Children categories

77. Chemical Processing (8)

77. Chemical Processing
Chapter Editors: Jeanne Mager Stellman and Michael McCann
Table of Contents
Tables and Figures
Chemical Industry
L. De Boer
Developing a Process Safety Management Programme
Richard S. Kraus
Major Unit Operations and Processes: An Overview
Sydney Lipton
Examples of Chemical Processing Operations
Chlorine and Caustic Production
The Chlorine Institute, Inc.
Paint and Coating Manufacture
Michael McCann
Plastics Industry
P.K. Law and T.J. Britton
Biotechnology Industry
Susan B. Lee and Linda B. Wolfe
Pyrotechnics Industry
J. Kroeger
Tables
Click a link below to view table in article context.
1. Chemical industry employment in selected countries
2. Some general site selection factors
3. Plant siting safety considerations
4. Facilities generally separated in overall plant layouts
5. General considerations in a process unit layout
6. Steps for limiting inventory
7. Tank separation & location considerations
8. Pumps in the chemicals process industry
9. Potential explosion sources in equipment
10. Volatile products of the decomposition of plastics
11. Microorganisms of industrial importance
12. Raw materials used in the manufacture of pyrotechnics
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

78. Oil and Natural Gas (1)

78. Oil and Natural Gas
Chapter Editor: Richard S. Kraus
Table of Contents
Petroleum Refining Process
Richard S. Kraus
Tables
Click a link below to view table in article context.
1. Summary of the history of refining processing
2. Principal products of crude oil refining
3. Overview of petroleum refining processes
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

79. Pharmaceutical Industry (2)

79. Pharmaceutical Industry
Chapter Editor: Keith D. Tait
Table of Contents
Tables and Figures
Pharmaceutical Industry
Keith D. Tait
Case Study: Effects of Synthetic Oestrogens on Pharmaceutical Workers: A United States Example
Dennis D. Zaebst
Tables
Click a link below to view table in article context.
1. Major categories of pharmaceutical agents
2. Solvents used in the pharmaceutical industry
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

80. Rubber Industry (12)

80. Rubber Industry
Chapter Editors: Louis S. Beliczky and John Fajen
Table of Contents
Tables and Figures
General Profile
Louis S. Beliczky and John Fajen
Rubber Tree Cultivation
Alan Echt
Tyre Manufacturing
James S. Frederick
Non-Tyre Industrial Products
Ray C. Woodcock
Case Study: Salt Bath Vulcaization
Beth Donovan Reh
1,3-Butadiene
Ronald L. Melnick
Engineering Controls
Ray C. Woodcock
Safety
James R. Townhill
Epidemiological Studies
Robert Harris
Rubber Contact Dermatitis and Latex Allergy
James S. Taylor and Yung Hian Leow
Ergonomics
William S. Marras
Environmental and Public Health Issues
Thomas Rhodarmer
Tables
Click a link below to view table in article context.
1. Some important rubber polymers
2. Worldwide rubber consumption for 1993
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Non-Tyre Industrial Products
Rubber products are made for countless applications, using processes similar to those described for tyre manufacturing. Non-tyre products, however, use a much greater variety of polymers and chemicals to give them the properties they need (see table 1). Compounds are carefully designed to reduce hazards such as dermatitis and nitrosamines in the factory and in products like surgical supplies, respirators and baby bottle nipples that are used in contact with the body. Often processing equipment is on a smaller scale than in tyre making, with more use of mill mixing. Roofing and landfill membranes are made on the largest calenders in the world. Some companies specialize in compounding rubber to the specifications of others who process it into many different kinds of products.
Reinforced products such as drive belts, air brake diaphragms and footwear are built up from calendered rubber, coated fabric or cord on a revolving drum or stationary form. Curing is usually by compression moulding to fix the final shape, sometimes using steam pressure and a bladder or airbag as with a tyre. More synthetic polymers are used in non-tyre products. They are not as sticky as natural rubber, so more solvent is used to clean and make the built-up layers tacky. Milling, calendering and solvents or adhesives are bypassed in some cases by going directly from the mixer to a cross-head extruder to build the product.
Non-reinforced products are formed and cured by transfer or injection moulding, extruded and cured in a hot air oven or formed in a compression mould from a pre-cut slug. Sponge rubber is made by agents in the compound that release gas when heated.
Rubber hose is built by braiding, knitting or spinning reinforcing cord or wire onto an extruded tube supported by air pressure or a solid mandrel, then extruding a cover tube over it. An extruded lead cover or nylon cross-wrap is then put on the hose for compression moulding and removed after curing, or else the hose is put into the pressurized steam vulcanizer bare. Nylon cross-wrap or extruded plastic are increasingly replacing the lead. Automotive curved hose is cut and pushed onto shaped mandrels for curing; in some cases robots are taking over this strenuous manual labour. A process also exists that uses chopped fibre for reinforcement and a movable die in the extruder to shape the hose.
Cements mixed from rubber and solvent are used to coat fabric for a host of products. Toluene, ethyl acetate and cyclohexane are common solvents. Fabric is dipped in thin cement, or rubber can be built up in increments of a few micrometres by applying thicker cement under a knife-edge over a roller. Curing is done on a continuous rotational vulcanizer or in an explosion-protected hot-air oven. Latex processes are being developed for coated fabrics to replace the cements.
Rubber cements are also commonly used as adhesives. Hexane, heptane, naphtha and 1,1,1-trichloroethane are common solvents for these products, but hexane is being replaced because of toxicity.
Latex is a typically very alkaline suspension of natural or synthetic rubber in water. Forms for gloves and balloons are dipped, or the latex compound can be foamed for carpet backing, extruded into an acetic acid coagulant solution and washed to produce thread, or spread on fabric. The product is dried and cured in an oven. Natural rubber latex is widely used in medical gloves and devices. Gloves are powdered with cornstarch, or treated in a chlorine solution to de-tackify the surface. Powder-free gloves are reportedly subject to spontaneous combustion when stored in large quantity in a hot area.
Hazards and Precautions
Rubber processing hazards include exposure to hot surfaces, pressurized steam, solvents, processing aids, curing fumes and noise. Dusting agents include stearates, talc, mica and cornstarch. The organic dusts are explosive. Finishing adds a variety of hazards such as punching, cutting, grinding, printing ink solvents and alkaline or acidic surface treatment washes.
