Children categories

53. Environmental Health Hazards (11)

53. Environmental Health Hazards
Chapter Editors: Annalee Yassi and Tord Kjellström
Table of Contents
Tables and Figures
Linkages between Environmental and Occupational Health
Annalee Yassi and Tord Kjellström
Food and Agriculture
Friedrich K. Käferstein
Industrial Pollution in Developing Countries
Niu Shiru
Developing Countries and Pollution
Tee L. Guidotti
Air Pollution
Isabelle Romieu
Land Pollution
Tee L. Guidotti and Chen Weiping
Water Pollution
Ivanildo Hespanhol and Richard Helmer
Energy and Health
L.D. Hamilton
Urbanization
Edmundo Werna
Global Climate Change and Ozone Depletion
Jonathan A. Patz
Species Extinction, Biodiversity Loss and Human Health
Eric Chivian
Tables
Click a link below to view table in article context.
1. Selected major "environmental disease" outbreaks
2. Foodborne-disease agents: epidemiology features
3. Major sources of outdoor air pollutants
4. Exposure-response relationship of PM10
5. Changes in ozone concentration: health outcomes
6. Morbidity & mortality: water-related diseases
7. Generating fuel electricity: health effects
8. Generating renewable electricity: health effects
9. Generating nuclear electricity: health effects
10. Housing & health
11. Urban infrastructure & health
12. Global status of major vector-borne diseases
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

54. Environmental Policy (10)

54. Environmental Policy
Chapter Editor: Larry R. Kohler
Table of Contents
Tables and Figures
Overview Occupational Safety and Health and the Environment - Two Sides of the Same Coin
Larry R. Kohler
Environment and the World of Work: An Integrated Approach to Sustainable Development, Environment and the Working Environment
Larry R. Kohler
Law and Regulations
Françoise Burhenne-Guilmin
International Environmental Conventions
David Freestone
Environmental Impact Assessments
Ron Bisset
Life-Cycle Assessment (Cradle-To-Grave)
Sven-Olof Ryding
Risk Assessment and Communication
Adrian V. Gheorghe and Hansjörg Seiler
Environmental Auditing - Definition and Methodology
Robert Coyle
Environmental Management Strategies and Workers’ Protection
Cecilia Brighi
Environmental Pollution Control: Making Pollution Prevention a Corporate Priority
Robert P. Bringer and Tom Zosel
Tables
Click a link below to view table in article context.
1. Scope of an environmental audit
2. Basic steps in environmental auditing
3. Voluntary agreements relevant to the environment
4. Environment-protection measures & collective agreements
5. Collective agreements on environment-protection
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

55. Environmental Pollution Control (11)

55. Environmental Pollution Control
Chapter Editors: Jerry Spiegel and Lucien Y. Maystre
Table of Contents
Tables and Figures
Environmental Pollution Control and Prevention
Jerry Spiegel and Lucien Y. Maystre
Air Pollution Management
Dietrich Schwela and Berenice Goelzer
Air Pollution: Modelling of Air Pollutant Dispersion
Marion Wichmann-Fiebig
Air Quality Monitoring
Hans-Ulrich Pfeffer and Peter Bruckmann
Air Pollution Control
John Elias
Water Pollution Control
Herbert C. Preul
Dan Region Sewage Reclamation Project: A Case Study
Alexander Donagi
Principles of Waste Management
Lucien Y. Maystre
Solid Waste Management and Recycling
Niels Jorn Hahn and Poul S. Lauridsen
Case Study: Canadian Multimedia Pollution Control and Prevention on the Great Lakes
Thomas Tseng, Victor Shantora and Ian R. Smith
Cleaner Production Technologies
David Bennett
Tables
Click a link below to view table in article context.
1. Common atmospheric pollutants & their sources
2. Measurement planning parameters
3. Manual measurement procedures for inorganic gases
4. Automated measurement procedures for inorganic gases
5. Measurement procedures for suspended particulate
6. Long-distance measurement procedures
7. Chromatographic air quality measurement procedures
8. Systematic air quality monitoring in Germany
9. Steps in selecting pollution controls
10. Air quality standards for sulphur dioxide
11. Air quality standards for benzene
12. Examples of best available control technology
13. Industrial gas: cleaning methods
14. Sample emission rates for industrial processes
15. Wastewater treatment operations & processes
16. List of investigated parameters
17. Parameters investigated at the recovery wells
18. Sources of waste
19. Criteria for selection of substances
20. Reductions in releases of dioxin & furan in Canada
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Principles of Waste Management
Environmental awareness is leading to a rapid transformation of waste management practices. Interpretation of this change is necessary before examining in more detail the methods that are applied to waste management and to the handling of residues.
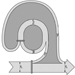
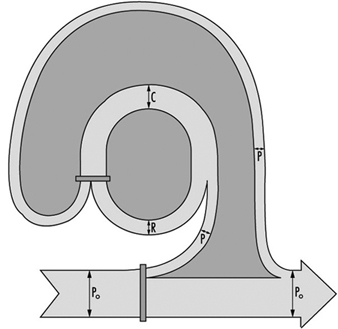
Modern principles of waste management are based on the paradigm of a geared connection between the biosphere and the anthroposphere. A global model (figure 1) relating these two spheres is based on the assumption that all materials drawn out of the environment end up as waste either directly (from the production sector) or indirectly (from the recycling sector), bearing in mind that all consumption waste flows back to this recycling sector either for recycling and/or for disposal.
Figure 1. A global model of the principles of waste management
From this perspective, recycling must be defined broadly: from the recycling of whole objects (returnables), to the recycling of objects for some of their spare parts (e.g., cars, computers), to the production of new materials (e.g., paper and cardboard, tin cans) or the production of similar objects (recycling, downcycling and so on). Over the long term, this model can be visualized as a steady-state system wherein goods end up as waste after a few days or often a few years.
Deductions from the Model
Some major deductions can be made from this model, provided the various flows are clearly defined. For purposes of this model:
- Po=the annual input of materials drawn from the environment (bio-, hydro- or lithospheres). In a steady state, this input is equal to the annual final disposal of waste.
- P=the annual production of goods from Po.
- C=the annual flow of goods in the anthroposphere.
- R=the annual flow of waste converted to goods through recycling. (In a steady state: C=R+ P)
- p=the effectiveness of production, measured as the ratio of P/Po.
- If r=the effectiveness of recycling, measured as the ratio of R/C, then the relationship is: C/Po=p(1-r).
- If C/Po=C*; then C* is the ratio of goods to the materials drawn out of nature.
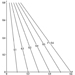
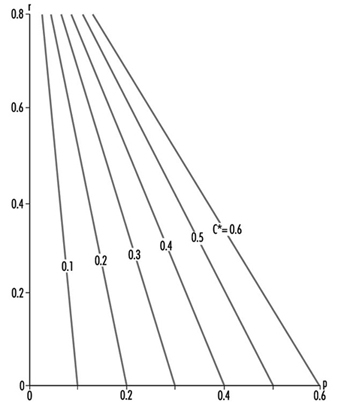
In other words, C* is a measure of the meshing of the connection between environment and anthroposphere. It is related to the efficiency of the production and of the recycling sectors. The relationship between C*, p and r, which is a utility function, can be charted as in figure 2, which shows the explicit trade-off between p and r, for a selected value of C*.
Figure 2. A utility function illustrating production recycling trade-offs
In the past, industry has developed along the line of an increase of the efficiency of production, p. Currently, in the late 1990s, the price of waste disposal through dispersion into the atmosphere, into bodies of water or into soils (uncontrolled tipping), or the burial of waste in confined deposit sites has increased very rapidly, as a result of increasingly stringent environmental protection standards. Under these conditions, it has become economically attractive to increase the effectiveness of recycling (in other words, to increase r). This trend will persist through the coming decades.
One important condition has to be met in order to improve the effectiveness of recycling: the waste to be recycled (in other words the raw materials of the second generation) must be as “pure” as possible (i.e., free of unwanted elements which would preclude the recycling). This will be achieved only through the implementation of a generalized policy of “non-mixing” of domestic, commercial and industrial waste at the source. This is often incorrectly termed sorting at the source. To sort is to separate; but the idea is precisely not to have to separate by storing the various categories of waste in separate containers or places until they are collected. The paradigm of modern waste management is non-mixing of waste at the source so as to enable an increase in the efficiency of recycling and thus to achieve a better ratio of goods per material drawn out of the environment.
