by a high-energy discharge. The combustible materials at the place of lightning stroke and its surroundings might ignite and burn off. At some strokes of lightning, very strong impulses are generated, and the energy is equalized in several steps. In other cases, long-lasting currents start to flow, sometimes reaching the order of magnitude of 10 A.
Mechanical heat energy
Technical practice is steadily coupled with friction. During mechanical operation, frictional heat is developed, and if heat loss is restricted to such an extent that heat accumulates in the system, its temperature may increase to a value that is dangerous for the environment, and fire may occur.
Friction sparks normally occur at metal technological operations because of heavy friction (grinding, chipping, cutting, hitting) or because of metal objects or tools dropping or falling on to a hard floor or during grinding operations because of metal contaminations within the material under grinding impact. The temperature of the spark generated is normally higher than the ignition temperature of the conventional combustible materials (such as for sparks from steel, 1,400-1,500 °C; sparks from copper-nickel alloys, 300-400 °C); however, the ignition ability depends on the whole heat content and the lowest ignition energy of the material and substance to be ignited, respectively. It has been proven in practice that friction sparks mean real fire risk in air spaces where combustible gases, vapours and dusts are present in dangerous concentrations. Thus, under these circumstances the use of materials that easily produce sparks, as well as processes with mechanical sparking, should be avoided. In these cases, safety is provided by tools that do not spark, i.e., made from wood, leather or plastic materials, or by using tools of copper and bronze alloys that produce sparks of low energy.
Hot surfaces
In practice, the surfaces of equipment and devices may warm up to a dangerous extent either normally or due to malfunction. Ovens, furnaces, drying devices, waste-gas outlets, vapour pipes, etc. often cause fires in explosive air spaces. Furthermore, their hot surfaces may ignite combustible materials coming close to them or by coming in contact. For prevention, safe distances should be observed, and regular supervision and maintenance will reduce the probability of the occurrence of dangerous overheating.
Fire Hazards of Materials and Products
The presence of combustible material in combustible systems represents an obvious condition of burning. Burning phenomena and the phases of the burning process fundamentally depend on the physical and chemical properties of the material involved. Therefore, it seems reasonable to make a survey of the flammability of the various materials and products with respect to their character and properties. For this section, the ordering principle for the grouping of materials is governed by technical aspects rather than by theoretical conceptions (NFPA 1991).
Wood and wood-based products
Wood is one of the most common materials in the human milieu. Houses, building structures, furniture and consumer goods are made of wood, and it is also widely used for products such as paper as well as in the chemical industry.
Wood and wood products are combustible, and when in contact with high-temperature surfaces and exposed to heat radiation, open flames or any other ignition source, will carbonize, glow, ignite or burn, depending upon the condition of combustion. To widen the field of their application, the improvement of their combustion properties is required. In order to make structural units produced from wood less combustible, they are typically treated with fire-retardant agents (e.g., saturated, impregnated, provided with surface coating).
The most essential characteristic of combustibility of the various kinds of wood is the ignition temperature. Its value strongly depends on some of the properties of wood and the test conditions of determination, namely, the wood sample’s density, humidity, size and shape, as well as the ignition source, time of exposure, intensity of exposure and the atmosphere during testing. It is interesting to note that the ignition temperature as determined by various test methods differs. Experience has shown that the inclination of clean and dry wood products to ignition is extremely low, but several fire cases caused by spontaneous ignition have been known to occur from storing dusty and oily waste wood in rooms with imperfect ventilation. It has been proven empirically that higher moisture content increases the ignition temperature and reduces the burning speed of wood. The thermal decomposition of wood is a complicated process, but its phases may clearly be observed as follows:
- The thermal decomposition with mass loss starts already in the range 120-200 °C; moisture content releases and the non-combustible degradates occur in the combustion space.
- At 200-280 °C, mainly endothermic reactions occur while the heat energy of ignition source is taken up.
- At 280-500 °C, the exothermic reactions of decomposition products are steadily accelerating as the primary process, while carbonization phenomena may be observed. In this temperature range, sustaining combustion has already developed. After ignition, burning is not steady in time because of the good heat-insulating ability of its carbonized layers. Consequently, the warming up of the deeper layers is limited and time consuming. When the surfacing of the combustible decomposition products is accelerated, burning will be complete.
- At temperatures exceeding 500 °C, the wood char forms residues. During its additional glowing, ash containing solid, inorganic materials is produced, and the process has come to an end.