For precautions, see the articles “Engineering Controls” and “Safety” in this chapter.
Microwave, electron beam and ultrasonic vulcanization are being developed to generate heat within the rubber instead of transferring it inefficiently from outside to inside. The industry is working hard to eliminate or find safer substitutes for lead, dusting agents and volatile organic solvents and to improve compounds for better and safer properties in processing and use.
Case Study: Salt Bath Vulcanization
Salt bath vulcanization is a liquid curing method (LCM), a common continuous vulcanization (CV) method. CV methods are desirable for producing products such as tubing, hoses and weather stripping. Salt is a good choice for a CV method because it requires relatively short-length curing units—it has good heat exchange properties and can be used at the necessary high temperatures (177 to 260°C). Also, the salt does not cause surface oxidation, and it is easy to clean off with water. The entire operation involves at least four main processes: the rubber is fed through a cold-feed vented (or vacuum) extruder, conveyed through the salt bath, rinsed and cooled and then cut and processed according to specification. The extrudate is either immersed in or showered by the molten salt, which is a eutectic (easily fusible) blend of nitrate and nitrite salts, such as 53% potassium nitrate, 40% sodium nitrite and 7% sodium nitrate. The salt bath is generally enclosed with access doors on one side and electric heating coils on the other.
A disadvantage of the salt bath LCM is that it has been associated with the formation of nitrosamines, which are suspected human carcinogens. These chemicals are formed when a nitrogen (N) and an oxygen (O) from a “nitrosating” compound bind to the amino group nitrogen (N) of the amine compound. The nitrate and nitrite salts used in the salt bath serve as nitrosating agents and combine with amines in the rubber compound to form nitrosamines. Rubber compounds that are nitrosamine precursors include: sulphenamides, secondary sulphenamides, dithiocarbamates, thiurams and diethylhydroxylamines. Some rubber compounds actually contain a nitrosamine, such as nitrosodiphenylamine (NDPhA), a retarder, or dinitrosopentamethylenetetramine (DNPT), a blowing agent. These nitrosamines are weakly carcinogenic, but they can “trans-nitrosate”, or transfer their nitroso- groups to other amines to form more carcinogenic nitrosamines. Nitrosamines that have been detected at salt bath operations include: nitrosodimethylamine (NDMA), nitrosopiperidine (NPIP), nitrosomorpholine (NMOR), nitrosodiethylamine (NDEA) and nitrosopyrrolidine (NPYR).
In the United States, both the Occupational Safety and Health Administration (OSHA) and the NIOSH consider NDMA to be an occupational carcinogen, but neither has established an exposure limit. In Germany, there are strict regulations for occupational exposures to nitrosamines: in general industry, the total nitrosamine exposure may not exceed 1 μg/m3. For certain processes, such as rubber vulcanization, total nitrosamine exposures may not exceed 2.5 μg/m3.
Eliminating the nitrosamine formation from CV operations can be done by either reformulating the rubber compounds or using a CV method other than a salt bath, such as hot air with glass beads or microwave curing. Both changes require research and development to ensure that the final product has all the same desirable properties as the former rubber product. Another option to reduce exposures is local exhaust ventilation. Not only does the salt bath need to be enclosed and properly ventilated, but also other areas along the line, such as places where the product is cut or drilled, need sufficient engineering controls to ensure that worker exposures be kept low.
1,3-Butadine
A colourless gas produced as a co-product in the manufacture of ethylene, 1,3-butadiene is used largely as a starting material in the manufacture of synthetic rubber (e.g., styrene-butadiene rubber (SBR) and polybutadiene rubber) and thermoplastic resins.
Health Effects
Animal studies. Inhaled butadiene is carcinogenic at multiple organ sites in rats and mice. In rats exposed to 0, 1,000, or 8,000 ppm butadiene for 2 years, increased tumour incidences and/or dose-response trends were observed in the exocrine pancreas, testis and brain of males and in the mammary gland, thyroid gland, uterus and Zymbal gland of females. Inhalation studies of butadiene in mice were conducted at exposures ranging from 6.25 to 1,250 ppm. Particularly noteworthy in mice were the induction of early malignant lymphomas and uncommon haemangiosarcomas of the heart. Malignant lung tumours were induced at all exposure concentrations. Other sites of tumour induction in mice included the liver, forestomach, Harderian gland, ovary, mammary gland and preputial gland. Non-neoplastic effects of butadiene exposure in mice included bone marrow toxicity, testicular atrophy, ovarian atrophy and developmental toxicity.
Butadiene is genotoxic to bone marrow cells of mice, but not rats, producing increases in sister chromatid exchanges, micronuclei and chromosomal aberrations. Butadiene is also mutagenic to Salmonella typhimurium in the presence of metabolic activation systems. The mutagenic activity of butadiene has been attributed to its metabolism to mutagenic (and carcinogenic) epoxide intermediates.
Human studies. Epidemiological studies have consistently found excess mortality from lymphatic and haematopoietic cancers associated with occupational exposure to butadiene. In the butadiene production industry, increases in lymphosarcomas in production workers were concentrated among men who were first employed before 1946. A case-control study of lymphatic and haematopoietic cancers in eight SBR facilities identified a strong association between leukaemia mortality and exposure to butadiene. Important characteristics of the leukaemia cases were that most were hired before 1960, worked in three of the plants and had been employed for at least 10 years in the industry. The International Agency for Research on Cancer (IARC) has classified as 1,3-butadiene probably carcinogenic to humans (IARC 1992).
A recent epidemiological study has provided data that confirm the excess in leukaemia mortality among SBR workers exposed to butadiene (Delzell et al. 1996). The site correspondence between lymphomas induced in mice exposed to butadiene and lymphatic and haematopoietic cancers associated with occupational exposure to butadiene is especially noteworthy. Furthermore, estimates of human cancer risk derived from data of butadiene-induced lymphomas in mice are similar to estimates of leukaemia risk determined from the new epidemiological data.