Waste Management Practices
Waste may be grouped into three major categories, depending on its production:
- from the primary sector of production (mining, forestry, agriculture, animal breeding, fishery)
- from the production and transformation industry (foods, equipment, products of all types)
- from the consumption sector (households, enterprises, transportation, trade, construction, services, etc.).
Waste can be also classified by legislative decree:
- municipal waste and mixed waste from enterprises which may be aggregated as municipal waste, since both consist of the same categories of waste and are of small size (vegetables, paper, metals, glass, plastics and so on), although in differing proportions.
- bulky urban waste (furniture, equipment, vehicles, construction and demolition waste other than inert material)
- waste subject to special legislation (e.g., hazardous, infectious, radioactive).
Management of municipal and ordinary commercial waste:
Collected by trucks, these wastes can be transported (directly or by road-to-road, road-to-rail or road-to-waterway transfer stations and long-distance transportation means) to a landfill, or to a treatment plant for material recovery (mechanical sorting, composting, biomethanization), or for energy recovery (grid or kiln incinerator, pyrolysis).
Treatment plants produce proportionally small quantities of residues which may be more hazardous for the environment than the original waste. For example, incinerators produce fly ashes with very high heavy metal and complex chemical content. These residues are often classified by legislation as hazardous waste and require appropriate management. Treatment plants differ from landfills because they are “open systems” with inputs and outputs, whereas landfills are essentially “sinks” (if one neglects the small quantity of leachate which deserves further treatment and the production of biogas, which may be an exploited source of energy on very large landfills).
Industrial and domestic equipment:
The present trend, which also has commercial contributions, is for the producers of the waste sectors (e.g., cars, computers, machines) to be responsible for the recycling. Residues are then either hazardous waste or are similar to ordinary waste from enterprises.
Construction and demolition waste:
The increasing prices of landfills is an incentive for a better sorting of such waste. Separation of the hazardous and burnable waste from the large quantity of inert materials allows the latter to be disposed of at a far lower rate than mixed waste.
Special waste:
Chemically hazardous waste must be treated through neutralization, mineralization, insolubilization or be made inert before it can be deposited in special landfills. Infectious waste is best burnt in special incinerators. Radioactive waste is subject to very strict legislation.
Management of Residues
Production and consumption waste which cannot be recycled, down-cycled, reused or incinerated to produce energy must eventually be disposed of. The toxicity for the environment of these residues should be reduced according to the principle of “best available technology at an acceptable price.” After this treatment, the residues should be deposited in sites where they will not contaminate the water and the ecosystem and spread into the atmosphere, into the sea or into lakes and streams.
Deposits of waste are usually dated by the combination of multilayer isolation (using clay, geotextiles, plastic foils and so on), the diversion of all exogenous water, and waterproof cover layers. Permanent deposits need to be monitored for decades. Restrictions on land use of a deposit site must also be controlled for long periods of time. Controlled drainage systems for leachates or gases are necessary in most cases.
More biochemically stable and chemically inert residues from waste treatment require less stringent conditions for their final disposal, making it less difficult to find a deposit site for them within the region of production of the waste. Export of wastes or their residues, which always awakens NIMBY (Not In My Back Yard) reactions, might thus be avoided.
Solid Waste Management and Recycling
Solid wastes are traditionally described as residual products, which represent a cost when one has to resort to disposal.
Management of waste encompasses a complex set of potential impacts on human health and safety, and the environment. The impacts, although the type of hazards may be similar, should be distinguished for three distinct types of operation:
- handling and storage at the waste producer
- collection and transportation
- sorting, processing and disposal.
One should bear in mind that health and safety hazards will arise where the waste is produced in the first place - in the factory or with the consumer. Hence, waste storage at the waste generator - and especially when waste is separated at source - may cause harmful impact on the nearby surroundings. This article will focus on a framework for understanding solid waste management practices and situating the occupational health and safety risks associated with the waste collection, transportation, processing and disposal industries.
Why Solid Waste Management?
Solid waste management becomes necessary and relevant when the structure of the society changes from agricultural with low-density and widespread population to urban, high-density population. Furthermore, industrialization has introduced a large number of products which nature cannot, or can only very slowly, decompose or digest. Hence, certain industrial products contain substances which, due to low degradability or even toxic characteristics, may build up in nature to levels representing a threat to humanity’s future use of the natural resources - that is, drinking water, agricultural soil, air and so on.
The objective of solid waste management is to prevent pollution of the natural environment.
A solid waste management system should be based on technical studies and overall planning procedures including:
- studies and estimates on waste composition and amounts
- studies on collection techniques
- studies on processing and disposal facilities
- studies on prevention of pollution of the natural environment
- studies on occupational health and safety standards
- feasibility studies.
The studies must include protection of the natural environment and occupational health and safety aspects, taking the possibilities of sustainable development into consideration. As it seldom is possible to solve all problems at one time, it is important at the planning stage to note that it is helpful to set up a list of priorities. The first step in solving environmental and occupational hazards is to recognize the existence of the hazards.
Principles of Waste Management
Waste management involves a complex and wide range of occupational health and safety relations. Waste management represents a “reverse” production process; the “product” is removal of surplus materials. The original aim was simply to collect the materials, reuse the valuable part of the materials and dispose of what remained at the nearest sites not used for agriculture purposes, buildings and so on. This is still the case in many countries.
Sources of waste can be described by the different functions in a modern society (see table 1).
Table 1. Sources of waste
|
Activity |
Waste description |
|
Industry |
Product residues |
|
Wholesale |
Default products |
|
Retail |
Transport packaging |
|
Consumer |
Transport packaging |
|
Construction and demolition |
Concrete, bricks, iron, soil, etc. |
|
Infrastructure activities |
Park waste |
|
Waste processing |
Rejects from sorting facilities |
Each type of waste is characterized by its origin or what type of product it was before it became waste. Hence, basically its health and safety hazards should be laid down upon the restriction of handling the product by the waste producer. In any case, storage of the waste may create new and stronger elements of hazards (chemical and/or biological activity in the storage period).
Solid waste management can be distinguished by the following stages:
- separation at source into specific waste fraction depending on material characteristics
- temporary storage at the waste producer in bins, sacks, containers or in bulk
- collection and transportation by vehicle:
- manual, horse team, motorized and so on
- open platform, closed truck body, compacting unit and so on
- transfer station: compaction and reloading to larger transport units
- recycling and/or waste processing facilities
- waste processing:
- manual or mechanical sorting out into different material fractions for recycling
- processing of presorted waste fractions to secondary raw materials
- processing for new (raw) materials
- incineration for volume reduction and/or energy recovery
- anaerobic digestion of organics for production of soil conditioner, fertilizer and energy (biogas)
- composting of organics for production of soil conditioner and fertilizer
- waste disposal:
- landfill, which should be designed and located to prevent migration of polluted water (landfill leachate), especially into drinking water resources (groundwater resources, wells and rivers).
Recycling of waste can take place at any stage of the waste system, and at each stage of the waste system, special occupational health and safety hazards may arise.
In low-income societies and non-industrial countries, recycling of solid waste is a basic income for the waste collectors. Typically, no questions are put on the health and safety hazards in these areas.
In the intensely industrialized countries, there is a clear trend for putting increased focus on recycling of the huge amounts of waste produced. Important reasons go beyond the direct market value of the waste, and include the lack of proper disposal facilities and the growing public awareness of the imbalance between consumption and protection of the natural environment. Thus, waste collection and scavenging have been renamed recycling to upgrade the activity in the mind of the public, resulting in a steeply growing awareness of the working conditions in the waste business.
Today, the occupational health and safety authorities in the industrialized countries are focusing on working conditions which, a few years ago, passed off unnoticed with unspoken acceptance, such as:
- improper heavy lifting and excessive amount of materials handled per working day
- inappropriate exposure to dust of unknown composition
- unnoticed impact by micro-organisms (bacteria, fungi) and endotoxins
- unnoticed exposure to toxic chemicals.