Fibres and textiles
The majority of the textiles produced from fibrous materials that are found in the close surrounding of people is combustible. Clothing, furniture and the built environment partly or totally consists of textiles. The hazard which they present exists during their production, processing and storing as well as during their wearing.
The basic materials of textiles are both natural and artificial; synthetic fibres are used either alone or mixed with natural fibres. The chemical composition of the natural fibres of plant origin (cotton, hemp, jute, flax) is cellulose, which is combustible, and these fibres have a relatively high ignition temperature (<<400°C). It is an advantageous feature of their burning that when brought to high temperature they carbonize but do not melt. This is especially advantageous for the medical treatments of burn casualties.
The fire hazardous properties of fibres of protein base of animal origin (wool, silk, hair) are even more favourable than those of fibres of plant origin, because a higher temperature is required for their ignition (500-600 °C), and under the same conditions, their burning is less intensive.
The plastics industry, utilizing several extremely good mechanical properties of polymer products, has also gained prominence in the textile industry. Among the properties of acrylic, polyester and the thermoplastic synthetic fibres (nylon, polypropylene, polyethylene), those associated with burning are the least advantageous. Most of them, in spite of their high ignition temperature (<<400-600 °C), melt when exposed to heat, easily ignite, burn intensively, drop or melt when burning and release considerably high quantities of smoke and toxic gases. These burning properties may be improved by addition of natural fibres, producing so-called textiles with mixed fibres. Further treatment is accomplished with flame-retardant agents. For the manufacture of textiles for industrial purposes and heat-protective clothing, inorganic, non-combustible fibre products (including glass and metal fibres) are already used in large quantities.
The most important fire hazard characteristics of textiles are the properties connected with ignitability, flame spread, heat generation and the toxic combustion products. Special testing methods have been developed for their determination. The test results obtained influence the fields of application for these products (tents and flats, furniture, vehicle upholstery, clothes, carpets, curtains, special protective clothing against heat and weather), as well as the stipulations to restrict the risks in their use. An essential task of industrial researchers is to develop textiles that sustain high temperature, treated with fire-retardant agents, (heavily combustible, with long ignition time, low flame spread rate, low speed of heat release) and produce small amounts of toxic combustion products, as well as to improve the unfavourable effect of fire accidents due to the burning of such materials.
Combustible and flammable liquids
In the presence of ignition sources, combustible and flammable liquids are potential sources of risk. First, the closed or open vapour space above such liquids provides a fire and explosion hazard. Combustion, and more frequently explosion, might occur if the material is present in the vapour-air mixture in suitable concentration. From this it follows that burning and explosion in the zone of combustible and flammable liquids may be prevented if:
- the ignition sources, air, and oxygen are excluded; or
- instead of oxygen, inert gas is present in the surrounding; or
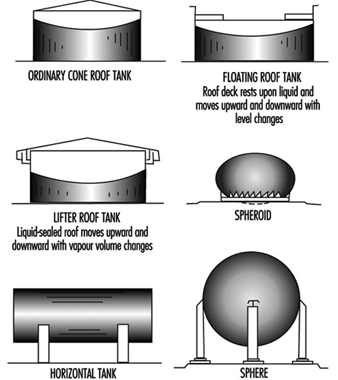
- the liquid is stored in a closed vessel or system (see Figure 1); or
- by proper ventilation, the development of the dangerous vapour concentration is prevented.
Figure 1. Common types of tanks for storage of flammable and combustible liquids.
In practice, a large number of material characteristics are known in connection with the dangerous nature of combustible and flammable liquids. These are closed-cup and open-cup flash points, boiling point, ignition temperature, rate of evaporation, upper and lower limits of the concentration for combustibility (flammable or explosive limits), the relative density of vapours compared to air and energy required for the ignition of vapours. These factors provide full information about the sensitivity for ignition of various liquids.
Nearly all over the world the flash point, a parameter determined by standard test under atmospherical conditions, is used as the basis to group the liquids (and materials behaving as liquids at relatively low temperatures) into categories of risk. The safety requirements for storage of liquids, their handling, the technological processes, and the electrical equipment to be set up in their zone should be elaborated for each category of flammability and combustibility. The zones of risk around the technological equipment should also to be identified for each category. Experience has shown that fire and explosion might occur—depending on the temperature and pressure of the system—within the range of concentration between the two flammable limits.
Gases
Although all materials—under a specific temperature and pressure—may become gases, the materials considered gaseous in practice are those that are in a gas state at normal temperature (~20 °C) and normal atmospheric pressure (~100 kPa).