Industrial Exposure and Control
Surveys of exposure in industries where butadiene is produced and utilized were conducted by the US National Institute for Occupational Safety and Health (NIOSH) in the mid-1980s. Exposures were greater than 10 ppm in 4% of the samples and less than 1 ppm in 81% of the samples. Exposures were not homogeneous within specific job categories, and excursions as high as 370 ppm were measured. Exposures to butadiene were probably much higher during the Second World War, when the synthetic rubber industry was undergoing rapid growth. Limited sampling from rubber tyre and hose manufacture plants were below the limit of detection (0.005 ppm) (Fajen, Lunsford and Roberts 1993).
Exposures to butadiene can be reduced by ensuring that fittings on closed-loop systems are not worn or incorrectly connected. Further measures to control potential exposures include: use of closed-loop systems for cylinder sampling, use of dual mechanical seals to control release from leaking pumps, use of magnetic gauges to monitor rail-car filling operations and use of a laboratory hood for cylinder voiding.
Engineering Controls
The manufacture of tyres and other rubber products exposes workers to a large variety of chemicals. These include many different powders, solids, oils and polymers used as compounding ingredients; anti-tack dusts to prevent sticking; mist, fumes and vapours generated by heating and curing rubber compounds; and solvents used for cements and process aids. The health effects related to most of these are not well known, except that they are usually chronic in nature rather than acute at typical exposure levels. Engineering controls are generally aimed at overall reduction of the level of dust, heated rubber emissions or curing fumes to which workers are exposed. Where there is exposure to specific chemicals, solvents or agents (such as noise) that are known to be harmful, control efforts can be targeted more specifically and in many cases the exposure can be eliminated.
Elimination or substitution of harmful materials is perhaps the most effective means of engineering control of hazards in rubber manufacturing. For example, β-naphthylamine contained as an impurity in an anti-oxidant was identified in the 1950s as a cause of bladder cancer and was banned. Benzene was once a common solvent but has been replaced since the 1950s by naphtha, or white gasoline, in which the benzene content has been steadily reduced (from 4-7% to commonly less than 0.1% of the mixture). Heptane has been used as a substitute for hexane and works just as well or better. Lead sheathing is being replaced by other materials for curing hose. Rubber compounds are being designed to reduce dermatitis in handling and the formation of nitrosamines in curing. Talcs used for anti-tack purposes are selected for low asbestos and silica content.
Rubber Compounding
Local exhaust ventilation is used for control of dust, mist and fumes in rubber compound preparation and mixing and in finishing processes involving buffing and grinding of rubber products (see figure 1). With good work practices and ventilation designs, dust exposures are usually well under 2 mg/m3. Effective maintenance of filters, hoods and mechanical equipment is an essential element of engineering control. Specific hood designs are given in the American Conference of Governmental Industrial Hygienists ventilation manual and the Rubber and the Plastics Research Association of Great Britain ventilation handbook (ACGIH 1995).
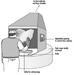
Figure 1. A canopy hood controls fumes in finishing a tube casting at an industrial rubber plant in Italy
Compounding chemicals have traditionally been scooped from bins into small bags on a weighing scale, then placed on a conveyor to be poured into the mixer or onto a mill. Dust exposures are controlled by a slotted side-draft hood behind the scale (see figure 2). and in some cases by slotted hoods at the edge of the stock bins. Dust control in this process is improved by substituting larger-particle-sized or granular forms for powders, by combining ingredients in a single (often heat-sealed) bag and by feeding compounds automatically from the storage bin to the transfer bag or directly to the mixer. Operator work practices also strongly influence the amount of dust exposure.
Figure 2. Slotted local exhaust ventilation at a compound weighing station
The Banbury mixer requires an effective enclosing hood to capture the dust from charging and to collect the fumes and oil mist coming from the heated rubber as it mixes. Well-designed hoods are often disrupted by drafts from pedestal fans used to cool the operator. Powered equipment is available to carry bags from pallets to the charging conveyor.
Mills are provided with canopy hoods to capture emissions of oil mist, vapours and fumes rising from the hot rubber. Unless more enclosed, these hoods are less effective in capturing dust when compounds are mixed on the mill or the mill is dusted with anti-tack powders (see figure 3). They are also sensitive to drafts from pedestal fans or misdirected general ventilation make-up air. A push-pull design has been used which places an air curtain in front of the operator directed up into the canopy. Mills are often raised to put the roller nip point out of the operator’s reach, and they also have a trip wire or bar in front of the operator to stop the mill in an emergency. Bulky gloves are worn that will be pulled into the nip before the fingers are caught.
Figure 3. A curtain at the edge of a canopy hood over a mixing mill helps contain dust.
Rubber slabs taken off mills and calenders are coated to keep them from sticking together. This is sometimes done by dusting the rubber with powder, but is now more often done by dipping it in a water bath (see figure 4). Applying the anti-tack compound this way greatly reduces dust exposure and improves housekeeping.
Figure 4. A rubber strip taken from a Banbury batch-off mill goes through a water bath to apply anti-tack compound.
Ray C. Woodcock
Dust and fumes are ducted to bag-house or cartridge-type dust collectors. In large installations, air is sometimes recirculated back into the factory. In that case, leak detection equipment is necessary to be sure contaminants are not recirculated. Odours from some ingredients such as animal glue make air recirculation undesirable. Rubber dust burns easily, so fire and explosion protection for ductwork and dust collectors are important considerations. Sulphur and explosive dusts such as cornstarch also have special fire-protection requirements.
Rubber Processing
Local exhaust hoods are often used at extruder heads to capture mist and vapours from the hot extrusion, which may then be directed into a water bath to cool it and suppress the emissions. Hoods are also used at many other emission points in the factory, such as grinders, dip tanks and laboratory test equipment, where air contaminants can easily be collected at the source.
The numbers and physical configurations of building stations for tyres and other products usually make them unsuitable for local exhaust ventilation. Confinement of solvents to covered containers as much as possible, along with careful work practices and adequate dilution air volume in the work area, are important for keeping exposures low. Gloves or applicator tools are used to minimize skin contact.
Curing presses and vulcanizers release large amounts of hot curing fumes when they are opened. Most of the visible emission is oil mist, but the mixture is also rich in many other organic compounds. Dilution ventilation is the control measure most often used, often in combination with canopy hoods or curtained enclosures over individual vulcanizers or groups of presses. Large volumes of air are required which, if not replaced by adequate make-up air, can disrupt ventilation and hoods in connecting buildings or departments. Operators should be positioned outside the hood or enclosure. If they must be under the hood, downdraft fresh air ventilators can be placed over their work stations. Otherwise, replacement air should be introduced adjacent to the enclosures but not directed into the canopy. The British occupational exposure limit for rubber curing fumes is 0.6 mg/m3 of cyclohexane soluble material, which is normally feasible with good practice and ventilation design.
