When a person is exposed to warm environmental conditions the physiological heat loss mechanisms are activated in order to maintain normal body temperature. Heat fluxes between the body and the environment depend on the temperature difference between:
- the surrounding air and objects like walls, windows, the sky, and so on
- the surface temperature of the person
The surface temperature of the person is regulated by physiological mechanisms, such as variations in the blood flow to the skin, and by evaporation of sweat secreted by the sweat glands. Also, the person can change clothing to vary the heat exchange with the environment. The warmer the environmental conditions, the smaller the difference between surrounding temperatures and skin or clothing surface temperature. This means that the “dry heat exchange” by convection and radiation is reduced in warm compared to cool conditions. At environmental temperatures above the surface temperature, heat is gained from the surroundings. In this case this extra heat together with that liberated by the metabolic processes must be lost through evaporation of sweat for the maintenance of body temperature. Thus evaporation of sweat becomes more and more critical with increasing environmental temperature. Given the importance of sweat evaporation it is not surprising that wind velocity and air humidity (water vapour pressure) are critical environmental factors in hot conditions. If the humidity is high, sweat is still produced but evaporation is reduced. Sweat which cannot evaporate has no cooling effect; it drips off and is wasted from a thermoregulatory point of view.
The human body contains approximately 60% water, about 35 to 40 l in an adult person. About one-third of the water in the body, the extracellular fluid, is distributed between the cells and in the vascular system (the blood plasma). The remaining two-thirds of the body water, the intracellular fluid, is located inside the cells. The composition and the volume of the body water compartments is very precisely controlled by hormonal and neural mechanisms. Sweat is secreted from the millions of sweat glands on the skin surface when the thermoregulatory centre is activated by an increase in body temperature. The sweat contains salt (NaCl, sodium chloride) but to a lesser extent than the extracellular fluid. Thus, both water and salt are lost and must be replaced after sweating.
Effects of Sweat Loss
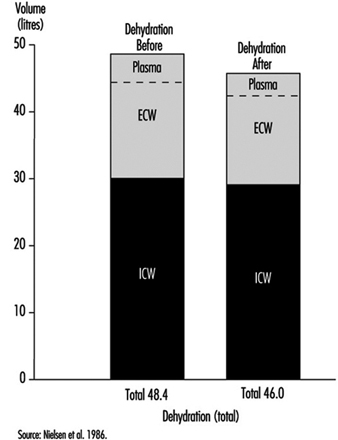
In neutral, comfortable, environmental conditions, small amounts of water are lost by diffusion through the skin. However, during hard work and in hot conditions, large quantities of sweat can be produced by active sweat glands, up to more than 2 l/h for several hours. Even a sweat loss of only 1% of body weight (» 600 to 700 ml) has a measurable effect on the ability to perform work. This is seen by a rise in heart rate (HR) (HR increases about five beats per minute for each per cent loss of body water) and a rise in body core temperature. If work is continued there is a gradual increase in body temperature, which can rise to a value around 40ºC; at this temperature, heat illness may result. This is partly due to the loss of fluid from the vascular system (figure 1). A loss of water from the blood plasma reduces the amount of blood which fills the central veins and the heart. Each heart beat will therefore pump a smaller stroke volume. As a consequence the cardiac output (the amount of blood which is expelled by the heart per minute) tends to fall, and the heart rate must increase in order to maintain the circulation and the blood pressure.
Figure 1. Calculated distributions of water in the extracellular compartment (ECW) and intracellular compartment (ICW) before and after 2 h of exercise dehydration at 30°C room temperature.
A physiological control system called the baroreceptor reflex system maintains the cardiac output and blood pressure close to normal under all conditions. The reflexes involve receptors, sensors in the heart and in the arterial system (aorta and carotid arteries), which monitor the degree of stretching of the heart and vessels by the blood which fills them. Impulses from these travel through nerves to the central nervous system, from which adjustments, in case of dehydration, cause a constriction in the blood vessels and a reduction in blood flow to splanchnic organs (liver, gut, kidneys) and to the skin. In this way the available blood flow is redistributed to favour circulation to the working muscles and to the brain (Rowell 1986).
Severe dehydration may lead to heat exhaustion and circulatory collapse; in this case the person cannot maintain the blood pressure, and fainting is the consequence. In heat exhaustion, symptoms are physical exhaustion, often together with headache, dizziness and nausea. The main cause of heat exhaustion is the circulatory strain induced by water loss from the vascular system. The decline in blood volume leads to reflexes which reduce circulation to the intestines and the skin. The reduction in skin blood flow aggravates the situation, since heat loss from the surface decreases, so the core temperature increases further. The subject may faint due to a fall in blood pressure and the resulting low blood flow to the brain. The lying position improves the blood supply to the heart and brain, and after cooling and having some water to drink the person regains his or her well-being almost immediately.