In respect to fire and explosion hazards, gases may be ranked in two main groups: combustible and non-combustible gases. According to the definition accepted in practice, combustible gases are those that burn in air with normal oxygen concentration, provided that the conditions required for burning exist. Ignition only occurs above a certain temperature, with the necessary ignition temperature, and within a given range of concentration.
Non-combustible gases are those that do not burn either in oxygen or in air with any concentration of air. A portion of these gases support combustion (e.g., oxygen), while the other portion inhibit burning. The non-combustible gases not supporting burning are called inert gases (nitrogen, noble gases, carbon dioxide, etc.).
In order to achieve economic efficiency, the gases stored and transported in containers or transporting vessels are typically in compressed, liquefied, or cooled-condensated (cryogenic) state. Basically, there are two hazardous situations in connection with gases: when they are in containers and when they are released from their containers.
For compressed gases in storage containers, external heat might considerably increase the pressure within the container, and the extreme overpressure might lead to explosion. Gaseous storage containers will typically include a vapour phase and a liquid phase. Because of changes in pressure and temperature, the extension of the liquid phase gives rise to the further compression of vapour space, while the vapour pressure of the liquid increases in proportion with the increase of temperature. As a result of these processes, critically dangerous pressure may be produced. Storage containers are generally required to contain the application of overpressure relief devices. These are capable of mitigating a hazardous situation due to higher temperatures.
If the storage vessels are insufficiently sealed or damaged, the gas will flow out to the free air space, mix with air and depending on its quantity and the way of its flowing, may cause the formation of a large, explosive air space. The air around a leaking storage vessel can be unsuitable for breathing and may be dangerous for people nearby, partly due to the toxic effect of some gases and partly due to the diluted concentration of oxygen.
Bearing in mind the potential fire hazard due to gases and the need for safe operation, one must get detailed knowledge of the following features of gases either stored or used, especially for industrial consumers: the chemical and physical properties of gases, ignition temperature, the lower and upper limits of concentration for flammability, the hazardous parameters of the gas in the container, the risk factors of the hazardous situation caused by the gases released into the open air, the extent of the necessary safety zones and the special measures to be taken in case of a possible emergency situation connected with fire-fighting.
Chemicals
Knowledge of the hazardous parameters of chemicals is one of the basic conditions of safe working. The preventive measures and requirements for protection against fire may be elaborated only if the physical and chemical properties connected with fire hazard are taken into consideration. Of these properties, the most important ones are the following: combustibility; ignitability; ability to react with other materials, water or air; inclination to corrosion; toxicity; and radioactivity.
Information on the properties of chemicals can be obtained from the technical data sheets issued by manufacturers and from the manuals and handbooks containing the data of hazardous chemicals. These provide users with information not only about the general technical features of materials, but also about the actual values of hazard parameters (decomposition temperature, ignition temperature, limit concentrations of combustion, etc.), their special behaviour, requirements for storage and fire-fighting, as well as recommendations for first aid and medical therapy.
The toxicity of chemicals, as potential fire hazard, may act in two ways. First, the high toxicity of certain chemicals themselves, may be hazardous in a fire. Second, their presence within the fire zone may effectively restrict fire-fighting operations.
The oxidizing agents (nitrates, chlorates, inorganic peroxides, permanganates, etc.), even if they themselves are non-combustible, largely contribute to the ignition of combustible materials and to their intensive, occasionally explosive burning.
The group of unstable materials includes the chemicals (acetaldehyde, ethylene oxide, organic peroxides, hydrogen cyanide, vinyl chloride) which polymerize or decompose in violent exothermic reactions spontaneously or very easily.
The materials sensitive to water and air are extremely dangerous. These materials (oxides, hydroxides, hydrides, anhydrides, alkali metals, phosphorus, etc.) interact with the water and air that are always present in the normal atmosphere, and start reactions accompanied by very high heat generation. If they are combustible materials, they will come to spontaneous ignition. However, the combustible components that initiate the burning may possibly explode and spread to the combustible materials in the surrounding area.
The majority of corrosive materials (inorganic acids—sulphuric acid, nitric acid, perchloric acid, etc.—and halogens —fluorine, chlorine, bromine, iodine) are strong oxidizing agents, but at the same time they have very strong destructive effects on living tissues, and therefore special measures have to be taken for fire-fighting.