Making and applying rubber cement presents special engineering control requirements for solvents. Mixing churns are sealed and vented to a solvent recovery system, while dilution ventilation controls vapour levels in the work area. The highest operator exposures come from reaching into churns to clean them. In applying rubber cement to fabric, a combination of local exhaust ventilation at emission points, covered containers, general ventilation in the workroom and properly directed make-up air controls worker exposure. Drying ovens are exhausted directly, or sometimes air is recirculated in the oven before it is exhausted. Carbon adsorption solvent recovery systems are the most common air-cleaning device. Recovered solvent is returned to the process. Fire-protection standards require that the flammable vapour concentration in the oven be maintained below 25% lower explosion limit (LEL), unless continuous monitoring and automatic controls are provided to ensure that the vapour concentration does not exceed 50% LEL (NFPA 1995).
Automation of processes and equipment often lowers exposure to airborne contaminants and physical agents by placing the operator at a greater distance, by confining the source or by reducing the generation of the hazard. Less physical strain on the body is also an important benefit of automation in processes and material handling.
Noise Control
Significant noise exposures often come from equipment such as braiders and belt grinders, air-exhaust ports, compressed air leaks and steam leaks. Noise-reducing enclosures are effective for braiders and grinders. Very effective silencers are made for air-exhaust ports. In some cases the ports can be ducted to a common header that vents elsewhere. Air noise from leaks can often be reduced by better maintenance, enclosure, design or good work practices to limit the noise cycle.
Work Practices
To prevent dermatitis and rubber allergies, rubber chemicals and fresh rubber batches should not come in contact with the skin. Where engineering controls are insufficient for this, long gauntlet gloves, or gloves and long-sleeved shirts, should be used to keep powders and rubber slabs off the skin. Work clothes should be kept separate from street clothing. Showers are recommended before changing to street clothing to remove residual contaminants from the skin.
Other protective equipment such as hearing protection and respirators may also be necessary at times. However, good practice dictates that priority always be given to substitution or other engineering solutions to reduce hazardous exposures in the workplace.
Safety
Mill Safety
Mills and calenders are used extensively throughout the rubber industry. Running nip accidents (getting caught in the rotating rolls) are major safety hazards during operation of these machines. In addition, there is a potential for accidents during repair and maintenance of these and other machines used in the rubber industry. This article discusses these safety hazards.
In 1973 in the United States, the National Joint Industrial Council for the Rubber Manufacturing Industry concluded that for in-running nip points, a safety device that depended on action of the operator could not be regarded as an effective method of preventing running nip accidents. This is especially true of mills in the rubber industry. Unfortunately, little has been done to force code changes. Currently there is only one safety device that does not require operator action to activate. The body bar is the only widely accepted automatic device that is an effective means of preventing mill accidents. However, even the body bar has limitations and cannot be used in all cases unless modifications are made to the equipment and work practice.
The problem of mill safety is not a simple one; there are several major issues involved:
- mill height
- the size of the operator
- auxiliary equipment
- the way the mill is worked
- the tack or stickiness of the stock
- stopping distance.
Mill height makes a difference as to where the operator works the mill. For mills less than
1.27 m high, where the height of the operator is greater than 1.68 m, there is a tendency to work too high on the mill or too close to the nip. This allows for a very short reaction time for the automatic safety to stop the mill.
The size of the operator also dictates how close the operator needs to get to the mill face to work the mill. Operators come in many different sizes, and often must operate the same mill. The majority of the time no adjustment is made to the mill safety devices.
Auxiliary equipment such as conveyors or loaders can often conflict with safety cables and ropes. Despite codes to the contrary, often the safety rope or cable is moved to allow for the operation of the auxiliary equipment. This can result in the operator working the mill with the safety cable behind the operator’s head.
While the height of the mill and the auxiliary equipment have a part in the way a mill is worked, there are other factors which enter into the picture. If there is no mixing roll below the mixer to distribute the rubber evenly on the mill, the operator will have to physically move the rubber from one side of the mill to the other by hand. The mixing and moving of the rubber exposes the operator to increased risk of strain or sprain injuries in addition to the hazard of the mill nip.
The tack or stickiness of the stock poses an additional hazard. If the rubber sticks to the mill roll and the operator has to pull it off the roll, a body bar becomes a safety hazard. Operators of mills with hot rubber have to wear gloves. Mill operators use knives. Tacky stock can grab a knife, glove or bare hand and pull it toward the running nip of the mill.
Even an automatic safety device will not be effective unless the mill can be stopped before the operator reaches the running nip of the mill. Stopping distances must be checked at least weekly and the brakes tested at the beginning of each shift. Dynamic electrical brakes must be checked on a regular basis. If the zero switch is not adjusted properly, the mill will move back and forth and damage to the mill will result. For some situations, disc brakes are preferred. With electrical brakes a problem can arise if the operator has activated the mill stop button and then tried an emergency mill stop. On some mills the emergency stop will not work after the mill stop button has been activated.
There have been some adjustments made that have improved mill safety. The following steps have greatly reduced exposure to running nip injuries on the mills:
- A body bar should be used on the working face of each mill, but only if the bar is adjustable for the height and reach of the operator.
- Mill brakes can be either mechanical or electrical, but they must be checked each shift and the distance checked weekly. The stopping distances should comply with the American National Standards Institute (ANSI) stopping distance recommendations.
- Where mixer mills have hot, tacky stock, a two-mill system has replaced the single-mill system. This has reduced operator exposure and improved the mixing of the stock.
- Where operators are required to move stock across a mill, a mixing roll should be added to reduce operator exposure.
- Current mill work practices have been reviewed to insure that the operator is not working too close to the running nip on the mill. This includes small lab mills, especially where a sample may require numerous passes through the running nip.
- Mill loaders have been added on mills to load stock. This has eliminated the practice of trying to load a mill using a fork truck, and has eliminated any conflict with the use of a body bar as a safety device.