Recycling
Recycling or salvaging is the word covering both reuse (use for the same purpose) and reclamation/recovery of materials or energy.
The reasons for implementing recycling may change depending on national and local conditions, and the key ideas in the arguments for recycling may be:
- detoxification of hazardous waste when high environmental standards are set by the authorities
- resource recovery in low income areas
- reduction of volume in areas where landfilling is predominant
- energy recovery in areas where conversion of waste to energy can replace fossil fuel (coal, natural gas, crude oil and so on) for energy production.
As previously mentioned, recycling can occur at any stage in the waste system, but recycling can be designed to prevent waste from being “born”. That is the case when products are designed for recycling and a system for repurchasing after end-use, for instance by putting a deposit on beverage containers (glass bottles and so on).
Hence, recycling may go further than mere implementation of reclamation or recovery of materials from the waste stream.
Recycling of materials implies, in most situations, separation or sorting of the waste materials into fractions with a minimum degree of fineness as a prerequisite to the use of the waste as a substitute for virgin or primary raw materials.
The sorting may be performed by waste producers (source separation), or after collection, meaning separation at a central sorting plant.
Source Separation
Source separation will, by today’s technology, result in fractions of waste which are “designed” for processing. A certain degree of source separation is inevitable, as some mixtures of waste fractions can be separated into usable material fractions again only by great (economic) effort. The design of source separation must always take the final type of recycling into consideration.
The goal of the source sorting system should be to avoid a mixing or pollution of the different waste fractions, which could be an obstacle to easy recycling.
The collection of source-sorted waste fractions will often result in more distinct occupational health and safety hazards than does collection in bulk. This is due to concentration of specific waste fractions - for instance, toxic substances. Sorting out of easily degradable organics may result in producing high levels of exposure to hazardous fungi, bacteria, endotoxins and so on, when the materials are handled or reloaded.
Central Sorting
Central sorting may be done by mechanical or manual methods.
It is the general opinion that mechanical sorting without prior source separation by today’s known technology should be used only for production of refuse derived fuel (RDF). Prerequisites for acceptable working conditions are total casing of the mechanical equipment and use of personal “space suits” when service and maintenance have to be carried out.
Mechanical central sorting with prior source separation has, with today’s technology, not been successful due to difficulties in reaching proper sorting efficiency. When the characteristics of the sorted out waste fractions become more clearly defined, and when these characteristics become valid on a national or international basis, then it can be expected that new proper and efficient techniques will be developed. The success of these new techniques will be closely linked to prudent consideration to obtaining acceptable working conditions.
Manual central sorting should imply prior source separation to avoid occupational health and safety hazards (dust, bacteria, toxic substances and so on). The manual sorting should be limited to only a limited number of waste fraction “qualities” to avoid foreseeable sorting mistakes at the source, and to facilitate easy control facilities at the plant’s reception area. As the waste fractions become more clearly defined, it will be possible to develop more and more devices for automatic sorting procedures to minimize direct human exposure to noxious substances.
Why Recycling?
It is important to note that recycling is not a waste processing method that should be seen independently of other waste management practices. In order to supplement recycling, it is necessary to have access to a properly managed landfill and perhaps to more traditional waste processing facilities such as incineration plants and composting facilities.
Recycling should be evaluated in connection with
- local supply of raw materials and energy
- what is substituted - renewable (i.e., paper/tree) resources or non-renewable (i.e., oil) resources.
As long as oil and coal are used as energy resources, for example, incineration of waste and refuse-derived fuel with energy recovery will constitute a viable waste management option based on energy recovery. Minimization of waste quantities by this method, however, must end in final deposits subject to extremely strict environmental standards, which may be very expensive.
Case Study: Canadian Multimedia Pollution Control and Prevention on the Great Lakes
The Challenge
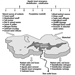
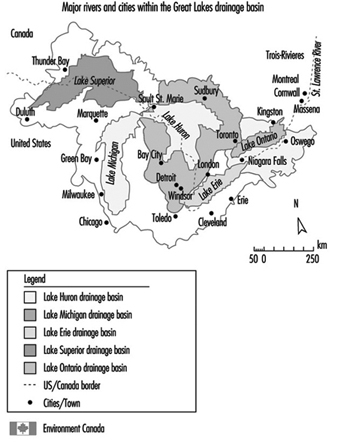
The Great Lakes are a shared resource between Canada and the United States (see figure 1). The five large lakes contain over 18% of the world’s surface water. The basin is home to one in every three Canadians (approximately 8.5 million ) and one in every nine Americans (27.5 million). The basin is the industrial heartland of both countries - one-fifth of the US industrial base and one-half of Canada’s. Economic activities around the Great Lakes basin generate an estimated 1 trillion dollars of wealth each year. Over time, increasing population and industrial activities created a variety of stresses on the lakes until the need for concerted action to protect the Great Lakes by the two countries was recognized in mid-century.
Figure 1. Great Lakes drainage basin: St. Lawrence River
The Response
Since the 1950s, both countries have put in place domestic and bilateral programmes to address gross pollution problems and also to respond to more subtle water quality concerns. As a result of these actions, Great Lakes waters are visibly cleaner than they were at mid-century, loadings of heavy metals and organic chemicals have decreased and contaminant levels in fish and aquatic birds have gone down significantly. The successes of Canada–United States actions to restore and protect the Great Lakes provide a model for bilateral cooperation on resource management, but challenges remain.
The Case Study in Perspective
The threats posed by persistent toxic substances, however, are long term in nature and their management requires a multimedia, comprehensive at-source approach. To achieve a long-term goal of virtual elimination of persistent toxic substances from the Great Lakes, environmental authorities, industries and other stakeholders in the basin were challenged to develop new approaches and programmes. The purpose of this case study report is to provide a brief summary of Canadian pollution control programmes and the progress achieved by 1995, and to outline initiatives for managing persistent toxics in the Great Lakes. Similar US initiatives and programmes are not discussed herein. Interested readers should contact the Great Lakes National Program Office of the US Environmental Protection Agency in Chicago for information on federal and state programmes for protecting the Great Lakes.
1970s–1980s
A significant problem acknowledged to be affecting Lake Erie in the 1960s was nutrient enrichment or eutrophication. The identified need for bilateral actions prompted Canada and the United States to sign the first Great Lakes Water Quality Agreement (GLWQA) in 1972. The Agreement outlined abatement goals for reducing phosphorus loadings primarily from laundry detergents and municipal sewage effluent. In response to this commitment Canada and Ontario enacted legislation and programmes for controlling point sources. Between 1972 and 1987, Canada and Ontario invested more than 2 billion dollars in sewage treatment plant construction and upgrading in the Great Lakes basin.
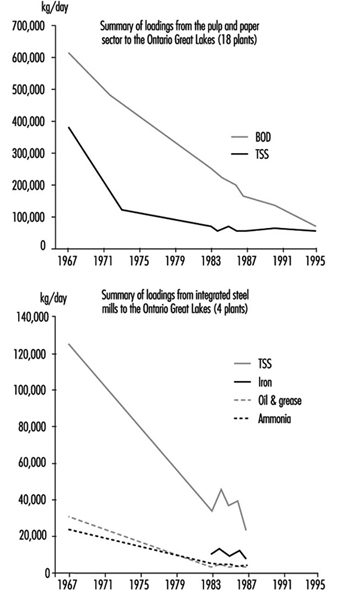
Figure 2. Progress on industrial abatement
The 1972 GLWQA also identified the need to reduce releases of toxic chemicals into the lakes from industries and other sources such as spills. In Canada, the promulgation of federal effluent (end of pipe) regulations in the 1970s for conventional pollutants from major industrial sectors (pulp and paper, metal mining, petroleum refining and so on) provided a national baseline standard, while Ontario established similar effluent guidelines tailored for local needs including the Great Lakes. Actions by industries and municipalities to meet these federal and Ontario effluent requirements produced impressive results; for example, phosphorus loadings from point sources to Lake Erie were reduced by 70% between 1975 and 1989, and discharges of conventional pollutants from the seven Ontario petroleum refineries were cut by 90% since the early 1970s. Figure 2 shows similar loading reduction trends for the pulp and paper and the iron and steel sectors.