If the processes causing the heat exhaustion “run wild”, it develops into heat stroke. The gradual reduction in skin circulation makes the temperature rise more and more, and this leads to a reduction, even a stop in sweating and an even faster rise in core temperature, which causes circulatory collapse and may result in death, or irreversible damage to the brain. Changes in the blood (such as high osmolality, low pH, hypoxia, cell adherence of the red blood cells, intravascular coagulation) and damage to the nervous system are findings in heat stroke patients. The reduced blood supply to the gut during heat stress can provoke tissue damage, and substances (endotoxins) may be liberated which induce fever in connection with heat stroke (Hales and Richards 1987). Heat stroke is an acute, life-threatening emergency further discussed in the section on “heat disorders”.
Together with water loss, sweating produces a loss of electrolytes, mainly sodium (Na+) and chloride (Cl–), but also to a lesser degree magnesium (Mg++), potassium (K+) and so on (see table 1). The sweat contains less salt than the body fluid compartments. This means that they become more salty after sweat loss. The increased saltiness seems to have a specific effect on the circulation via effects on vascular smooth muscle, which controls the degree to which the vessels are open. However, it is shown by several investigators to interfere with the ability to sweat, in such a way that it takes a higher body temperature to stimulate the sweat glands—the sensitivity of the sweat glands becomes reduced (Nielsen 1984). If the sweat loss is replaced only by water, this may lead to a situation where the body contains less sodium chloride than in the normal state (hypo-osmotic). This will cause cramps due to the malfunction of nerves and muscles, a condition known in earlier days as “miner’s cramps” or “stoker’s cramps”. It can be prevented by addition of salt to the diet (drinking beer was a suggested preventive measure in the UK in the 1920s!).
Table 1. Electrolyte concentration in blood plasma and in sweat
|
Electrolytes and other |
Blood plasma concen- |
Sweat concentrations |
|
Sodium (Na+) |
3.5 |
0.2–1.5 |
|
Potassium (K+) |
0.15 |
0.15 |
|
Calcium (Ca++) |
0.1 |
small amounts |
|
Magnesium (Mg++) |
0.02 |
small amounts |
|
Chloride (Cl–) |
3.5 |
0.2–1.5 |
|
Bicarbonate (HCO3–) |
1.5 |
small amounts |
|
Proteins |
70 |
0 |
|
Fats, glucose, small ions |
15–20 |
small amounts |
Adapted from Vellar 1969.
The decreased skin circulation and sweat gland activity both affect thermoregulation and heat loss in such a way that core temperature will increase more than in the fully hydrated state.
In many different trades, workers are exposed to external heat stress—for example, workers in steel plants, glass industries, paper mills, bakeries, mining industries. Also chimney sweeps and firefighters are exposed to external heat. People who work in confined spaces in vehicles, ships and aircraft may also suffer from heat. However, it must be noted that persons working in protective suits or doing hard work in waterproof clothes can be victims of heat exhaustion even in moderate and cool environmental temperature conditions. Adverse effects of heat stress occur in conditions where the core temperature is elevated and the sweat loss is high.
Rehydration
The effects of dehydration due to sweat loss may be reversed by drinking enough to replace the sweat. This will usually take place during recovery after work and exercise. However, during prolonged work in hot environments, performance is improved by drinking during activity. The common advice is thus to drink when thirsty.
But, there are some very important problems in this. One is that the urge to drink is not strong enough to replace the simultaneously occurring water loss; and secondly, the time needed to replace a large water deficit is very long, more than 12 hours. Lastly, there is a limit to the rate at which water can pass from the stomach (where it is stored) to the intestine (gut), where the absorption takes place. This rate is lower than observed sweat rates during exercise in hot conditions.
There have been a large number of studies on various beverages to restore body water, electrolytes and carbohydrate stores of athletes during prolonged exercise. The main findings are as follows:
- The amount of the fluid which can be utilized—that is, transported through the stomach to the intestine—is limited by the “gastric emptying rate”, which has a maximum of about 1,000 ml/h.
- If the fluid is “hyperosmotic” (contains ions/molecules in higher concentrations than the blood) the rate is slowed down. On the other hand “iso-osmotic fluids” (containing water and ions/molecules to the same concentration, osmolality, as blood) are passed at the same rate as pure water.
- Addition of small amounts of salt and sugar increases the rate of uptake of water from the gut (Maughan 1991).
With this in mind you can make your own “rehydration fluid” or choose from a large number of commercial products. Normally water and electrolyte balance is regained by drinking in connection with meals. Workers or athletes with large sweat losses should be encouraged to drink more than their urge. Sweat contains about 1 to 3 g of NaCl per litre. This means that sweat losses of above 5 l per day may cause a deficiency in sodium chloride, unless the diet is supplemented.
Workers and athletes are also counselled to control their water balance by weighing themselves regularly—for example, in the morning (at same time and condition)—and try to maintain a constant weight. However, a change in body weight does not necessarily reflect the degree of hypohydration. Water is chemically bound to glycogen, the carbohydrate store in the muscles, and liberated when glycogen is used during exercise. Weight changes of up to about 1 kg may occur, depending on the glycogen content of the body. The body weight “morning to morning” also shows changes due to “biological variations” in water contents—for example, in women in relation to the menstrual cycle up to 1 to 2 kg of water can be retained during the premenstrual phase (“premenstrual tension”).