Currently technology exists to improve mill safety. In Canada, for example, a rubber mill cannot be operated without a body bar on the working face or front of the mill. Countries receiving older equipment from other countries need to adjust the equipment to fit their workforce.
Calender Safety
Calenders have many configurations of machines and auxiliary equipment, making it difficult to be specific on calender safety. For a more in-depth study in calender safety, see National Joint Industrial Council for the Rubber Manufacturing Industry (1959, 1967).
Unfortunately, when a calender or any other piece of equipment has been transferred from one company to another or one country to another, often the accident history is not included. This has resulted in the removal of guards and in dangerous work practices that had been changed because of a prior incident. This has led to history repeating itself, with accidents that have occurred in the past reoccurring. Another problem is language. Machines with the controls and instructions in a different language from the user country makes safe operation more difficult.
Calenders have increased in speed. The braking ability of these machines has not always kept pace with the equipment. This is especially true around the calender rolls. If these rolls cannot be stopped in the recommended stopping distance, an additional method must be used to protect employees. If necessary, the calender should be equipped with a sensing device that will slow the machine when the rolls are approached during operation. This has proven very effective in keeping employees from getting too close to the rolls during the operation of the machine.
Some of the other major areas identified by the National Joint Industrial Council are still a source of injuries today:
- clearing jams and adjusting material
- running nip injuries, especially at wind-ups
- threading up
- communications.
An effective, well understood lockout programme (see below) will do much to reduce or eliminate injuries from the clearing of jams or the adjusting of material while the machine is in operation. Proximity devices that slow the rolls when they are approached may help deter an adjustment attempt.
Running nip injuries remain a problem, especially at wind-ups. Speeds at the wind-up must be adjustable to allow for a slow start-up at the beginning of the roll. Safeties must be available in the event of a problem. A device that slows the roll when it is approached will tend to discourage an attempt to adjust a liner or fabric during the wind-up. Telescoping rolls are a special temptation for even experienced operators.
The problem of threading-up incidents has increased with the speed and complexity of the calender train and the amount of auxiliary equipment. Here the existence of a single line control and good communications are essential. The operator may not be able to see all of the crew. Everyone must be accounted for and communications must be clear and easily understood.
The need for good communications is essential to safe operation when a crew is involved. Critical times are when adjustments are being made or when the machine is started at the beginning of a run or started after a shut-down which had been caused by a problem.
The answer to these problems is a well-trained crew that understands the problems of calender operation, a maintenance system that maintains all safety devices is working condition and a system that audits both.
Machine Lockout
The concept of machine lockout is not new. While lockout has been generally accepted in maintenance programmes, very little has been done to gain acceptance in the operating area. Part of the problem is the recognition of the hazard. A typical lockout standard requires that “if the unexpected movement of equipment or release of energy could cause injury to an employee then that equipment should be locked out”. Lockout is not limited to electrical energy, and not all energy can be locked out; some things must be blocked in position, pipes must be disconnected and blanked, stored pressure must be relieved. While the lockout concept is viewed in some industries as a way of life, other industries have not accepted it due to the fear of the cost of locking out.
Central to the concept of lockout is control. Where the person is at risk for injury as the result of movement, the power source(s) must be disabled and the person or persons at risk should have control. All situations requiring lockout are not easy to identify. Even when they are identified, it is not easy to change work practices.
Another key to a lockout programme which is often overlooked is the ease with which a machine or line can be locked out or the power isolated. Older equipment was not designed or installed with lockout in mind. Some machines were installed with a single breaker for several machines. Other machines have multiple power sources, making lockout more complicated. To add to this problem, motor control room breakers are often changed or feed additional equipment, and the documentation of the changes is not always kept current.
The rubber industry has seen general acceptance of lockout in maintenance. While the concept of protecting one’s self from the dangers of unexpected movement is not new, the uniform use of lockout is. In the past, maintenance personnel used different means to protect themselves. This protection was not always consistent due to other pressures such as production, and not always effective. For some of the equipment in the industry, the lockout answer is complex and not easily understood.
The tyre press is an example of a piece of equipment for which there is little consensus on the exact time and method for lockout. While the complete lockout of a press for an extensive repair is straightforward, there is no consensus about lockout in such operations as mould and bladder changes, mould cleaning and unjamming equipment.
The tyre machine is another example of difficulty in lockout compliance. Many of the injuries in this area have not been to maintenance personnel, but rather to operators and tyre technicians making adjustments, changing drums, loading or unloading stock or unjamming equipment and to janitorial employees cleaning the equipment.
It is difficult to have a successful lockout programme if the lockout is time consuming and difficult. Where possible, the means to disconnect should be available at the equipment, which helps with ease of identification and can eliminate or reduce the possibility of someone being in the danger zone when the energy is returned to the equipment. Even with changes that make identification easier, no lockout can ever be considered complete unless a test is made to be sure the correct power isolation devices were used. In the case of work with electrical wiring, a test should be made after the disconnect is pulled to ensure that all power has been disconnected.
An effective lockout programme must include the following:
- The equipment should be designed to facilitate a lockout for all energy sources.
- Lockout sources must be identified correctly.
- Work practices requiring lockout must be identified.
- All employees affected by lockout should have some training in lockout.
- Employees who are required to lockout should be trained and advised that lockout is expected and that anything less is unacceptable under any circumstances.
- The programme needs to be audited on a regular basis to make sure that it is effective.
Epidemiological Studies
In the 1920s and 1930s, reports from the United Kingdom showed that rubber workers had higher death rates than did the general population, and that the excess deaths were from cancers. Thousands of different materials are used in manufacturing rubber products and which if any of these might be associated with the excess deaths in the industry was not known. Continued concern for the health of rubber workers led to joint company-union occupational health research programmes within the US rubber industry at Harvard University and at the University of North Carolina. The research programmes continued through the decade of the 1970s, after which they were supplanted by jointly sponsored company-union health surveillance and health maintenance programmes based, at least in part, on findings of the research effort.
Work in the Harvard research programme focused generally on mortality in the rubber industry (Monson and Nakano 1976a, 1976b; Delzell and Monson 1981a, 1981b; Monson and Fine 1978) and on respiratory morbidity among rubber workers (Fine and Peters 1976a, 1976b, 1976c; Fine et al. 1976). An overview of the Harvard research has been published (Peters et al. 1976).