By the mid-1970s evidence of elevated concentrations of toxic chemicals in Great Lakes fish and wildlife, reproductive abnormalities in some fish-eating birds and population decline in a number of species implicated persistent bioaccumulative toxic substances, which became the new focus for the binational protection effort. Canada and the United States signed a second Great Lakes Water Quality Agreement in 1978, in which the two countries pledged to “restore and maintain the chemical, physical and biological integrity of the waters of the Great Lakes Ecosystem”. A key challenge was the policy “that the discharge of toxic substances in toxic amounts be prohibited and the discharge of any or all persistent toxic substances be virtually eliminated”. The call for virtual elimination was necessary, as persistent toxic chemicals may concentrate and accumulate in the food chain, causing severe and irreversible damages to the ecosystem, whereas chemicals which are not persistent needed to be kept below levels which cause immediate harm.
In addition to tighter controls on point sources, Canada and Ontario developed and/or strengthened controls on pesticides, commercial chemicals, hazardous wastes and non-point sources of pollution such as dump sites and incinerators. Government initiatives became more multimedia oriented, and the concept of “cradle to grave” or “responsible care” for chemicals became the new environmental management philosophy for government and industries alike. A number of persistent toxic pesticides were banned under the federal Pest Control Products Act (DDT, Aldrin, Mirex, Toxaphene, Chlordane) and the Environmental Contaminants Act was used to (1) prohibit commercial, manufacturing and processing uses of persistent toxics (CFC, PPB, PCB, PPT, Mirex, lead) and (2) to limit chemical releases from specific industrial operations (mercury, vinyl chloride, asbestos).
By the early 1980s, results from these programmes and measures and similar American efforts started producing evidence of a rebound. Contaminant levels in Great Lakes sediments, fish and wildlife were on the decline, and noted environmental improvements included the return of bald eagles to the Canadian shore of Lake Erie, a 200-fold increase in cormorant population, a resurgence in osprey on Georgian Bay and the re-establishment in the Toronto Harbour area of common terns - all have been affected by levels of persistent toxic substances in the past, and their recovery illustrates the success of this approach to date.
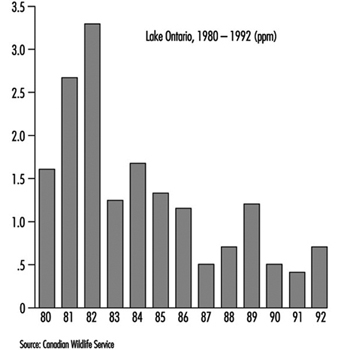
Figure 3. Mirex in herring gull eggs
The trend toward reduced concentrations for some of the persistent toxic substances in fish, wildlife and sediments levelled off by the mid-1980s (see Mirex in herring gull eggs in figure 3). It was concluded by scientists that:
- While the water pollution and contaminants control programmes in place were helpful, they were not enough to bring about further reductions in contaminant concentrations.
- Additional measures were required for non-point sources of persistent toxics including contaminated sediments, long range atmospheric input of pollutants, abandoned dump sites and so on.
- Some pollutants can persist in the ecosystem at minute concentrations and can bioaccumulate in the food chain for a long time.
- The most efficient and effective approach for dealing with persistent toxics is to prevent or eliminate their generation at source rather than virtually eliminate their release.
It was generally agreed that achieving virtual elimination in the environment through the application of zero-discharge philosophy to sources and the ecosystem approach to Great Lakes water quality management needed to be further strengthened and promoted.
To reaffirm their commitment to the virtual elimination goal for persistent toxic substances, Canada and the United States amended the 1978 Agreement through a protocol in November 1987 (United States and Canada 1987). The protocol designated areas of concern where beneficial uses have been impaired around the Great Lakes, and required the development and implementation of remedial action plans (RAPs) for both point and non-point sources in the designated areas. The protocol also stipulated lakewide management plans (LAMPs) to be used as the main framework for resolving whole-lake impairment of beneficial uses and for coordinating control of persistent toxic substances impacting each of the Great Lakes. Furthermore, the protocol included new annexes for establishing programmes and measures for airborne sources, contaminated sediments and dump sites, spills and control of exotic species.
1990s
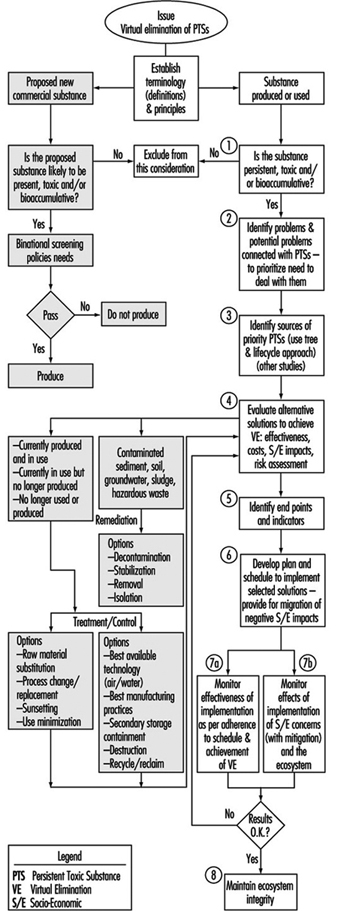
Following the signing of the 1987 protocol, the goal of virtual elimination was strongly promoted by environmental interest groups on both sides of the Great Lakes as concerns about the threat of persistent toxics increased. The International Joint Commission (IJC), the binational advisory body created under the 1909 Boundary Waters Treaty, also strongly advocated the virtual elimination approach. An IJC binational task force recommended a strategy for Virtual Elimination in 1993 (see figure 4). By the mid-1990s, the IJC and the parties are attempting to define a process for implementing this strategy, including considerations for socioeconomic impacts.
Figure 4. Decision-making process for virtual elimination of persistent toxic substances from the Great Lakes
The governments of Canada and Ontario responded in a number of ways to control or reduce the release of persistent toxics. The important programmes and initiatives are briefly summarized below.
Canadian Environmental Protection Act (CEPA)
In 1989, Environment Canada consolidated and streamlined its legal mandates into a single statute. CEPA provides the federal government with comprehensive powers (e.g., information gathering, regulations making, enforcement) over the entire life cycle of chemicals. Under CEPA, the New Substances Notification Regulations establish screening procedures for new chemicals so that persistent toxics that cannot be adequately controlled will be prohibited from being imported, manufactured or used in Canada. The first phase of the Priority Substances List (PSL I) assessment programme was completed in 1994; 25 of the 44 substances assessed were found to be toxic under the definition of CEPA, and the development of management strategies for these toxic chemicals was initiated under a Strategic Options Process (SOP); an additional 56 priority substances will be nominated and assessed in phase II of the PSL programme by the year 2000. The National Pollutant Release Inventory (NPRI) was implemented in 1994 to mandate industrial and other facilities that meet the reporting criteria to annually report their releases to air, water and land, and their transfers in waste, of 178 specified substances. The inventory, modelled on the Toxic Release Inventory (TRI) in the United States, provides an important database for prioritizing pollution prevention and abatement programmes.
Canada-Ontario Agreement (COA)
In 1994, Canada and Ontario set out a strategic framework for coordinated action to restore, protect and conserve the Great Lakes ecosystem with a key focus on reducing the use, generation or release of 13 Tier I persistent toxic substances by the year 2000 (Canada and Ontario 1994). COA also targets an additional list of 26 priority toxics (Tier II) for significant reductions. Specifically for Tier I substances, COA will: (1) confirm zero discharge of five banned pesticides (Aldrin, DDT, Chlordane, Mirex, Toxaphene); (2) seek to decommission 90% of high-level PCBs, destroy 50% now in storage and accelerate destruction of low-level PCBs in storage; and (3) seek 90% reduction in the release of the remaining seven Tier I substances (benzo(a)pyrene, hexachlorobenzene, alkyl-lead, octachlorostyrene, PCDD (dioxins) PCDF (furans) and mercury).