The dangerous characteristic of radioactive elements and compounds is increased by the fact that the radiation emitted by them may be harmful in several ways, besides that such materials may be fire hazards themselves. If in a fire the structural containment of the radioactive objects involved becomes damaged, λ-radiating materials might be released. They can have a very strong ionizing effect, and are capable of the fatal destruction of living organisms. Nuclear accidents can be accompanied by fires, the decomposition products of which bind radioactive (α-and β-radiating) contaminants by adsorption. These may cause permanent injuries to the persons taking part in rescue operations if they penetrate into their bodies. Such materials are extremely dangerous, because the persons affected do not perceive any radiation by their sensing organs, and their general state of health does not seem to be any worse. It is obvious that if radioactive materials burn, the radioactivity of the site, the decomposition products and the water used for fire-fighting should be kept under constant observation by means of radioactive signalling devices. The knowledge of these factors has to be taken into account for the strategy of intervention and all additional operations. The buildings for handling and storing radioactive materials as well as for their technological use need to be built of non-combustible materials of high fire resistance. At the same time, high-quality, automatic equipment for detecting, signalling and extinguishing a fire should be provided.
Explosives and blasting agents
Explosive materials are used for many military and industrial purposes. These are chemicals and mixtures which, when affected by strong mechanical force (hitting, shock, friction) or starting ignition, suddenly transform to gases of large volume through an extremely rapid oxidizing reaction (e.g., 1,000-10,000 m/s). The volume of these gases is the multiple of the volume of the explosive material already exploded, and they will exert very high pressure on the surroundings. During an explosion, high temperatures can arise (2,500-4,000 °C) that promote the ignition of the combustible materials in the zone of explosion.
Manufacture, transport and storage of the various explosive materials are governed by rigorous requirements. An example is NFPA 495, Explosive Materials Code.
Besides the explosive materials used for military and industrial purposes, the inductive blasting materials and pyrotechnical products are also treated as hazards. In general, mixtures of explosive materials are often used (picric acid, nitroglycerin, hexogene, etc.), but mixtures of materials capable of explosion are also in use (black powder, dynamite, ammonium nitrate, etc.). In the course of acts of terrorism, plastic materials have become well-known, and are, in essence, mixtures of brisant and plasticizing materials (various waxes, Vaseline, etc.).
For explosive materials, the most effective method of protection against fire is the exclusion of ignition sources from the surroundings. Several explosive materials are sensitive to water or various organic materials with an ability to oxidate. For these materials, the requirements for the conditions of storage and the rules for storing in the same place together with other materials should be carefully considered.
Metals
It is known from practice that nearly all the metals, under certain conditions, are capable of burning in atmospheric air. Steel and aluminium in large structural thickness, on the basis of their behaviour in fire, are clearly evaluated as non-combustible. However, the dusts of aluminium, iron in fine distribution and metal cottons from thin metal fibres can easily be ignited and thus burn intensively. The alkali metals (lithium, sodium, potassium), the alkaline-earth metals (calcium, magnesium, zinc), zirconium, hafnium, titanium, etc. ignite extremely easily in the form of a powder, filings or thin bands. Some metals have such a high sensitivity that they are stored separately from air, in inert gas atmospheres or under a liquid that is neutral for the metals.
The combustible metals and those that are conditioned to burn produce extremely violent burning reactions that are high-speed oxidation processes releasing considerably higher quantities of heat than observed from the burning of combustible and flammable liquids. The burning of metal dust in the case of settled powder, following the preliminary phase of glowing-ignition, might grow to rapid burning. With stirred-up dusts and clouds of dusts that might result, severe explosions can occur. The burning activity and affinity for oxygen of some metals (such as magnesium) are so high that after being ignited they will continue to burn in certain media (e.g., nitrogen, carbon dioxide, steam atmosphere) that are used for extinguishing fires derived from combustible solid materials and liquids.
Extinguishing metal fires presents a special task for fire-fighters. The choice of the proper extinguishing agent and the process in which it is applied are of great importance.
Fires of metals may be controlled with very early detection, the rapid and appropriate action of fire-fighters using the most effective method and, if possible, removal of metals and any other combustible materials from the zone of burning or at least a reduction of their quantities.
Special attention should be given to the protection against radiation when radioactive metals (plutonium, uranium) burn. Preventive measures have to be taken to avoid the penetration of toxic decomposition products into living organisms. For example, alkali metals, because of their ability to react violently with water may be extinguished with dry fire-extinguishing powders only. Burning of magnesium cannot be extinguished with water, carbon dioxide, halons or nitrogen with good success, and more important, if these agents are used in fire-fighting, the hazardous situation will become even more severe. The only agents that can be applied successfully are the noble gases or in some cases boron trifluoride.