The University of North Carolina group engaged in a combination of epidemiological and environmental research. The early efforts were primarily descriptive studies of rubber workers’ mortality experience and investigations of conditions of work (McMichael, Spirtas and Kupper 1974; McMichael et al. 1975; Andjelkovich, Taulbee and Symons 1976; Gamble and Spirtas 1976; Williams et al. 1980; Van Ert et al. 1980). The major focus, however, was in analytic studies on associations between work-related exposures and disease (McMichael et al. 1976a; McMichael et al. 1976b; McMichael, Andjelkovich and Tyroler 1976; Lednar et al. 1977; Blum et al. 1979; Goldsmith, Smith and McMichael 1980; Wolf et al. 1981; Checkoway et al. 1981; Symons et al. 1982; Delzell, Andjelkovich and Tyroler 1982; Arp, Wolf and Checkoway 1983; Checkoway et al. 1984; Andjelkovich et al. 1988). Noteworthy were findings regarding associations between exposures to hydrocarbon solvent vapours and cancers (McMichael et al. 1975; McMichael et al. 1976b; Wolf et al. 1981; Arp, Wolf and Checkoway 1983; Checkoway et al. 1984) and associations between exposures to airborne particulate materials and pulmonary disability (McMichael, Andjelkovich and Tyroler 1976; Lednar et al. 1977).
At the University of North Carolina, the initial analytic studies of leukaemia among rubber workers showed excess cases among workers who had a history of working in jobs in which solvents were used (McMichael et al. 1975). Exposure to benzene, a common solvent in the rubber industry many years ago, and a recognized cause of leukaemia, was immediately suspected. More detailed analyses, however, showed that the excess leukaemias were generally lymphocytic, while exposures to benzene had commonly been associated with the myeloblastic type (Wolf et al. 1981). It was surmised that some agent other than benzene could be involved. A very painstaking review of records of solvent use and solvent sources of supply for one large company showed that use of coal-based solvents, including both benzene and xylene, had a much stronger association with lymphocytic leukaemia than did use of petroleum-based solvents (Arp, Wolf and Checkoway 1983). Coal-based solvents are generally contaminated with polynuclear aromatic hydrocarbons, including compounds which have been shown to cause lymphocytic leukaemia in experimental animals. Further analyses in this study showed an even stronger association of lymphocytic leukaemia with exposures to carbon disulphide and carbon tetrachloride than with exposures to benzene (Checkoway et al. 1984). Exposures to benzene are hazardous, and exposures to benzene in workplaces should be eliminated or minimized to the extent possible. A conclusion, however, that eliminating benzene from use in rubber processes will eliminate future excesses of leukaemia, particularly of lymphocytic leukaemia, among rubber workers may be incorrect.
Special studies at the University of North Carolina of rubber workers who had taken disability retirement showed that disabling pulmonary disease, such as emphysema, was more likely to have occurred among people with a history of work in curing, curing preparation, finishing and inspection than among workers in other jobs (Lednar et al. 1977). All of these work areas involve exposures to dusts and fumes which can be inhaled. In these studies it was found that a history of smoking generally more than doubled the risk of pulmonary disability retirement, even in the dusty jobs which themselves were associated with disability.
Epidemiological studies were under way in the European and Asian rubber industries (Fox, Lindars and Owen 1974; Fox and Collier 1976; Nutt 1976; Parkes et al. 1982; Sorahan et al. 1986; Sorahan et al. 1989; Kilpikari et al. 1982; Kilpikari 1982; Bernardinelli, Marco and Tinelli 1987; Negri et al. 1989; Norseth, Anderson and Giltvedt 1983; Szeszenia-Daborowaska et al. 1991; Solionova and Smulevich 1991; Gustavsson, Hogstedt and Holmberg 1986; Wang et al. 1984; Zhang et al. 1989) at about the same time and continued after those of Harvard and the University of North Carolina in the United States. Findings of excess cancers at various sites were commonly reported. Several studies showed an excess of lung cancer (Fox, Lindars and Owen 1974; Fox and Collier 1976; Sorahan et al. 1989; Szeszenia-Daborowaska et al. 1991; Solionova and Smulevich 1991; Gustavsson, Hogstedt and Holmberg 1986; Wang et al. 1984), associated, in some cases, with a history of work in curing. This finding was duplicated in some studies in the United States (Monson and Nakano 1976a; Monson and Fine 1978) but not in others (Delzell, Andjelkovich and Tyroler 1982; Andjelkovich et al. 1988).
The mortality experience among a cohort of workers in the German rubber industry has been reported (Weiland et al. 1996). Mortality from all causes and from all cancers was significantly elevated in the cohort. Statistically significant excesses in mortality from lung cancer and from pleural cancer were identified. The excess of mortality from leukaemia among German rubber workers barely failed to reach statistical significance.
A case-control study of lymphatic and haematopoietic cancers in eight styrene-butadiene rubber (SBR) facilities identified a strong association between leukaemia mortality and exposure to butadiene. The IARC has concluded that 1,3-butadiene is probably carcinogenic to humans (IARC 1992). A more recent epidemiological study has provided data that confirm the excess in leukaemia mortality among SBR workers exposed to butadiene (Delzell et al. 1996).
Over the years, epidemiological studies among rubber workers have led to the identification of workplace hazards and to improvements in their control. The area of occupational epidemiological research in greatest need of improvement at this time is assessment of past exposures of study subjects. Progress is being made in both research techniques and in databases in this area. Although questions regarding causal associations remain, continued epidemiological progress will surely lead to continued improvements in control of exposures in the rubber industry and, consequently, to continued improvement in the health of rubber workers.
Acknowledgement: I would like to recognize the pioneering efforts of Peter Bommarito, former president of the United Rubber Workers Union, who was primarily responsible for causing research to be done in the US rubber industry in the 1970s and 1980s on the health of rubber workers.
Rubber Contact Dermatitis and Latex Allergy
Contact Dermatitis
Adverse skin reactions have been reported frequently among workers who have direct contact with rubber and with the hundreds of chemicals used in the rubber industry. These reactions include irritant contact dermatitis, allergic contact dermatitis, contact urticaria (hives), aggravation of pre-existing skin diseases and other less common skin disorders such as oil folliculitis, xerosis (dry skin), miliaria (heat rash) and depigmentation from certain phenol derivatives.