The COA approach is to seek quantitative reductions wherever feasible, and sources are challenged to apply pollution prevention and other means to meet the COA targets. Fourteen projects have already been launched by federal Ontario staff to achieve reduction/elimination of Tiers I and II substances.
Toxic Substances Management Policy
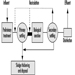
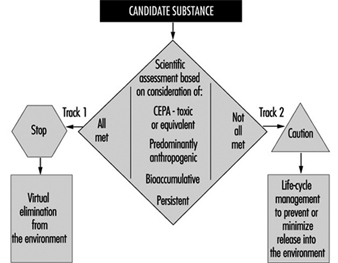
In recognition of the need for a preventive and precautionary approach, Environment Canada announced in June 1995 a national Toxic Substances Management Policy as the framework for efficient management of toxic substances in Canada (Environment Canada 1995a). The policy adopts a two-track approach (see figure 5) that recognizes management actions must be tailored to the characteristics of chemicals; that is:
- to virtually eliminate from the environment substances that are predominantly anthropogenic, persistent, bioaccumulative and toxic (Track I)
- to implement full life cycle (cradle-to-grave) management of all other substances of concern (Track II).
Figure 5. Selection of management objectives under the Toxic Substances Management Policy
A set of scientifically based criteria (Environment Canada 1995b) (see table 1) will be used to categorize substances of concern into the two tracks. If a substance identified for either track is not adequately controlled under existing programmes, additional measures will be identified under the multi-stakeholder Strategic Options Process. The policy is consistent with the Great Lakes Water Quality Agreement and will direct and frame a number of domestic programmes by defining their ultimate environmental objective, but the means and pace of achieving the ultimate objective will vary by chemical and source. Further, Canada’s position on persistent toxics will also be framed by this policy in international discussions.
Table 1. Criteria for the selection of substances for Track 1 toxic substances management policy
|
Persistence |
Bioaccumulation |
Toxicity |
Predominantly Anthropogenic |
|
|
Medium |
Half-life |
|||
|
Air |
≥2 days |
BAF≥5,000 |
CEPA-toxic |
Concentration |
Chlorine Action Plan
A comprehensive approach to managing chlorinated substances within the context of the Toxic Substances Management Policy was announced in October 1994 by Environment Canada (Environment Canada 1994). The approach will be to prune the chlorine-use tree with a five-part action plan that will (1) target action on critical uses and products, (2) improve scientific understanding of chlorine and its impact on health and the environment, (3) detail socioeconomic implications, (4) improve public access to information and (5) promote international actions on chlorinated substances. Chlorine use has already decreased in Canada in recent years, for example by 45% in the pulp and paper sector since 1988. Implementation of the Chlorine Action Plan will accelerate this reduction trend.
Great Lakes Pollution Prevention Initiative
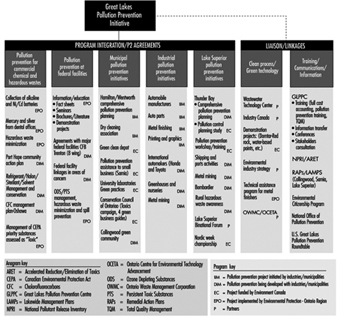
A strong pollution prevention programme has been put in place for the Great Lakes basin. Since March 1991, Environment Canada and the Ontario Ministry of the Environment and Energy have been working together with industries and other stakeholders to develop and implement pollution prevention projects, in contrast to waste treatment or reducing pollution after its generation. In 1995/96, more than 50 projects will cover commercial chemicals, hazardous waste management, federal facilities, industries, municipalities and the Lake Superior basin. Figure 6 provides an overview of these projects, which fall into two main categories: programme integration or voluntary agreements. The figure also shows programme linkages with other programmes discussed earlier (NPRI, RAP, LAMP) and a number of institutions that work with Environment Canada closely on green technologies and clean processes, as well as on training, information and communications. Pollution prevention projects can produce impressive results, as evidenced by the Automotive Manufacturers, who have undertaken 15 pilot projects recently, thereby reducing or eliminating 2.24 million kilograms of targeted substances from the manufacture of automobiles at the Ontario facilities of Chrysler, Ford and General Motors.
Figure 6. Great Lakes pollution prevention
Accelerated Reduction/Elimination of Toxics (ARET)
ARET is a cooperative multi-stakeholder initiative launched in 1994 that seeks the eventual elimination of 14 priority toxics with an interim target (by the year 2000) of a 90% reduction/elimination and reduced emission (50%) of 87 less harmful toxic substances (ARET Secretariat 1995). As of 1995, more than 200 companies and government agencies are participating in this voluntary initiative. Together, they reduced emissions by 10,300 tonnes in comparison with the 1988 base year and are committed to an additional 8,500 tonnes reduction by the year 2000.
Binational and international strategies
In addition to the above domestic initiatives, Canada and the United States are currently developing a binational strategy to coordinate agency action and to establish shared goals for persistent toxics in the Great Lakes basin. Goals and objectives similar to the Canada-Ontario Agreement for the Tiers I and II substances and a similar US list will be adopted. Joint projects will be developed and implemented to facilitate information exchange and agency action on priority chemicals such as PCBs and mercury. By taking an aggressive approach to virtual elimination as outlined above, Canada will be able to assume a leadership role in promoting international action on persistent toxics. Canada hosted a United Nations conference in June 1995 in Vancouver to focus global dialogue on persistent organic pollutants (POP) and to explore pollution prevention approaches to reducing their emissions around the world. Canada also co-chairs the United Nations Economic Commission for Europe (UNECE) workgroup to develop a protocol for persistent organic pollutants under the Convention on Long Range Transboundary Air Pollution.
An Example—Dioxins and Furans
For more than a decade, polychlorinated dibenzo-dioxins and furans have been recognized as a group of persistent toxics of concern to the Canadian environment and the Great Lakes. Table 2 summarizes federal actions and the reductions in releases achieved to date, illustrating the mix of programmes and initiatives which has resulted in significant reductions of these toxics. In spite of these impressive results, dioxins and furans will remain priorities under the Toxic Substances Management Policy, the Chlorine Action Plan, the Canada Ontario Agreement and the binational strategy outlined above, because virtual elimination requires further reductions.
Table 2. Summary of reductions in releases of dioxin and furan in Canada
|
Sources of Emissions |
Reductions |
Reporting Period |
Canadian Government Initiatives |
|
Bleached kraft pulpmill effluents |
82% |
1989-94 |
CEPA defoamer, wood chip and |
|
2,4,5-T—pesticide |
100% |
1985 |
Banned from use under PCPA |
|
2,4-D—pesticide |
100% |
1987-90 |
Dioxin content and use heavily |
|
Pentachlorophenol |
|
|
|
|
PCBs |
23% |
1984-93 |
CCME PCB Action Plan |
|
Incineration |
|
|
|
CCME: Canadian Council of Environmental Ministers; CEPA: Canadian Environmental Protection Act; PCPA: Pest Control Products Act.
Summary
There has been a significant improvement in the water quality of the Great Lakes as a result of pollution control actions taken by governments and stakeholders in Canada and the United States since the early 1970s. This case study report provides a summary of the Canadian effort and successes in dealing with gross pollution and conventional pollutants. It also outlines the evolution of a new approach (the Toxic Substances Management Policy, the Chlorine Action Plan, pollution prevention, voluntary action, stakeholder consultations and so on) for dealing with the much more difficult problems with persistent toxic substances in the Great Lakes. Comprehensive programmes (COA, NPRI, SOP, PSL and so on) that are being put in place with the aim of achieving the virtual elimination goal are briefly described. Details of the Canadian approach are contained in the listed references.
Cleaner Production Technologies
Prevention, Control and Remediation
Conventionally, there are three ways of addressing pollution: prevention, control and remediation. These form a hierarchy, in which the first priority or option is prevention, followed by control measures, with remediation as a poor third. Pollution abatement can refer to any means that lessens pollution, or a mitigation of pollution; in practice, it usually means control. Though the hierarchy of the three ideas is in terms of preference or priority, this is not always so in practice: there may be regulatory pressures to choose one path rather than another; one strategy may be less expensive than another, or remediation may be the most urgent - for example, in the event of a major spill or the hazardous dissemination of pollutants from a contaminated site.