Plastics and rubber
Plastics are macromolecular organic compounds produced synthetically or by modification of natural materials. The structure and shape of these macromolecular materials, produced by polymerizational, polyadditional or polycondensational reactions, will strongly influence their properties. The chain molecules of thermoplastics (polyamides, polycarbonates, polyesters, polystyrene, polyvinyl chloride, polymethyl-metacrylate, etc.) are linear or branched, the elastomers (neoprene, polysulphides, isoprene, etc.) are lightly cross-linked, while thermosetting plastics (duroplastics: polyalkydes, epoxy resins, polyurethanes, etc.) are densely cross-linked.
Natural caoutchouc is used as raw material by the rubber industry, and after being vulcanized, rubber is produced. The artificial caoutchoucs, the structure of which is similar to that of natural chaoutchouc, are polymers and co-polymers of butadiene.
The range of products from plastics and rubber used in nearly all fields of everyday life is steadily widening. Use of the large variety and excellent technical properties of this group of materials results in items such as various building structures, furniture, clothes, commodities, parts for vehicles and machines.
Typically, as organic materials, plastics and rubber also are considered to be combustible materials. For the description of their fire behaviour, a number of parameters are used that can be tested by special methods. With the knowledge of these parameters, one can allocate the fields of their application (determined, pointed out, set), and the fire safety provisions can be elaborated. These parameters are combustibility, ignitability, ability to develop smoke, inclination to produce toxic gases and burning dripping.
In many cases the ignition temperature of plastics is higher than that of wood or any other materials, but in most cases they ignite more easily, and their burning takes place more rapidly and with higher intensity. Fires of plastics are often accompanied by the unpleasant phenomena of large quantities of dense smoke being released that can strongly restrict visibility and develop various toxic gases (hydrochloric acid, phosgene, carbon monoxide, hydrogen cyanide, nitrous gases, etc.). Thermoplastic materials melt during burning, then flow and depending on their location (if mounted in or on a ceiling) produce drops which remain in the burning area and might ignite the combustible materials underneath.
The improvement of burning properties represents a complex problem and a “key issue” of plastics chemistry. Fire-retardant agents inhibit combustibility, ignition will be slower, the rate of combustion will fall, and flame propagation will slow down. At the same time, the quantity and optical density of smoke will be higher and the gas mixture produced will be more toxic.
Dusts
With regard to physical state, dusts belong to the solid materials, but their physical and chemical properties differ from those of those same materials in compact form. It is known that industrial accidents and catastrophes are caused by dust explosions. Materials that are non-combustible in their usual form, such as metals, may initiate an explosion in the form of dust mixed with air when affected by any ignition source, even of low energy. The hazard of an explosion also exists with dusts of combustible materials.
Dust can be an explosion hazard not only when floating in the air, but also when settled. In layers of dust, heat may accumulate, and slow burning may develop in the inside as a result of the increased ability of particles to react and their lower thermal conductivity. Then the dust may be stirred up by flashes, and the possibility of dust explosion will grow.
Floating particles in fine distribution present a more severe hazard. Similar to the explosion properties of combustible gases and vapours, dusts also have a special range of air-dust concentration in which an explosion may occur. The lower and upper limit values of explosion concentration and the width of concentration range depend on the size and distribution of particles. If the dust concentration exceeds the highest concentration leading to an explosion, a portion of the dust is not destroyed by fire and absorbs heat, and as a consequence the explosion pressure developed remains below the maximum. The moisture content of air also influences the occurrence of an explosion. At higher humidity, the ignition temperature of the cloud of dust will increase in proportion with the heat quantity necessary for the evaporation of humidity. If an inert foreign dust is mixed in a cloud of dust, the explosivity of the dust-air mixture will be reduced. The effect will be the same if inert gases are mixed in the mixture of air and dust, because the oxygen concentration necessary for burning will be lower.
Experience has shown that all the ignition sources, even of minimum ignition energy, are capable of igniting dust clouds (open flames, electric arc, mechanical or electrostatic spark, hot surfaces, etc.). According to test results obtained in laboratory, the energy demand for ignition of dust clouds is 20 to 40 times higher than in the case of mixtures of combustible vapour and air.
The factors that influence the explosion hazard for settled dusts are the physical and thermal engineering properties of the dust layer, the glowing temperature of the dust and the ignition properties of the decomposition products released by the dust layer.