Irritant contact dermatitis is the most frequent reaction and is caused by either acute exposure to strong chemicals or by cumulative exposure to weaker irritants such as those found in wet work and in repeated use of solvents. Allergic contact dermatitis is a delayed type of allergic reaction from the accelerators, vulcanizers, anti-oxidants and anti-ozonants which are added during rubber manufacture. These chemicals are often present in the final product and may cause contact dermatitis in both the end-product user as well as in rubber workers, especially Banbury, calender and extruder operators and assemblers.
Some workers acquire contact dermatitis through exposure in work which does not permit the use of chemical-protective clothing (CPC). Other workers also develop allergy to CPC itself, most commonly from rubber gloves. A valid positive patch test to the suspected allergen is the key medical test which is used to differentiate allergic contact dermatitis from irritant contact dermatitis. It is important to remember that allergic contact dermatitis may coexist with irritant contact dermatitis as well as with other skin disorders.
Dermatitis may be prevented by automated mixing and preblending of chemicals, provision of exhaust ventilation, substitution of known contact allergens with alternative chemicals and improved materials handling to reduce skin contact.
Natural Rubber Latex (NRL) Allergy
NRL allergy is an immunoglobulin E–mediated, immediate, Type I allergic reaction, most always due to NRL proteins present in medical and non-medical latex devices. The spectrum of clinical signs ranges from contact urticaria, generalized urticaria, allergic rhinitis (inflammation of nasal mucosa), allergic conjunctivitis, angio-oedema (severe swelling) and asthma (wheezing) to anaphylaxis (severe, life-threatening allergic reaction). Highest risk individuals are patients with spina bifida, health care workers and other workers with significant NRL exposure. Predisposing factors are hand eczema, allergic rhinitis, allergic conjunctivitis or asthma in individuals who frequently wear gloves, mucosal exposure to NRL and multiple surgical procedures. Fifteen deaths following NRL exposure during barium enema examinations have been reported to the US Food and Drug Administration. Thus the route of exposure to NRL proteins is important and includes direct contact with intact or inflamed skin and mucosal exposure, including inhalation, to NRL-containing glove powder, especially in medical facilities and in operating rooms. As a result, NRL allergy is a major worldwide medical, occupational health, public health and regulatory problem, with the number of cases having increased dramatically since the mid-1980s.
Diagnosis of NRL allergy is strongly suggested if there is a history of angio-oedema of the lips when inflating balloons and/or itching, burning, urticaria or anaphylaxis when donning gloves, undergoing surgical, medical and dental procedures or following exposure to condoms or other NRL devices. Diagnosis is confirmed by either a positive wear or use test with NRL gloves, a valid positive intracutaneous prick test to NRL or a positive RAST (radioallergosorbent test) blood test for latex allergy. Severe allergic reactions have occurred from prick and wear tests; epinephrine and resuscitation equipment free of NRL should be available during these procedures.
NRL allergy may be associated with allergic reactions to fruit, especially bananas, chestnuts and avocados. Hyposensitization to NRL is not yet possible, and NRL avoidance and substitution is imperative. Prevention and control of NRL allergy includes latex avoidance in health care settings for affected workers and patients. Substitute synthetic non-NRL gloves should be available, and in many cases low-allergen NRL gloves should be worn by co-workers to accommodate those with NRL allergy, in order to minimize symptoms and to decrease induction of NRL allergy. Continued cooperation among government, industry and health care professionals is necessary to control latex allergy, as discussed in the Health care facilities chapter.
Ergonomics
Ergonomics is the science of assessing the relationship between workers and their work environment. This science includes not only an assessment of musculoskeletal risk due to the design of the work, but also includes a consideration of the cognitive processes involved in work that may lead to human errors.
Jobs in the rubber and tyre industry have been identified with an increased risk of particular types of musculoskeletal disorders. In particular, back injuries appear to be prominent. A sample of materials-handling jobs in the tyre and rubber industry has indicated that the high-risk jobs result in low-back disorder injury rates that are approximately 50% higher than that of general industry. An assessment of jobs indicates that these problems typically arise from jobs requiring the manual transport of rubber products. These jobs include rubber processing (Banbury) operations, tyre builders, tyre finishers and tyre transporters both in the factory and warehouse environment. Wrist problems such as carpal tunnel syndrome and tenosynovitis also appear to be prominent in tyre construction. An examination of tyre manufacturing operations suggests that shoulder problems would be expected. However, as expected, injury records tend to under-report the risk of shoulder injuries due to a lack of sensitivity to the problem. Finally, there appear to be some cognitive processing issues involved in the tyre industry. These are apparent in the inspection tasks and are often exacerbated by poor lighting.
There are several workplace-related risk factors believed to be responsible for these musculoskeletal problems in the tyre and rubber industry. Risk factors consist of static, awkward postures in the back, shoulders and wrists, rapid motions in the wrist and back, and large weights handled, as well as large forces applied to the trunk while handling large pieces of rubber during tyre building. A study of factors associated with low-back disorder risk indicates that greater weight is handled by workers in the tyre building industry than in other fields and these loads are handled at greater than average distances from the body. Furthermore, these forces and weights are often imposed on the body during asymmetric motions of the trunk, such as bending. The duration of the force applications in this type of work is also problematic. Often in a tyre-building operation, lengthy applications of force are required which diminish the worker’s available force over time. Finally, tyre and rubber workplaces are often warm and exposed to dirt and dust. The heat within the workplace will tend to increase the caloric demands of the job, thus increasing the energy demands. Resin and dust within the workplace increase the likelihood that workers will be wearing gloves while performing their tasks. This glove use will increase the required tension in the forearm muscles that control the fingers. In addition, when workers wear gloves they will increase their grip force since they cannot perceive when an object is about to slip out of their hands. Solutions to these ergonomic-related problems include the simple rearrangement of the workplace (e.g., raising or lowering of the work or moving the workstations in order to eliminate large twisting or lateral bending motions of the trunk; the latter can often be accomplished by reorienting origins and destinations of lifting tasks from 180º twists to 90º turns). Often more significant changes are needed. These may range from incorporating adjustable workstations such as scissors jacks or lift tables, to incorporating lifting assistance devices such as lifts and cranes, to fully automating the workstation. There is obviously a large cost associated with some of these solutions to the problem. Therefore the key to proper ergonomic design is to make only the changes that are necessary and to determine the effect of the change in terms of the change in musculoskeletal risk. Fortunately, new methods for quantifying the extent of the risk associated with a given design of the workplace are becoming available. For example, a risk model has been reported that assesses the risk of occupationally related low-back disorder given the demands of the job (Marras et al. 1993; 1995). Models have also been developed that assess the loading of the spine due to dynamic trunk activities (Marras and Sommerich 1991; Granata and Marras 1993). Thus, models are becoming available for the assessment of workplace designs in the industry that are capable of addressing the issue of how much exposure to a workplace is too much.