Pollution prevention
Pollution prevention can be defined as a strategy or strategies which avoid the creation of pollutants in the first place. In Barry Commoner’s phrase, “If it’s not there, it can’t pollute.” Thus, if a chemical whose use results in pollution is eliminated, there will be “zero discharge” (or “zero emission”) of the pollutant. Zero discharge is more convincing if the chemical is not replaced by another chemical - an alternative or substitute - which results in a different pollutant.
One central strategy of pollution prevention is the banning, elimination or the phasing out (“sunsetting”) of specified chemicals or classes of chemical. (Alternatively, use-restrictions may be specified.) Such strategies are laid down in the form of laws or regulations by national governments, less often by international instruments (conventions or treaties) or by sub-national governments.
A second strategy is pollution reduction, again in the context of prevention rather than control. If the use of a chemical which results in pollution is reduced, then the result will almost always be less pollution. Pollution reduction strategies are exemplified in North America by toxics use reduction (TUR) programmes and in Europe by “clean technology programmes”.
Unlike bans and phase-outs, which usually apply to all (relevant) workplaces within a political jurisdiction, pollution reduction programmes apply to specific workplaces or classes of workplace. These are usually industrial manufacturing (including chemical manufacturing) workplaces over a certain size, in the first instance, though the principles of pollution reduction can be applied generally - for example, to mines, power plants, construction sites, offices, agriculture (in regard to chemical fertilizers and pesticides) and municipalities. At least two US states (Michigan and Vermont) have legislated TUR programmes for individual households which are also workplaces.
Pollution reduction can result in the elimination of specific chemicals, thus achieving the same aims as bans and phase-outs. Again, this would result in zero discharge of the pollutant concerned, but requirements to eliminate specific chemicals are not part of pollution reduction programmes; what is prescribed is a general programme with a flexible range of specified methods. A requirement to eliminate a specific chemical is an example of a “specification standard”. A requirement to institute a general programme is a “performance standard” because it allows flexibility in the mode of implementation, though a specific mandatory target (outcome) for a general programme would (confusingly) count as a specification standard. When they have to choose, businesses usually prefer performance to specification standards.
Pollution control
Pollution control measures cannot eliminate pollution; all they can do is to mitigate its effects on the environment. Control measures are instituted “at the end of the (waste) pipe”. The usefulness of control measures will depend on the pollutant and the industrial circumstance. The main methods of pollution control, in no particular order, are:
- the capture and subsequent storage of pollutants
- filtration, whereby airborne or waterborne pollutants are removed from the waste stream by physical methods such as meshes, filters and other permeable barriers (such as coke)
- precipitation, whereby the pollutant is chemically precipitated and then captured in its transformed state or captured by physical methods such as an electrostatic charge
- destruction - for example, incineration, or neutralization, whereby pollutants are transformed chemically or biologically into substances which are less harmful
- dilution, whereby the pollutant is diluted or flushed in order to lessen its effects on any one organism or on an ecosystem; or concentration to lessen the effect of disposal
- evaporation or dissolution - for example, dissolving a gas in water
- utilization - for example, transforming a pollutant into a potentially useful (though not necessarily less toxic) product (such as sulphur dioxide into sulphuric acid or using solid waste as hard core or road bed)
- out-of-process recycling (where the recycling is not an integral part of the production process)
- media-shift, whereby a waste-stream is diverted from one medium, such as air, soil or water, to another, on the rationale that the medium-shift makes the pollutant less harmful
- state-changes—a change to the solid, liquid or gaseous state on the rationale that the new state is less harmful.
Pollution remediation
Remediation is needed to the extent that pollution prevention and control fail. It is also very expensive, with the costs not always accruing to the polluter. The modes of remediation are:
The clean-up of contaminated sites
Clean-up has a common sense meaning, as when an employer is required to “clean up his act”, which can mean a large number of different things. Within environmental protection, clean-up is a technical term meaning a branch or a mode of remediation. Even within this restricted use of the term, clean-up can mean (1) the removal of pollutants from a contaminated site or (2) the rehabilitation of a site so that it is restored to its full use-potential. Again, clean-up sometimes refers to nothing more than the containment of pollutants within a site, area or body of water—for example, by capping, sealing or the construction of an impermeable floor.
To be successful, clean-up has to be 100% effective, with full protection for workers, bystanders and the general public. A further consideration is whether the clean-up materials, methods and technology do not create further hazards. Though it is desirable to use engineering controls to protect clean-up workers, there will almost always be a need for appropriate personal protective equipment. Normally, workers engaged in remediation are classified as hazardous-waste workers, though aspects of such work are undertaken by fire fighters and municipal workers, among others.
A large number of physical, chemical, biological and biotechnological agents and methods are used in the clean-up of contaminated sites.
Hazardous-waste treatment
Most treatment of hazardous (or toxic) waste now takes place in purpose-built facilities by hazardous-waste workers. From an environmental point of view, the test of effectiveness of a hazardous-waste facility is that it produces no outputs which are not inert or virtually inert, such as silica, insoluble inorganic compounds, insoluble and non-corrosive slags, gaseous nitrogen or carbon dioxide - though carbon dioxide is a “greenhouse gas” which causes climate change and is, thus, a further environmental detriment.
A further test is that the facility be energy efficient - that is, energy is not wasted - and as energy non-intensive as possible (i.e., the ratio of energy use to the volume of waste treated be as low as possible). A general rule of thumb (it is fortunately not a universal law) is that the more effective the pollution (or waste) abatement strategy, the more energy is consumed, which by sustainable development criteria is another detriment.
Even when the workers are properly protected, it is easy to see the drawbacks of hazardous-waste treatment as a mode of addressing pollution. Pollution prevention methods can be applied to the operation of the treatment process but they cannot be applied to the principal “input” - the waste to be treated. Hazardous-waste treatment facilities will usually require at least as much energy to treat the waste as was expended in its creation, and there will always be further waste as an output, however inert or non-toxic.
Spills and leaks
The same considerations will apply to chemical spills and leaks as to the clean-up of contaminated sites, with the further hazards caused by the urgency of the clean-up. Workers cleaning up spills and leaks are almost always emergency workers. Depending on the scale and the nature of the pollutant, leaks and spills can become major industrial accidents.
The Modes of Pollution Prevention
Definition and philosophy
The definition of pollution prevention may seem to be a trivial matter, but it is important because advocates of pollution prevention want, as a principle of policy, to see a single-minded and aggressive prevention strategy at the expense of control methods, and to avoid remediation. The more strictly pollution prevention is defined, they say, the more likely it is to succeed as a practical strategy. Conversely, the more widely employers are allowed to define the term, the more likely their activities are to result in a mix of the same old (failed) strategies. Employers sometimes reply that even toxic waste can have a market value, and control methods have their place, so pollution is really only potential pollution. Besides, zero discharge is impossible and leads only to false expectations and misguided strategies. Proponents of pollution prevention respond that unless we have zero discharge as an aim or practical ideal, pollution prevention will not succeed and environmental protection will not improve.
Most of the strict definitions of pollution prevention have, as a sole or central element, the avoidance of the use of chemicals which result in pollutants so that pollution is not created in the first place. Some of the most important definitional controversies concern recycling, which is dealt with in the context of pollution prevention below.
Objectives
One possible objective of pollution prevention is zero discharge of pollutants. This is sometimes referred to as “virtual elimination”, since even zero discharge cannot solve the problem of contaminants already in the environment. Zero discharge of pollutants is possible using pollution prevention methods (while control methods cannot achieve zero in theory and are even less effective in practice, usually owing to lax enforcement). For instance, we can envisage automobile production in which there is zero discharge of pollutants from the plant; other waste is recycled and the product (the car) consists of parts which are reusable or recyclable. Certainly, zero discharge of specific pollutants has been achieved - for example, by modifying the production process in wood pulp mills so that no dioxins or furans are discharged in the effluent. The aim of zero discharge has also been written into environmental laws and into the policies of bodies commissioned to abate pollution.