The control of water and electrolytes
The volume of the body water compartments—that is, the extracellular and intracellular fluid volumes—and their concentrations of electrolytes is held very constant through a regulated balance between intake and loss of fluid and substances.
Water is gained from the intake of food and fluid, and some is liberated by metabolic processes, including combustion of fat and carbohydrates from food. The loss of water takes place from the lungs during breathing, where the inspired air takes up water in the lungs from moist surfaces in the airways before it is exhaled. Water also diffuses through the skin in small amount in comfortable conditions during rest. However, during sweating water can be lost at rates of more than 1 to 2 l/h for several hours. The body water content is controlled. Increased water loss by sweating is compensated for by drinking and by a reduction in urine formation, while excess water is excreted by increased urine production.
This control both of intake and output of water is exerted through the autonomic nervous system, and by hormones. Thirst will increase the water intake, and the water loss by the kidneys is regulated; both the volume and electrolyte composition of urine are under control. The sensors in the control mechanism are in the heart, responding to the “fullness” of the vascular system. If the filling of the heart is reduced—for example, after a sweat loss—the receptors will signal this message to the brain centres responsible for the sensation of thirst, and to areas which induce a liberation of anti-diuretic hormone (ADH) from the posterior pituitary. This hormone acts to reduce the urine volume.
Similarly, physiological mechanisms control the electrolyte composition of the body fluids via processes in the kidneys. The food contains nutrients, minerals, vitamins and electrolytes. In the present context, the intake of sodium chloride is the important issue. The dietary sodium intake varies with eating habits, between 10 and 20 to 30 g per day. This is normally much more than is needed, so the excess is excreted by the kidneys, controlled by the action of multiple hormonal mechanisms (angiotensin, aldosterone, ANF, etc.) which are controlled by stimuli from osmoreceptors in the brain and in the kidneys, responding to the osmolality of primarily Na+ and Cl– in the blood and in the fluid in the kidneys, respectively.
Interindividual and Ethnic Differences
Differences between male and female as well as younger and older persons in reaction to heat might be expected. They differ in certain characteristics which might influence heat transfer, such as surface area, height/weight ratio, thickness of insulating skin fat layers, and in physical ability to produce work and heat (aerobic capacity » maximal oxygen consumption rate). Available data suggest that heat tolerance is reduced in older persons. They start to sweat later than do young individuals, and older people react with a higher blood flow in their skin during heat exposure.
Comparing the sexes it has been observed that women tolerate humid heat better than men do. In this environment the evaporation of sweat is reduced, so the slightly greater surface/mass area in women could be to their advantage. However, aerobic capacity is an important factor to be considered when comparing individuals exposed to heat. In laboratory conditions the physiological responses to heat are similar, if groups of subjects with the same physical work capacity (“maximal oxygen uptake”—VO2 max) are tested—for instance, younger and older males, or males versus females (Pandolf et al. 1988). In this case a certain work task (exercise on a bicycle ergometer) will result in the same load on the circulatory system—that is, the same heart rate and the same rise in core temperature—independent of age and sex.
The same considerations are valid for comparison between ethnic groups. When differences in size and aerobic capacity are taken into account, no significant differences due to race can be pointed out. But in daily life in general, older persons do have, on average, a lower VO2 max than younger persons, and females a lower VO2 max than males in the same age group.
Therefore, when performing a specific task which consists of a certain absolute work rate (measured, e.g., in Watts), the person with a lower aerobic capacity will have a higher heart rate and body temperature and be less able to cope with the extra strain of external heat, than one with a higher VO2 max.
For occupational health and safety purposes a number of heat stress indices have been developed. In these the large interindividual variation in response to heat and work are taken into account, as well as the specific hot environments for which the index is constructed. These are treated elsewhere in this chapter.
Persons exposed repeatedly to heat will tolerate the heat better after even a few days. They become acclimatized. Sweating rate is increased and the resulting increased cooling of the skin leads to a lower core temperature and heart rate during work under the same conditions.
Therefore, artificial acclimation of personnel who are expected to be exposed to extreme heat (firefighters, rescue personnel, military personnel) will probably be of benefit to reduce the strain.
Summing up, the more heat a person produces, the more must be dissipated. In a hot environment the evaporation of sweat is the limiting factor for heat loss. Interindividual differences in the capacity for sweating are considerable. While some persons have no sweat glands at all, in most cases, with physical training and repeated exposure to heat, the amount of sweat produced in a standard heat stress test is increased. Heat stress results in an increase in heart rate and core temperature. Maximal heart rate and/or a core temperature of about 40ºC sets the absolute physiological limit for work performance in a hot environment (Nielsen 1994).