Environmental and Public Health Issues
All rubber products start out as a “rubber compound”. Rubber compounds start with a rubber polymer, either natural or one of the many synthetic polymers, fillers, plasticizers, anti-oxidants, process aids, activators, accelerators and curatives. Many of the chemical ingredients are classified as hazardous or toxic chemicals, and some may be listed as carcinogens. Handling and processing of these chemicals create both environmental and safety concerns.
Hazardous Waste
Ventilation systems and dust collectors are necessary for workers handling and weighing the rubber chemicals and for workers mixing and processing the uncured rubber compound. Personal protection equipment may also be necessary for these workers. The material collected in the dust collectors must be tested to determine whether it is a hazardous waste. It would be a hazardous waste if it is reactive, corrosive, flammable or contains chemicals that are listed hazardous as wastes.
Hazardous waste must be listed on a manifest and sent for disposal at a hazardous wastesite. Non-hazardous waste can go to local sanitary landfills or may have to go to an industrial landfill, depending on applicable environmental regulations.
Air Pollution
Some rubber products require a rubber cement application in the manufacturing process. Rubber cements are made by mixing the uncured rubber compound with a solvent. The solvents used in this process are usually classified as volatile organic compounds (VOCs). Processes that use VOCs must have some type of emission-control equipment. This equipment can be a solvent recovery system or a thermal oxidizer. A thermal oxidizer is an incineration system that destroys the VOCs by combustion and usually requires a fuel supplement such as natural gas. Without emission control equipment the VOCs can cause health concerns in the factory and in the community. If the VOCs are photochemically reactive, they will affect the ozone layer.
When rubber parts are cured and the curing vessel is opened, curing fumes rush out of the vessel and from the rubber part. These fumes will be in the form of smoke, steam or both. Curing fumes can carry unreacted chemicals, plasticizers, mould lubes and other materials out into the atmosphere. Emission controls are needed.
Ground and Water Pollution
Storage and handling of VOCs must be done with extreme caution. In past years, VOCs were stored in underground storage tanks, which in some cases resulted in leaks or spills. Leaks and/or spills around underground storage tanks generally result in soil and groundwater contamination, which triggers expensive soil and groundwater remediation. The best storage choice is above-ground tanks with good secondary containment for spill prevention.
Waste Rubber
Every manufacturing process has process and finished goods scrap. Some of the process scrap can be reprocessed in the intended product or other product processes. However, once the rubber is cured or vulcanized, it can no longer be reprocessed. All cured process and finished goods scrap becomes waste material. Disposal of scrap or waste rubber products has become a worldwide problem.
Every household and business in the world uses some type of rubber product. Most rubber products are classified as non-hazardous materials and therefore would be non-hazardous waste. However, rubber products such as tyres, hose and other tubular products create an environmental problem as related to disposal after their useful life.
Tyres and tubular products cannot be buried in a landfill because the void areas trap air, which causes the products to rise to the surface over time. Shredding the rubber products eliminates this problem; however, shredding requires special equipment and is very expensive.
Smoldering tyre fires can generate large amounts of irritating smoke that can contain a wide variety of toxic chemicals and particulates.
Incineration of Scrap Rubber
One of the options for disposing of scrap rubber products and process scrap rubber from the manufacturing processes is incineration. Incineration might initially seem to be the best solution for disposal of the numerous “worn out” rubber products that exist in the world today. Some rubber-manufacturing companies have looked at incineration as a means of disposing of scrap rubber parts as well as cured and uncured rubber-process scrap. In theory, the rubber could be burned to generate steam that could be used back in the factory.
Unfortunately, it is not that simple. The incinerator must be designed so as to handle air emissions and would most likely require scrubbers to remove such contaminants as chlorine. Chlorine emissions generally would come from burning products and scrap that contain chloroprene polymers. The scrubbers generate an acidic discharge that may have to be neutralized prior to discharge.
Almost all rubber compounds contain some type of fillers, either carbon blacks, clays, calcium carbonates or hydrated silica compounds. When these rubber compounds are burned, they generate ash equivalent to the filler loading in the rubber compound. The ash is collected either by wet scrubbers or dry scrubbers. Both methods must be analysed for heavy metals prior to disposal. Wet scrubbers most likely will produce a wastewater that contains 10 to 50 ppm zinc. This much zinc being discharged into a sewage system will create problems at the treatment plant. If this occurs, then a treatment system for the removal of zinc must be installed. This treatment system then generates a zinc-containing sludge that must be shipped out for disposal.
Dry scrubbers generate an ash that must be collected for disposal. Both wet and dry ash is difficult to handle, and disposal can be a problem since most landfills do not accept this type of waste. Both wet and dry ash can be very alkaline if the rubber compounds being burned are heavily loaded with calcium carbonate.
Finally, the amount of steam generated is not enough to supply the full amount necessary to operate a rubber-manufacturing facility. The scrap rubber supply is inconsistent, and efforts are currently underway to reduce scrap, which would reduce the fuel supply. The maintenance cost of an incinerator designed to burn rubber scrap and rubber products is also very high.
When all of these costs are taken into consideration, incineration of scrap rubber may be the least cost-effective method of disposal.
Conclusion
Perhaps the best solution to environmental and health concerns associated with manufacturing rubber products would be good engineering control for producing and compounding powdered chemicals used in rubber compounds, and recycling programmes for all uncured and cured rubber process scrap and products. The powdered chemicals collected in dust-collector systems could be added back to rubber compounds with the appropriate engineering controls, which would eliminate the landfilling of these chemicals.
Controlling the environmental and health issues in the rubber industry can be done, but it will not come easy or be free. The cost associated with controlling environmental and health problems must be added back to the cost of rubber products.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."