In practice, zero discharge often gives way to target reductions - for example, a 50% reduction in pollution emissions by such-and-such a year. These targets or interim targets are usually in the form of “challenges” or aims by which to measure the success of the pollution prevention programme. They are rarely the product of a feasibility analysis or calculation, and there are invariably no penalties attached to failure to attain the target. Nor are they measured with any precision.
Reductions would have to be measured (as opposed to estimated) by variations on the formula:
Pollution (P) = Toxicity of the pollutant (T) × Volume (V) of the discharges
or:
P = T x V x E (exposure potential).
This is very difficult in theory and expensive in practice, though it could be done in principle by utilizing hazard assessment techniques (see below). The whole issue suggests that resources would be better allocated elsewhere - for example, in ensuring that proper pollution prevention plans are produced.
In regard to chemical pesticides, the objective of use-reduction can be achieved by the methods of integrated pest management (IPM), though this term, too, is capable of a wide or a strict definition.
Methods
The main methods of pollution prevention are:
- The elimination or phasing out of specific hazardous chemicals
- Input substitution - replacing a toxic or hazardous substance with a non-toxic or less hazardous substance or with a non-toxic process. Examples are the substitution of water-based for synthetic organic dyes in the printing industry; water - or citrus-based solvents for organic solvents; and, in some applications, the substitution of vegetable for mineral oils. Examples of non-chemical substitution include the substitution of pellet blasting technology for the use of fluid chemical paint strippers; the use of high-pressure hot water systems instead of caustic cleaning; and the substitution of kiln-drying for the use of pentachlophenols (PCPs) in the lumber industry.
In all cases, it is necessary to perform a substitution analysis to ensure that substitutes are genuinely less hazardous than what they replace. This is at least a matter of organized common sense, and at best the application of hazard assessment techniques (see below) to the chemical and its proposed substitute.
- Product reformulation - substituting for an existing end-product an end-product which is non-toxic or less toxic upon use, release or disposal
Whereas input substitution refers to the raw materials and adjuncts at the “front end” of the production process, product reformulation approaches the issue from the final product end of the production cycle.
General programmes to produce products which are more environmentally benign are examples of “economic conversion”. Examples of particular measures in the area of product reformulation include the production of rechargeable batteries instead of throw-away types and the use of water-based product coatings instead of those based on organic solvents and the like.
Again, substitution analysis will be necessary to ensure that the net environmental benefit is greater for the reformulated products that it is for the originals.
- Production unit redesign modernization or modification, which results in less chemical use or in the use of less toxic substances.
- Improved operation and maintenance of the production unit and production methods, including better housekeeping, more efficient production quality control, and process inspections.
Examples are spill prevention measures; the use of spill-proof containers; leak prevention; and floating lids for solvent tanks.
- Using less and reusing more. For instance, some degreasing operations take place too frequently on a single item. In other cases, chemicals can be used more sparingly in each operation. De-icing fluids can sometimes be reused, a case of “extended use”.
- Closed-loop methods and in-process recycling. Strictly speaking, a closed-loop process is one in which there are no emissions into the workplace or into the outside environment, not even waste water into surface water or carbon dioxide into the atmosphere. There are only inputs, finished products, and inert or non-toxic wastes. In practice, closed-loop methods eliminate some, but not all, hazardous releases. To the extent that this is achieved, it will count as a case of in-process recycling (see below).
Recycling
Any definition of pollution prevention is likely to result in a number of “grey areas” in which it is not easy to distinguish prevention measures from emission controls. For instance, to qualify as a prevention method, a phase of a production process may have to be “an integral part of the production unit”, but how far away the phase has to be from the periphery of the production process in order to qualify as a prevention measure is not always clear. Some processes may be so remote from the heart of an operation that they look more like an “add on” process and, thus, more like an “end of pipe” control measure than a prevention method. Again, there are unclear cases like a waste pipe that provides the feedstock for a neighbouring plant: taken together, the two plants provide a kind of closed loop; but the “upstream” plant still produces effluent and, thus, fails the prevention test.
Similarly with recycling. Conventionally, there are three types of recycling:
- in-process recycling - for example, when dry-cleaning solvent is filtered, cleaned and dried, then reused within a single process
- out-of-process but on-site, as when pesticide production waste is cleaned and then reused as the so-called inert base in a new production run
- out-of-process and off-site.
Of these, the third is usually ruled out as not qualifying as pollution prevention: the more remote the recycling site, the less of a guarantee that the recycled product is actually reused. There are also hazards in the transporting of waste to be recycled, and the financial uncertainty that the waste will have a continuous market value. Similar, though less acute, considerations apply to out-of-process but on-site recycling: there is always a possibility that the waste will not actually be recycled or, if recycled, not actually reused.
In the initial pollution prevention strategies of the 1980s, on-site but out-of-process recycling was ruled out as not being a genuine pollution prevention measure. There was a fear that an effective pollution prevention programme would be compromised or diluted by too great an emphasis on recycling. In the mid-1990s, some policy-makers are prepared to entertain on-site, out-of-process recycling as a legitimate pollution prevention method. One reason is that there are genuine “grey areas” between prevention and control. Another reason is that some on-site recycling really does do what it is supposed to do, even though it may not technically qualify as pollution prevention. A third reason is business pressure: employers see no reason why techniques should be ruled out it they serve the purposes of a pollution prevention programme.
Pollution prevention planning
Planning is an essential part of pollution prevention methodology, not least because the gains in both industrial efficiency and environmental protection are likely to be in the longer term (not immediate), reflecting the sort of planning that goes into product design and marketing. The production of periodic pollution prevention plans is the most usual way of realizing pollution prevention planning. There is no single model for such plans. One proposal envisages:
- aims and objectives
- chemical inventories and estimates of discharges into the environment
- pollution prevention methods used and methods proposed
- responsibilities and action in the event of the plan not being fulfilled or realized.
Another proposal envisages:
- a review of production processes
- identification of pollution prevention opportunities
- a ranking of the opportunities and a schedule for the implementation of the selected options
- measures of the success of the plan after the implementation period.
The status of such plans varies widely. Some are voluntary, though they can be spelled out in law as a (voluntary) code of practice. Others are mandatory in that they are required (1) to be kept on-site for inspection or (2) submitted to a regulatory authority on completion or (3) submitted to a regulatory authority for some form of scrutiny or approval. There are also variations, such as requiring a plan in the event that a “voluntary” plan is, in some way, inadequate or ineffective.
The degree to which mandatory plans are prescriptive also varies - for example, in regard to penalties and sanctions. Few authorities have the power to require specific changes in the content of pollution prevention plans; almost all have the power to require changes in the plan in the event that the formal requirements have not been met - for example, if some plan headings have not been addressed. There are virtually no examples of penalties or sanctions in the event that the substantive requirements of a plan have not been met. In other words, legal requirements for pollution prevention planning are far from traditional.
Issues surrounding the production of pollution prevention plans concern the degree of confidentiality of the plans: in some cases, only a summary becomes public, while in other cases, plans are released only when the producer fails in some way to comply with the law. In almost no cases do the requirements for pollution prevention planning override existing provisions regarding the trade secrecy or the business confidentiality of inputs, processes or the ingredients of products. In a few cases, community environmental groups have access to the planning process, but there are virtually no cases of this being required by law, nor are the legal rights of workers to participate in the production of plans widespread.
Legislation
In the Canadian provinces of British Columbia and Ontario, pollution prevention measures are “voluntary”; their effectiveness depends on “moral suasion” on the part of governments and environmentalists. In the United States, about half (26) of the states have some form of legislation, while in Europe, several northern countries have legislated clean technology programmes. There is quite a wide variety in both the content and the effectiveness of such legislation. Some laws define pollution prevention strictly; others define it widely or loosely and cover a wide variety of environmental protection activities concerning pollution and waste, not just pollution prevention. The New Jersey law is highly prescriptive; those of the Commonwealth of Massachusetts and the States of Minnesota and Oregon involve a high degree of government scrutiny and assistance; that of Alaska is little more than a statement of the government’s intentions.
Health, safety and employment
Pollution prevention is of central concern to occupational health: if the use of toxic substances decreases, there will almost always be a corresponding decrease in worker exposure to toxic substances and, thus, in industrial diseases. This is a prime case of prevention “at the source” of the hazard and, in many cases, the elimination of hazards by “engineering controls”
(i.e., methods), the first and best line of defence against chemical hazards. However, such preventive measures are different from one traditional strategy, which is the “total isolation” or the “total enclosure” of a chemical process. While total enclosure is highly useful and highly desirable, it does not count as a pollution prevention method since it controls, rather that reduces intrinsically, an existing hazard.
The pollutants which pose hazards to workers, communities and the physical environment alike, have usually been addressed primarily because of their impact on human communities (environmental health). Though the greatest exposures are often received by workers within a workplace (workplace pollution), this has not, so far, been the prime focus of pollution prevention measures. The Massachusetts legislation, for instance, aims to reduce the risks to the health of workers, consumers and the environment without shifting the risks between workers, consumers and parts of the environment (New Jersey is similar). But there was no attempt to focus on workplace pollution as a major detriment, nor was there a requirement to accord a primacy to the chief human exposures to hazards - often the workers. Nor is there any requirement to train workers in the discipline of pollution prevention.
There are several reasons for this. The first is that pollution prevention is a new discipline in the context of a general, traditional failure to see environmental protection as a function of processes utilized and adopted within workplaces. A second reason is that worker-management co-determination in the area of environmental protection is not well advanced. Workers in many countries have legal rights, for instance, to joint workplace health and safety committees; to refuse unsafe or unhealthy work; to health and safety information; and to training in health and safety issues and procedures. But there are few legal rights in the parallel and often overlapping area of environmental protection, such as the right to joint union-management environment committees; the right of employees to “blow the whistle” (go public) on an employer’s anti-environmental practices; the right to refuse to pollute or to degrade the outside environment; the right to environmental information; and the right to participate in workplace environmental audits (see below).
The impacts of pollution prevention planning on employment are hard to gauge. The explicit aim of pollution prevention initiatives is often to increase industrial efficiency and environmental protection at the same time and by the same set of measures. When this happens, the usual effect is to decrease overall employment within any given workplace (because of technological innovation) but to increase the skills required and then to increase job security (because there is planning for a longer-term future). To the extent that the use of raw materials and adjuncts is reduced, there will be decreased chemical manufacturing employment, though this is likely to be offset by the implied transition of feedstock to speciality chemicals and by the development of alternatives and substitutes.
There is one aspect of employment which pollution prevention planning cannot address. Pollution emissions from a single facility may decrease but to the extent that there is an industrial strategy to create wealth and value-added employment, an increase in the number of production facilities (however “clean”) will tend to nullify the environmental protection gains already achieved. The most notorious failing in environmental protection measures - that pollution emission reductions and controls are nullified by an increase in the number of sources - applies, unfortunately, to pollution prevention as well as to any other form of intervention. Ecosystems, according to one respected theory, have a “carrying capacity”, and that limit can be reached equally by a small number of highly polluting or “dirty” sources or by a correspondingly large number of clean ones.
Workplace environmental audits
Pollution prevention planning can form part of or be accommodated in a workplace environmental audit. Though there are many versions of such audits, they are likely to be in the form of a “site audit” or “production audit”, in which the whole production cycle is subjected to both an environmental and a financial analysis.
There are roughly three areas of sustainable development and environmental protection which can be covered in a workplace audit:
- the conservation of natural resource inputs - for example, minerals, water and wood products
- energy use, which may also include consideration of energy sources, energy efficiency, energy intensiveness and energy conservation
- pollution prevention, control and remediation.
To the extent that pollution prevention is successful, there will be a corresponding decrease in the importance of control and remediation measures; pollution prevention measures can form a major part of a workplace environmental audit.
Traditionally, businesses were able to “externalize” environmental detriments through such means as the profligate use of water or unloading their wastes onto the outside community and the environment. This has led to demands for taxes on the “front end” such as water use or on “outputs” such as environmentally unfriendly products or on wastes (“pollution taxes”).
In this way, costs to business are “internalized”. However, it has proved difficult to put the right price on the inputs and on the detriments - for example, the cost to communities and the environment of wastes. Nor is it clear that pollution taxes reduce pollution in proportion to the amounts levied; taxes may well “internalize” costs, but they otherwise only add to the cost of doing business.
The advantage of environmental auditing is that the audit can make economic sense without having to “cost” externalities. For instance, the “value” of waste can be calculated in terms of resource input loss and energy “non-utilization” (inefficiency) - in other words, of the difference in value between resources and energy on one side and the value of the product on the other. Unfortunately, the financial side of pollution prevention planning and its part in workplace environmental audits is not well advanced.
Hazard assessment
Some pollution prevention schemes work without any hazard evaluation - that is, without criteria to decide whether a plant or facility is more or less environmentally benign as a result of pollution prevention measures. Such schemes may rely on a list of chemicals which are objects of concern or which define the scope of the pollution prevention programme. But the list does not grade chemicals as to their relative hazardousness, nor is there a guarantee that a chemical substitute not on the list is, in fact, less hazardous than a listed chemical. Common sense, not scientific analysis, tells us how to go about implementing a pollution prevention programme.
Other schemes rest on criteria for assessing hazardousness, that is, on hazard assessment systems. They work, essentially, by laying down a number of environmental parameters, such as persistence and bioaccumulation in the environment, and a number of human health parameters which serve as measures of toxicity - for example, acute toxicity, carcinogenicity, mutagenicity, reproductive toxicity and so on.
There is then a weighted scoring system and a decision procedure for scoring those parameters on which there is inadequate information on the chemicals to be scored. Relevant chemicals are then scored and ranked, then (often) assembled in groups in descending order of hazardousness.
Though such schemes are sometimes devised with a specific purpose in mind - for example, for assessing priorities for control measures or for elimination (banning) - their essential use is as an abstract scheme which can be used for a large variety of environmental protection measures, including pollution prevention. For instance, the top group of scored chemicals could be the prime candidates for a mandatory pollution prevention programme, or they could be candidates for phasing-out or substitution. In other words, such schemes do not tell us how much we should reduce environmental health hazards; they tell us only that any measures we take should be informed by the hazard assessment scheme.
For instance, if we have to make decisions about substituting a less hazardous chemical for a more dangerous one, we can use the scheme to tell us whether, prima facie, the substitution decision is a good one: we run both chemicals through the scheme to determine whether there is a wide or merely a narrow gap between them regarding their hazardousness.
There are two sorts of considerations which rarely fall within the scope of hazard assessment schemes. The first is exposure data, or the potential for human exposure to the chemical. The latter is difficult to calculate, and, arguably, it distorts the “intrinsic hazard” of the chemicals concerned. For instance, a chemical could be accorded an artificially low priority on the grounds that its exposure potential is low; though it may, in fact, be highly toxic and relatively easy to deal with.
The second sort of consideration is the socioeconomic impact of eliminating or reducing the use of the chemical concerned. While we can start to make substitution decisions on the basis of the hazard analysis, we would have to make a further and distinct socioeconomic analysis and consider, for example, the social utility of the product associated with the chemical use (which may, e.g., be a useful drug), and we would also have to consider the impact on workers and their communities. The reason for keeping such analysis separate is that it is impossible to score the results of a socioeconomic analysis in the same way that the intrinsic hazards of chemicals are scored. There are two entirely distinct sets of values with different rationales.
However, hazard assessment schemes are crucial in assessing the success of pollution prevention programmes. (They are also relatively new, both in their impact and their utility.) For instance, it is possible to apply them without reference to risk assessments, risk analysis and (with reservations) without reference to cost-benefit analysis. An earlier approach to pollution was to first do a risk assessment and only then decide what sort of action, and how much, was necessary to reduce the risk to an “acceptable” level. The results were rarely dramatic. Hazard assessment, on the other hand, can be utilized very quickly and in such a way that it does not delay or compromise the effectiveness of a pollution prevention programme. Pollution prevention is, above all, a pragmatic programme capable of constantly and speedily addressing pollution issues as they arise and before they arise. It is arguable that traditional control measures have reached their limit and only the implementation of comprehensive pollution prevention programmes will be capable of addressing the next phase of environmental protection in a practical and effective way.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."